IB MYP Integrated Science- Chemistry- Types of fuels-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Types of fuels -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Types of fuels -Study Notes -As per latest Syllabus.
Key Concepts:
Types of fuels
IB MYP Integrated Science -Concise Summary Notes- All Topics
Types of Fuels
Fuels are substances that release energy when they undergo combustion (burning). This energy is used for heating, electricity generation, and transportation.
What is Combustion?
Combustion is a chemical reaction between a fuel and oxygen that releases energy in the form of heat and often light.

\( \mathrm{Fuel + O_2 \rightarrow CO_2 + H_2O + energy} \)
Classification of Fuels
Fuels can be classified based on their physical state and source.
1. Solid Fuels
| 2. Liquid Fuels
| 3. Gaseous Fuels
|
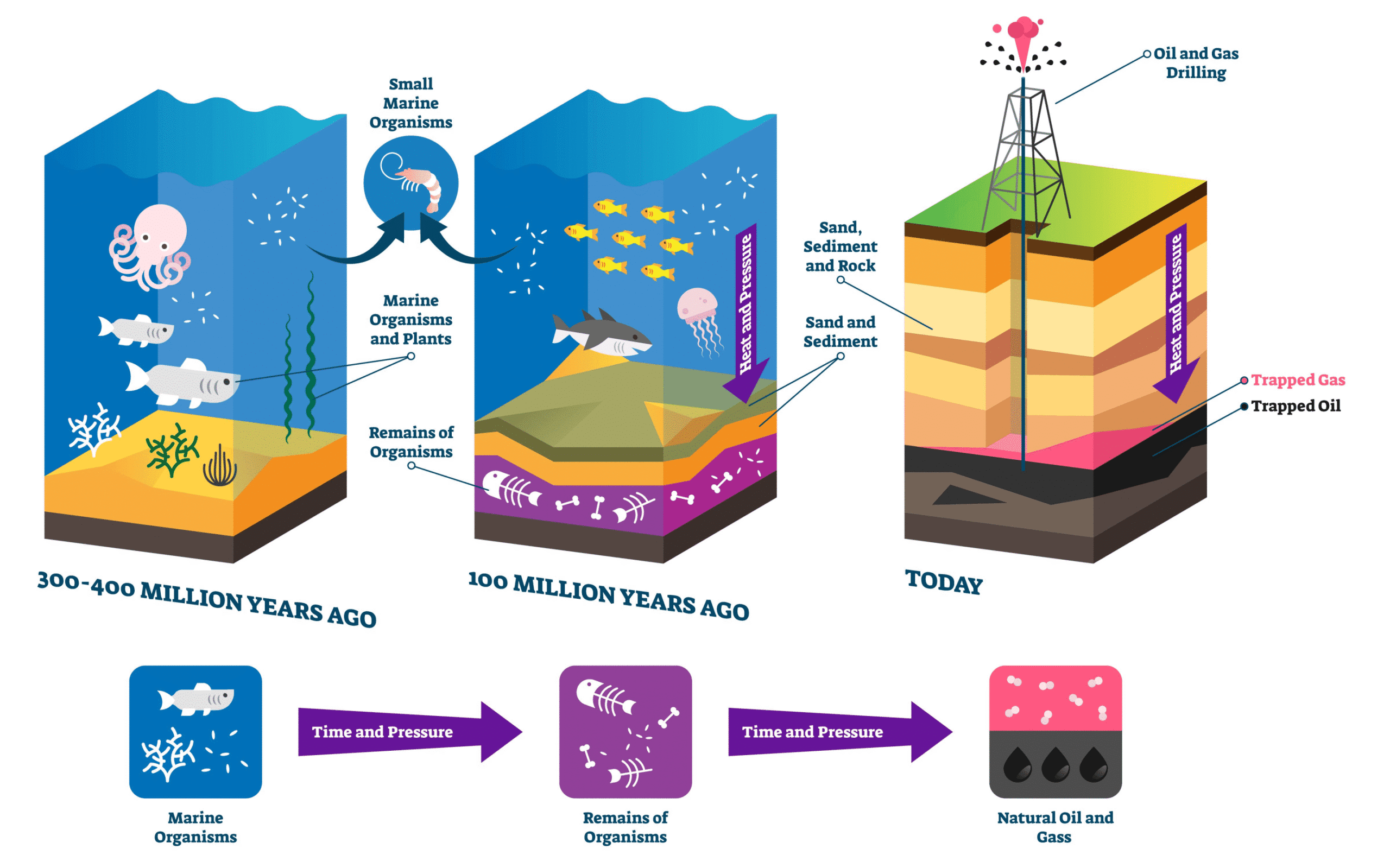
Renewable and Non-Renewable Fuels
Non-renewable fuels
| Renewable fuels
|
Characteristics of a Good Fuel
- High energy output (high calorific value)
- Easy to store and transport
- Burns cleanly with minimal pollution
- Safe and economical
Environmental Impact
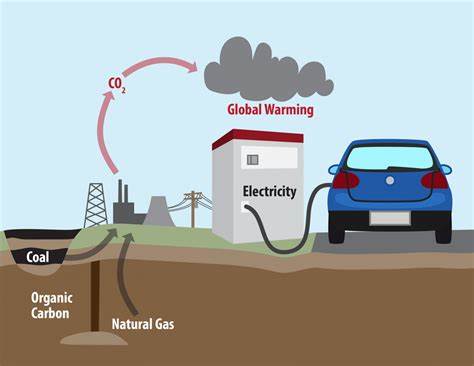
- Burning fossil fuels produces \( \mathrm{CO_2} \), contributing to climate change
