IB MYP Integrated Science- Chemistry- States and properties of matter-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – States and properties of matter -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – States and properties of matter -Study Notes -As per latest Syllabus.
Key Concepts:
States and properties of matter
IB MYP Integrated Science -Concise Summary Notes- All Topics
States and Properties of Matter
Matter exists in different states depending on the arrangement, movement, and energy of its particles. The three main states are solids, liquids, and gases, and each has distinct physical properties explained by the particle model
States of Matter Overview
The properties of each state depend on:
- Arrangement of particles
- Movement of particles
- Forces of attraction between particles
Solids
| Liquids
| Gases
|
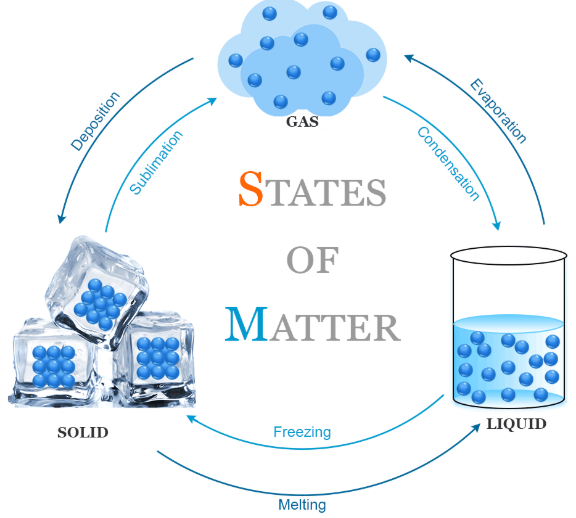
Changes of State
Changes of state occur when energy is transferred, affecting particle movement and spacing.
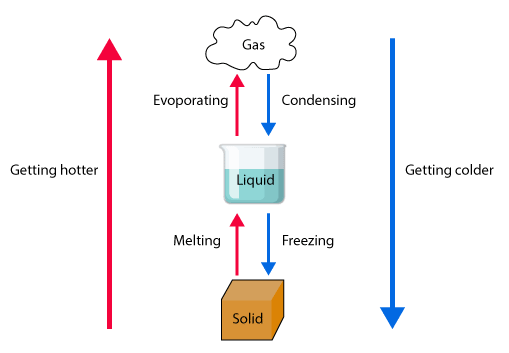
- Melting: solid → liquid
- Boiling/Evaporation: liquid → gas
- Condensation: gas → liquid
- Freezing: liquid → solid
- Sublimation: solid → gas (without liquid stage)
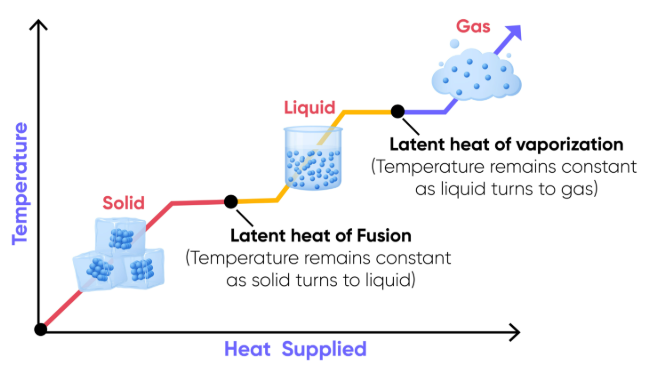
Heating and Cooling Curves
During changes of state, temperature remains constant while energy is used to break or form intermolecular forces.
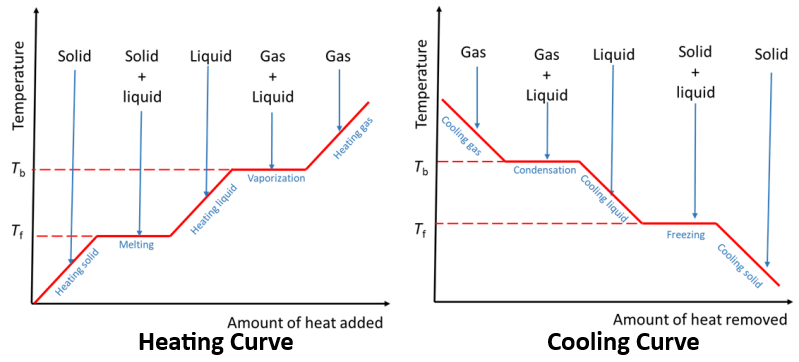
Latent Heat
Latent heat is the energy required to change the state of a substance without changing its temperature.
Effect of Temperature
Increasing temperature increases particle kinetic energy:
\( \mathrm{Higher\ temperature \rightarrow faster\ particles} \)
This leads to expansion and changes of state.
Real-World Applications
- Refrigeration uses evaporation and condensation
- Boiling is used in cooking and sterilisation
- Sublimation is used in dry ice and air fresheners
Example 1:
Explain why liquids can flow but solids cannot.
▶️ Answer/Explanation
Liquid particles can move past each other
Solid particles are fixed in position
Conclusion: Liquids flow due to mobile particles.
Example 2:
Explain why gases have low density.
▶️ Answer/Explanation
Particles are far apart
Large empty spaces
Conclusion: Low particle density leads to low mass per volume.
Example 3:
A substance is heated from solid to gas.
Explain the changes in particle arrangement and energy during this process.
▶️ Answer/Explanation
Step 1: Solid → Liquid
Particles gain energy and break fixed positions
Step 2: Liquid → Gas
Particles gain more energy and move far apart
Step 3: Energy changes
Energy used to overcome intermolecular forces
Conclusion: Increased energy leads to greater particle movement and separation.