IB MYP Integrated Science- Chemistry- Reaction kinetics-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Reaction kinetics -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Reaction kinetics -Study Notes -As per latest Syllabus.
Key Concepts:
Reaction kinetics (energy changes, enzymes, rates, factors affecting rates/collision theory)
IB MYP Integrated Science -Concise Summary Notes- All Topics
Reaction Kinetics (Energy Changes, Rates, Collision Theory, Enzymes)
Reaction kinetics is the study of how fast chemical reactions occur and the factors that influence their speed. It also explains the energy changes involved when bonds are broken and formed during reactions.
Energy Changes in Reactions
Chemical reactions involve two key processes:
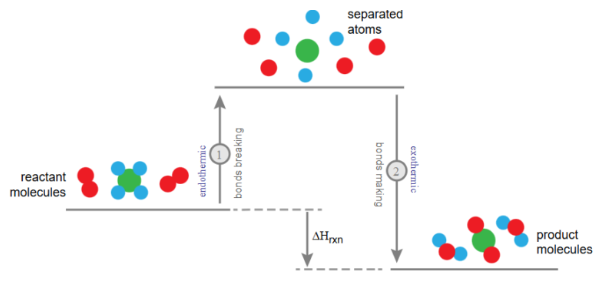
- Breaking bonds (requires energy)
- Forming bonds (releases energy)
The overall energy change depends on the balance between these processes.
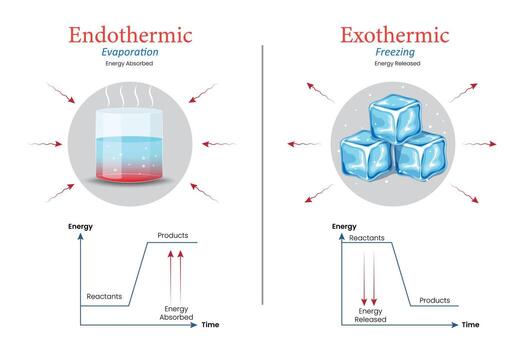
- Exothermic reactions: release more energy than they absorb
- Endothermic reactions: absorb more energy than they release
\( \mathrm{Exothermic: \text{energy released} > \text{energy absorbed}} \)
\( \mathrm{Endothermic: \text{energy absorbed} > \text{energy released}} \)
Activation Energy
The activation energy is the minimum energy required for reactant particles to successfully collide and react.
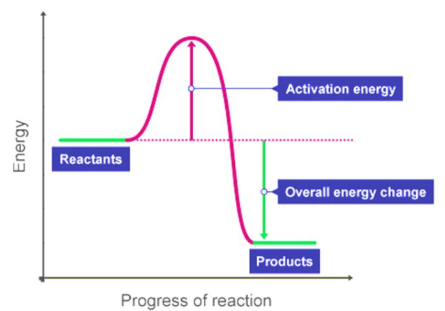
\( \mathrm{Reactants \xrightarrow{E_a} Products} \)
Even in exothermic reactions, activation energy must be supplied initially.
Energy Profile Diagrams
Energy profile diagrams show how energy changes during a reaction:
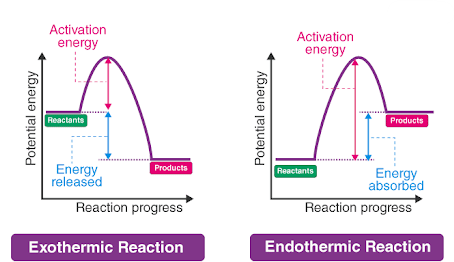
- Peak represents activation energy
- Difference between reactants and products shows energy change
- Exothermic → products lower energy
- Endothermic → products higher energy
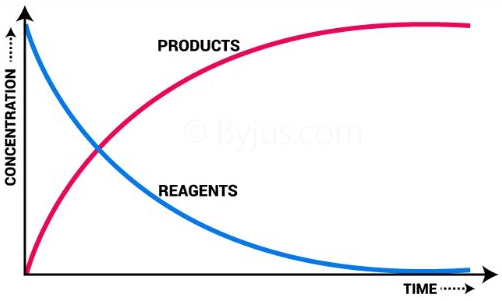
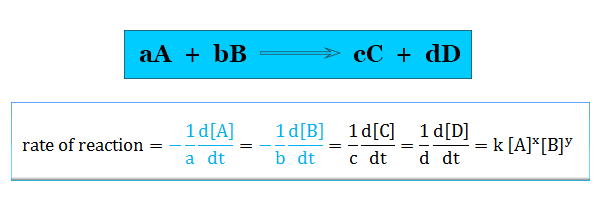
Rate of Reaction
The rate of reaction measures how quickly reactants are used up or products are formed.
