IB MYP Integrated Science- Chemistry- Radioactivity and decay-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Radioactivity and decay -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Radioactivity and decay -Study Notes -As per latest Syllabus.
Key Concepts:
Radioactivity and decay
IB MYP Integrated Science -Concise Summary Notes- All Topics
Radioactivity and Decay
Radioactivity is the spontaneous emission of radiation from unstable atomic nuclei. These unstable nuclei undergo radioactive decay to become more stable.
Why Do Nuclei Decay?
An atom becomes unstable when there is an imbalance between protons and neutrons in the nucleus. To achieve stability, it releases energy and particles.
Types of Radioactive Decay
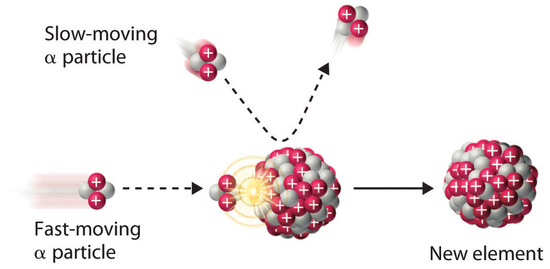
1. Alpha (α) Decay
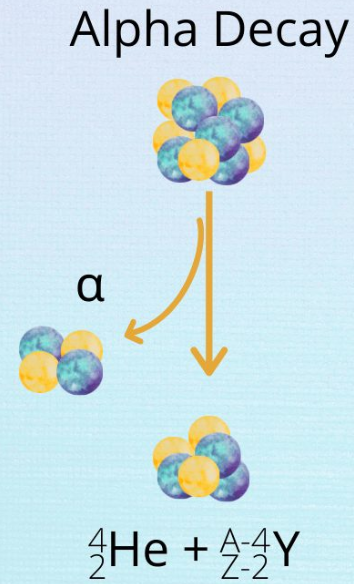
\( \mathrm{^{A}_{Z}X \rightarrow ^{A-4}_{Z-2}Y + ^{4}_{2}He} \)
- Emits a helium nucleus
- Mass number decreases by 4
- Atomic number decreases by 2
- Low penetration, highly ionising
2. Beta (β) Decay
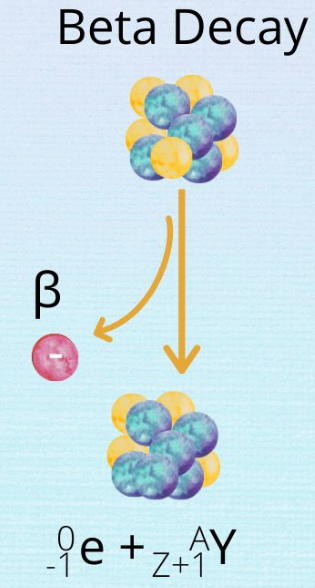
\( \mathrm{^{A}_{Z}X \rightarrow ^{A}_{Z+1}Y + ^{0}_{-1}e} \)
- A neutron changes into a proton and electron
- Mass number remains the same
- Atomic number increases by 1
- Medium penetration
3. Gamma (γ) Radiation
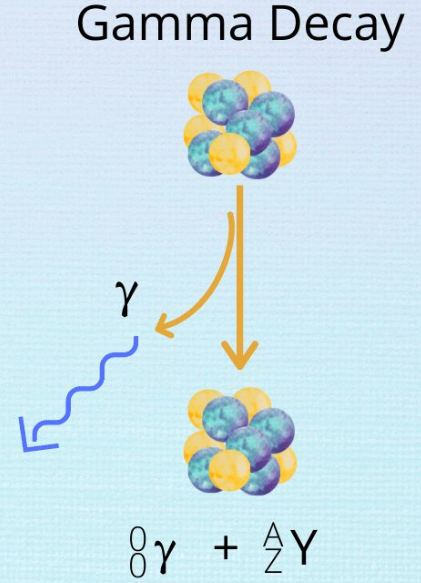
\( \mathrm{^{A}_{Z}X^* \rightarrow ^{A}_{Z}X + \gamma} \)
- No change in mass or atomic number
- Releases excess energy
- Highly penetrating
Half-Life
Half-life is the time taken for half of the radioactive nuclei in a sample to decay.