- Incomplete combustion produces carbon monoxide (CO), a toxic gas
- Air pollution from smoke and particulates
Complete vs Incomplete Combustion
Complete combustion (excess oxygen):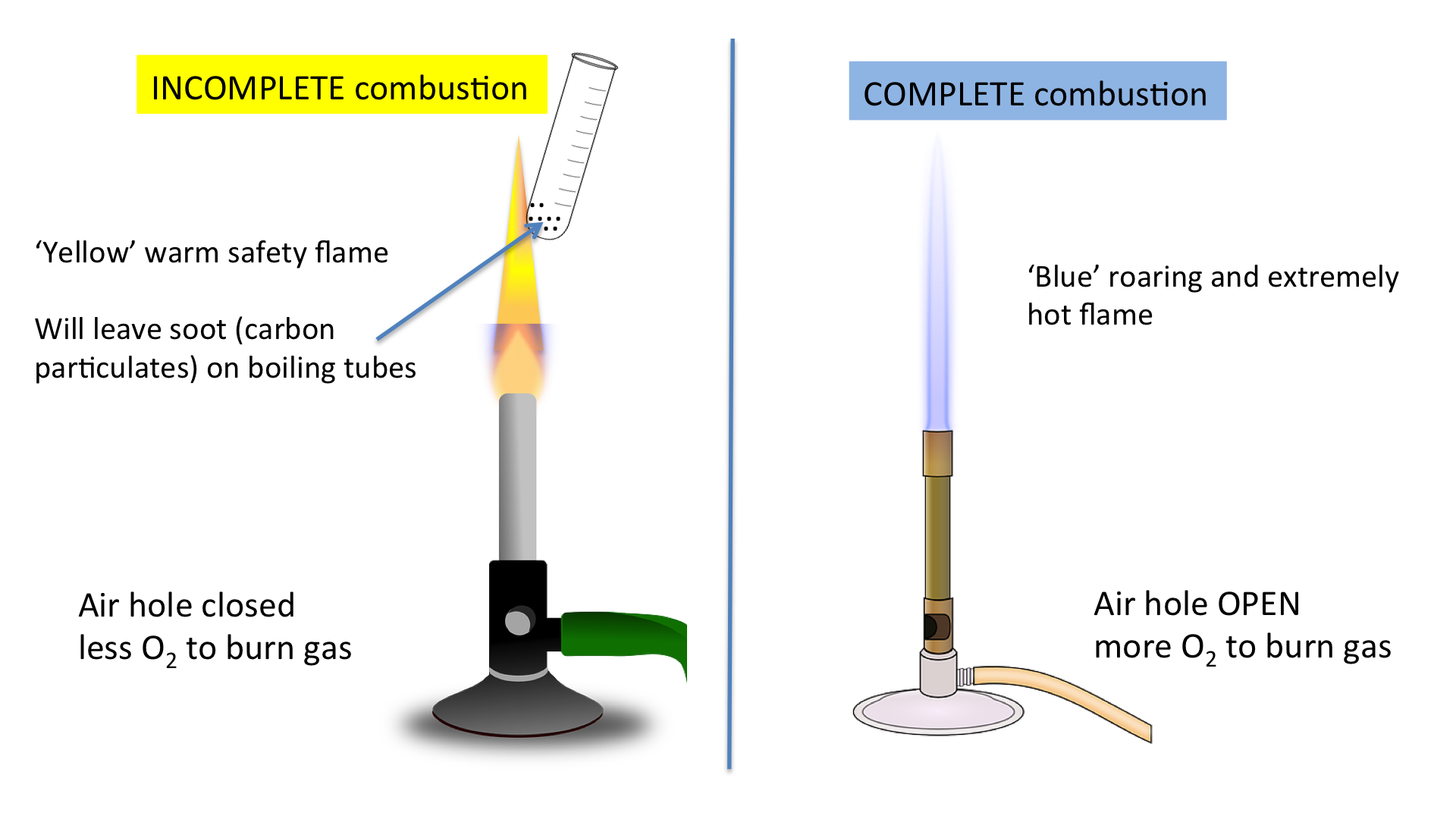
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Incomplete combustion (limited oxygen):
\( \mathrm{CH_4 + O_2 \rightarrow CO + H_2O} \)
Real-World Applications
- Fuels power vehicles and industries
- Used for cooking and heating
- Electricity generation in power plants
Example 1:
Why are gaseous fuels considered more efficient than solid fuels?
▶️ Answer/Explanation
They burn more completely
Produce less smoke and residue
Conclusion: Cleaner and more efficient combustion.
Example 2:
Explain the difference between renewable and non-renewable fuels.
▶️ Answer/Explanation
Renewable fuels can be replenished quickly
Non-renewable fuels take millions of years to form
Conclusion: Renewable fuels are more sustainable.
Example 3 :
Explain why incomplete combustion is dangerous and how it can be prevented.
▶️ Answer/Explanation
Step 1: Problem
Produces carbon monoxide (CO)
Step 2: Danger
CO is toxic and prevents oxygen transport in blood
Step 3: Prevention
Ensure sufficient oxygen supply
Use proper ventilation
Conclusion: Complete combustion is safer and more efficient.