\( \mathrm{Rate = \frac{change\ in\ amount}{time}} \)
Rates can be measured by:
- Volume of gas produced
- Mass loss
- Change in concentration
Collision Theory
Collision theory explains that reactions occur when particles collide:
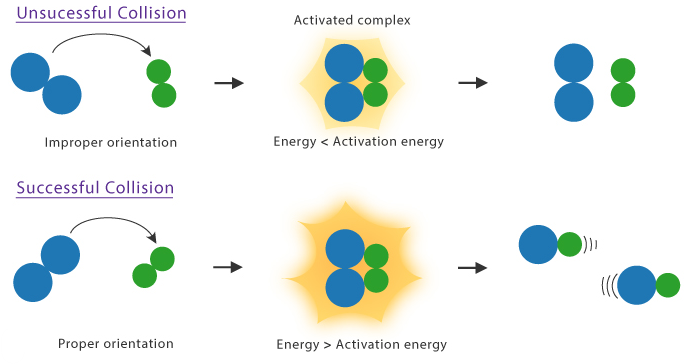
- Particles must collide
- Collisions must have energy ≥ activation energy
- Correct orientation is required
Only successful collisions lead to a reaction.
Factors Affecting Rate of Reaction
Temperature
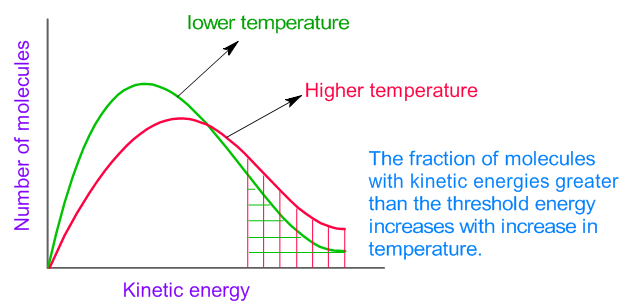
Higher temperature → more kinetic energy → more frequent and energetic collisions
Concentration / Pressure
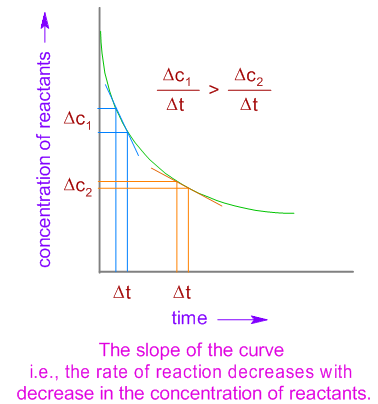
Higher concentration → more particles → higher collision frequency
Surface Area
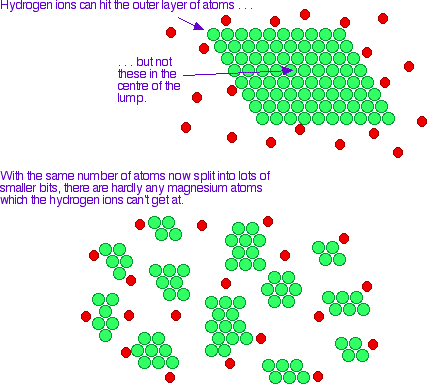
Smaller particles → greater surface area → more exposed particles for collisions
Catalysts
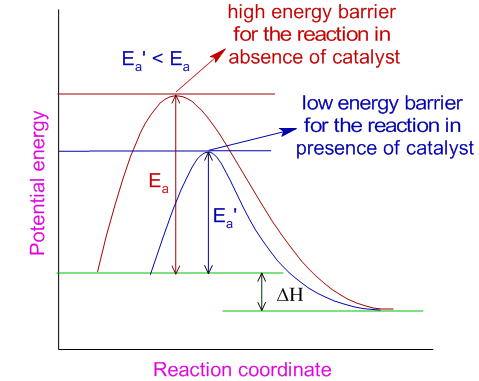
Catalysts lower activation energy and provide an alternative reaction pathway
Enzymes (Biological Catalysts)
Enzymes are highly specific protein catalysts that control biochemical reactions in living organisms.