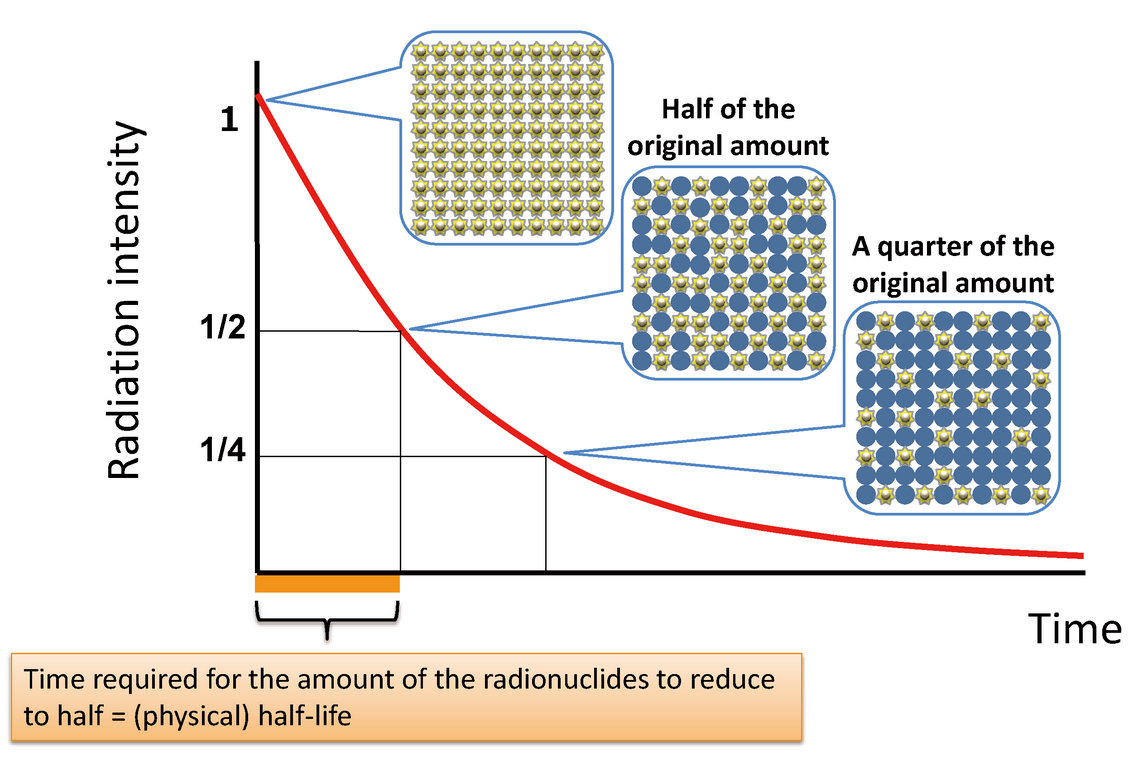
\( \mathrm{N = N_0 \left(\frac{1}{2}\right)^n} \)
Where \( n \) is the number of half-lives.
Random Nature of Decay
Radioactive decay is random and cannot be predicted for a single atom, but it follows predictable patterns for large numbers of atoms.
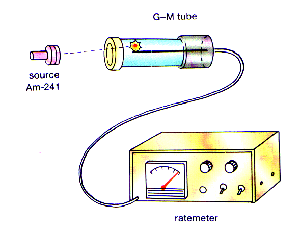
Detection of Radiation
| Uses of Radioactivity
| Hazards of Radiation
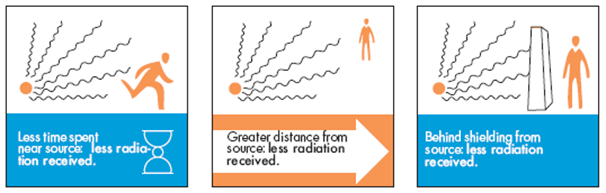
| Safety Precautions
|
Real-World Importance
- Used in medicine and industry
- Helps determine age of fossils and rocks
- Important for energy production
Example 1:
What happens to the atomic and mass numbers during alpha decay?
▶️ Answer/Explanation
Mass number decreases by 4
Atomic number decreases by 2
Conclusion: Alpha decay reduces both numbers.
Example 2:
A radioactive sample has a half-life of 5 years. If the initial mass is 40 g, what remains after 10 years?
▶️ Answer/Explanation
10 years = 2 half-lives
\( 40 \rightarrow 20 \rightarrow 10 \)
Conclusion: 10 g remains.
Example 3:
A nucleus undergoes beta decay followed by gamma emission. Describe the changes in atomic number and mass number.
▶️ Answer/Explanation
Step 1: Beta decay
Atomic number increases by 1
Mass number unchanged
Step 2: Gamma emission
No change in atomic or mass number
Conclusion: Overall, atomic number increases by 1 while mass number remains the same.