IB MYP Integrated Science- Chemistry- Reactivity series of metals-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Reactivity series of metals -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Reactivity series of metals -Study Notes -As per latest Syllabus.
Key Concepts:
Reactivity series of metals
IB MYP Integrated Science -Concise Summary Notes- All Topics
Reactivity Series of Metals
The reactivity series is a list of metals arranged in order of their reactivity, from most reactive to least reactive. It helps predict how metals will react and how they can be extracted from their ores.
The Reactivity Series 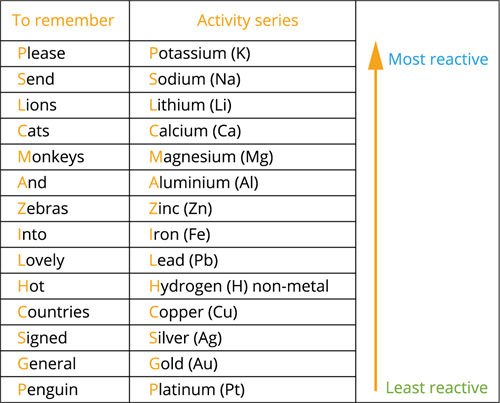
\( \mathrm{K,\ Na,\ Ca,\ Mg,\ Al,\ Zn,\ Fe,\ Cu,\ Ag,\ Au} \)
Metals at the top are highly reactive, while those at the bottom are less reactive.
Key Features of the Reactivity Series
- More reactive metals lose electrons more easily
- Less reactive metals are more stable
- Reactivity is linked to electron configuration
Reactions with Water
- Very reactive metals (K, Na, Ca) react vigorously with cold water
- Moderately reactive metals react with steam
- Less reactive metals do not react
Example:
\( \mathrm{2Na + 2H_2O \rightarrow 2NaOH + H_2} \)
Reactions with Acids
Metals above hydrogen react with acids to produce hydrogen gas.
\( \mathrm{Metal + Acid \rightarrow Salt + H_2} \)
Example:
\( \mathrm{Zn + 2HCl \rightarrow ZnCl_2 + H_2} \)
Displacement Reactions
A more reactive metal can displace a less reactive metal from its compound.
\( \mathrm{Zn + CuSO_4 \rightarrow ZnSO_4 + Cu} \)
Extraction and the Reactivity Series
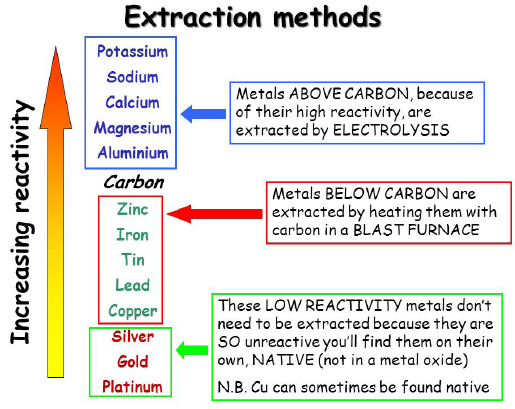
- Top metals → extracted by electrolysis
- Middle metals → extracted using carbon
- Bottom metals → found native
Corrosion and Reactivity
More reactive metals corrode more easily.
- Iron rusts in presence of water and oxygen
- Less reactive metals like gold do not corrode
Link to Electron Loss
Reactivity depends on how easily a metal loses electrons:
\( \mathrm{Metal \rightarrow Metal^{+} + e^-} \)
Real-World Applications
- Predicting reactions in industry
- Preventing corrosion (galvanising)
- Designing extraction methods
Example 1:
Explain why copper does not react with dilute hydrochloric acid.
▶️ Answer/Explanation
Copper is below hydrogen in the reactivity series
Cannot displace hydrogen
Conclusion: No reaction occurs.
Example 2:
Predict the result when iron is added to copper sulfate solution.
▶️ Answer/Explanation
Iron is more reactive than copper
Displacement occurs
\( \mathrm{Fe + CuSO_4 \rightarrow FeSO_4 + Cu} \)
Conclusion: Copper is displaced.
Example 3 :
A metal X reacts with water but does not react with dilute acid. Suggest its position in the reactivity series and explain.
▶️ Answer/Explanation
Step 1: Reaction with water
Indicates metal is quite reactive
Step 2: No reaction with acid
Unusual — suggests limited conditions or protective layer
Conclusion: Metal X is likely high in the reactivity series but may form a protective oxide layer affecting acid reaction.
