IB MYP Integrated Science- Chemistry- Trends, periods, groups-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Trends, periods, groups -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Trends, periods, groups -Study Notes -As per latest Syllabus.
Key Concepts:
Trends, periods, groups
IB MYP Integrated Science -Concise Summary Notes- All Topics
Periodic Table (Trends, Periods, and Groups)
The periodic table is a systematic arrangement of elements in order of increasing atomic number. It allows scientists to identify patterns (trends) in properties and predict the behavior of elements.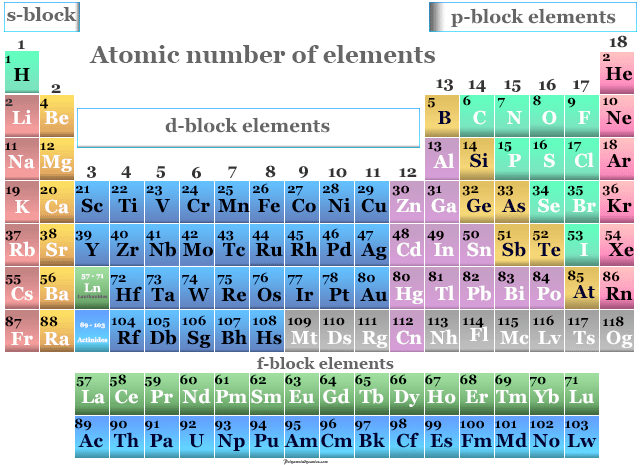
Structure of the Periodic Table
The periodic table is organised into periods (horizontal rows) and groups (vertical columns).
Periods
- Horizontal rows in the periodic table
- Elements in the same period have the same number of electron shells
- Atomic number increases from left to right
Groups
- Vertical columns in the periodic table
- Elements in the same group have the same number of valence electrons
- Similar chemical properties and reactivity
Key Groups
- Group 1: Alkali metals (very reactive metals)
- Group 7: Halogens (reactive non-metals)
- Group 0/18: Noble gases (very unreactive)
Trends Across a Period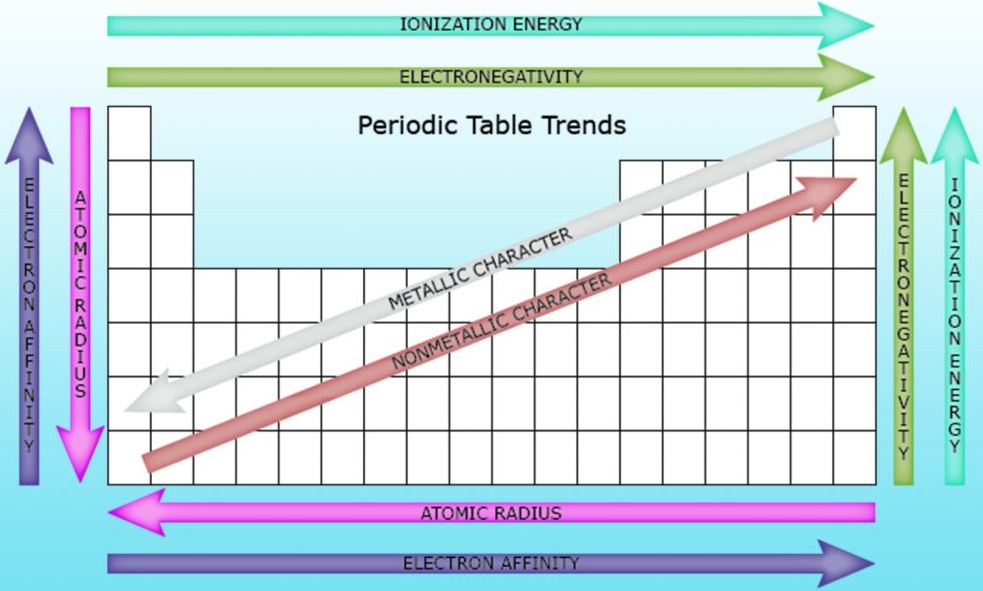
As you move from left to right across a period:
- Atomic number increases
- Number of valence electrons increases
- Atomic radius decreases
- Reactivity of metals decreases, non-metals increases
Trends Down a Group
As you move down a group:
- Number of shells increases
- Atomic radius increases
- Reactivity changes depending on group
Group Trends Examples
Group 1 (Alkali Metals)

- Reactivity increases down the group
- Atoms lose electrons more easily
Group 7 (Halogens)
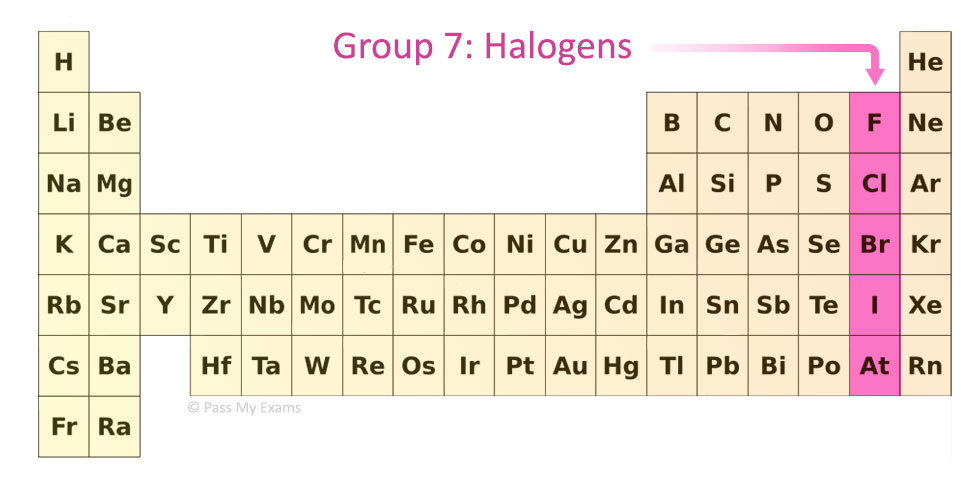
- Reactivity decreases down the group
- Atoms gain electrons less easily
Group 0 (Noble Gases)
![]()
- Very low reactivity
- Full outer electron shell
Link to Electron Configuration
- Group number → number of valence electrons
- Period number → number of electron shells
Metals and Non-Metals
- Metals are on the left side
- Non-metals are on the right side
- Metalloids lie in between
Real-World Importance
- Predicting reactivity of elements
- Designing materials and chemicals
- Understanding chemical behavior in industry and biology
Example 1:
Explain why elements in the same group have similar chemical properties.
▶️ Answer/Explanation
They have the same number of valence electrons
Chemical reactions involve outer electrons
Conclusion: Same valence electrons → similar reactivity.
Example 2:
Explain why atomic radius decreases across a period.
▶️ Answer/Explanation
More protons increase nuclear attraction
Electrons are pulled closer
Conclusion: Stronger attraction reduces atomic size.
Example 3:
Compare the reactivity of sodium and potassium using periodic trends.
▶️ Answer/Explanation
Step 1: Position
Both are in Group 1
Step 2: Down the group
Potassium has more shells
Step 3: Attraction
Outer electron is further from nucleus → weaker attraction
Conclusion: Potassium is more reactive than sodium.