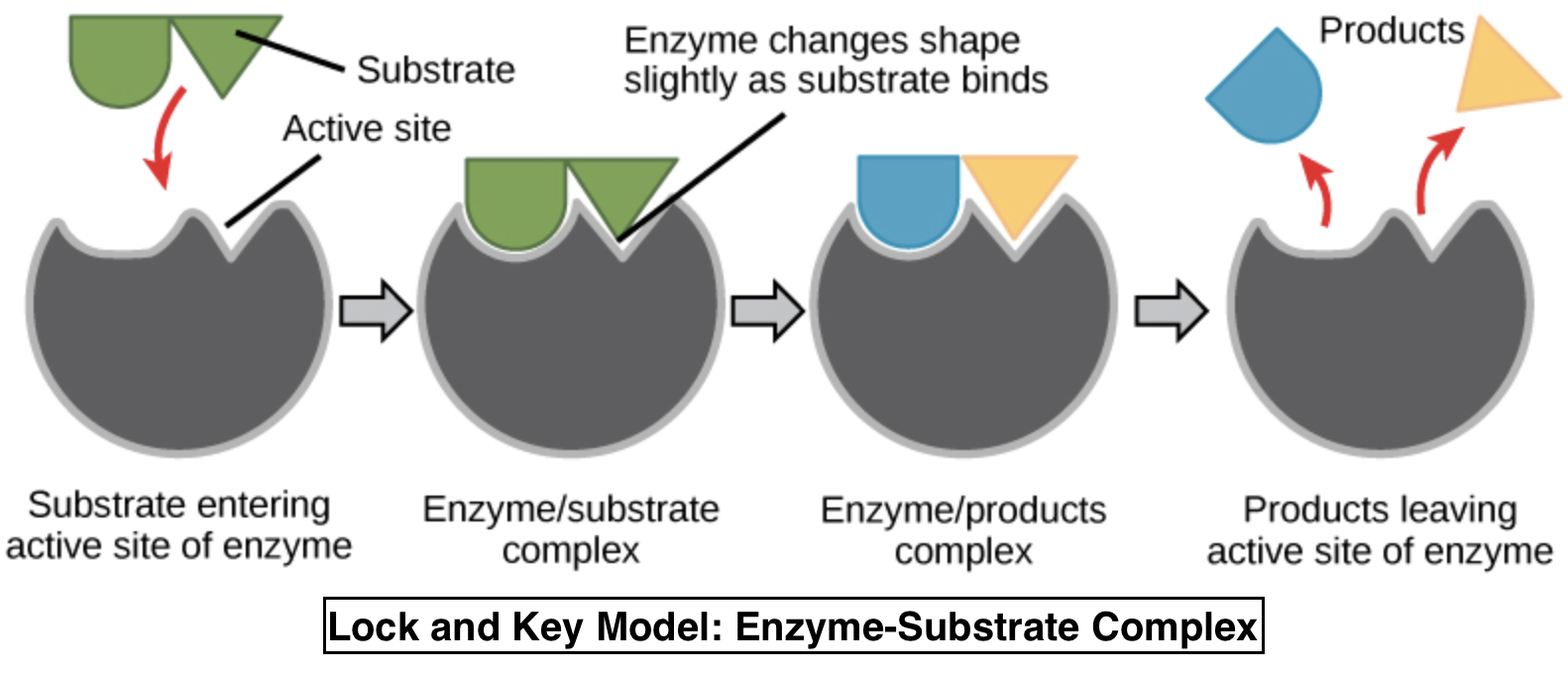
- Each enzyme has an active site where the substrate binds
- Forms an enzyme-substrate complex
- Reaction occurs → products released
This is often described using the lock-and-key model.
Effect of Temperature and pH on Enzymes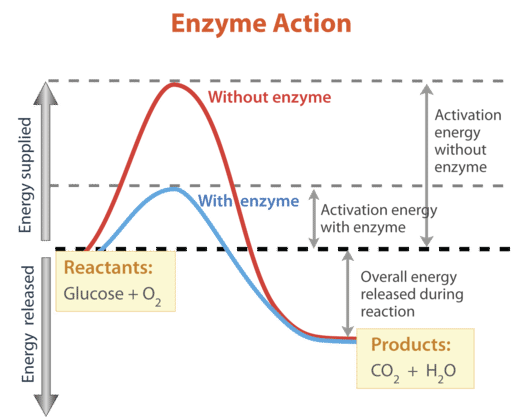
- Low temperature → slower reaction
- Optimum temperature → fastest rate
- High temperature → enzyme denatures (loses shape)
- pH affects shape of active site
Real-World Importance
- Enzymes control digestion and metabolism in the body
- Catalysts are used in industrial processes to increase efficiency
- Reaction rates affect food spoilage and preservation
Example 1:
Explain why increasing temperature increases the rate of reaction using collision theory.
▶️ Answer/Explanation
Particles gain kinetic energy
Move faster → more collisions
More particles exceed activation energy
Conclusion: Increased successful collisions increase reaction rate.
Example 2:
Describe how enzymes speed up reactions.
▶️ Answer/Explanation
Substrate binds to active site
Forms enzyme-substrate complex
Lowers activation energy
Conclusion: Enzymes increase reaction rate without being used up.
Example 3 :
A reaction is carried out at different temperatures. The rate increases rapidly at first but then decreases at very high temperatures.
Explain this observation.
▶️ Answer/Explanation
Step 1: Increase in rate
Higher temperature → more kinetic energy → more collisions
Step 2: Decrease in rate
Enzyme denatures at high temperature
Conclusion: Rate increases initially but decreases due to enzyme denaturation.
