IB MYP Integrated Science- Chemistry- Word and chemical reactions and formulas-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Word and chemical reactions and formulas -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Word and chemical reactions and formulas -Study Notes -As per latest Syllabus.
Key Concepts:
Word and chemical reactions and formulas
IB MYP Integrated Science -Concise Summary Notes- All Topics
Word and Chemical Equations (Reactions and Formulae)
A chemical reaction is a process in which substances (reactants) are converted into new substances (products). These reactions can be represented using word equations and chemical equations.
Word Equations
A word equation shows the names of the reactants and products.
Reactant + Reactant → Product + Product
Example:
Magnesium + Oxygen → Magnesium oxide
Chemical Equations
Chemical equations use symbols and formulae to represent reactions.
\( \mathrm{Mg + O_2 \rightarrow MgO} \)
Balancing Chemical Equations
Chemical equations must be balanced to obey the law of conservation of mass, which states that atoms are neither created nor destroyed in a reaction.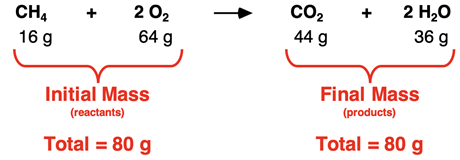
Balanced equation:
\( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)
The number of each type of atom must be the same on both sides.
State Symbols
State symbols show the physical state of substances:
- \( \mathrm{(s)} \) solid
- \( \mathrm{(l)} \) liquid
- \( \mathrm{(g)} \) gas
- \( \mathrm{(aq)} \) aqueous (dissolved in water)
Example:
\( \mathrm{2H_2(g) + O_2(g) \rightarrow 2H_2O(l)} \)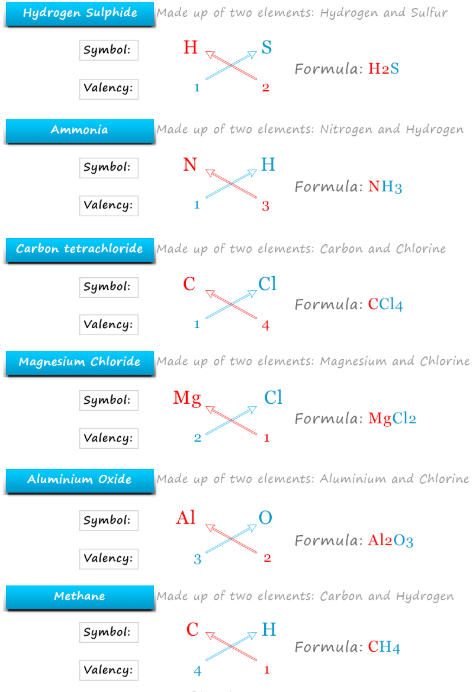
Types of Chemical Reactions
- Combination: two substances combine to form one product
- Decomposition: one substance breaks down into simpler substances
- Displacement: one element replaces another
- Combustion: reaction with oxygen producing energy
Writing Chemical Formulae
Chemical formulae show the ratio of atoms in a compound.
For ionic compounds, charges must balance:
\( \mathrm{Na^+ + Cl^- \rightarrow NaCl} \)
\( \mathrm{Ca^{2+} + 2Cl^- \rightarrow CaCl_2} \)
Real-World Importance
Chemical reactions are essential in industries, energy production, and biological processes such as respiration.
Example 1:
Write the word and balanced chemical equation for the reaction between hydrogen and oxygen.
▶️ Answer/Explanation
Word equation:
Hydrogen + Oxygen → Water
Chemical equation:
\( \mathrm{2H_2 + O_2 \rightarrow 2H_2O} \)
Conclusion: The equation is balanced with equal atoms on both sides.
Example 2:
Balance the equation: \( \mathrm{Fe + O_2 \rightarrow Fe_2O_3} \)
▶️ Answer/Explanation
Balanced equation:
\( \mathrm{4Fe + 3O_2 \rightarrow 2Fe_2O_3} \)
Explanation: Adjust coefficients so Fe and O atoms are equal on both sides.
Example 3:
Calcium reacts with water to form calcium hydroxide and hydrogen gas.
a) Write the word equation
b) Write the balanced chemical equation with state symbols
▶️ Answer/Explanation
Word equation:
Calcium + Water → Calcium hydroxide + Hydrogen
Chemical equation:
\( \mathrm{Ca(s) + 2H_2O(l) \rightarrow Ca(OH)_2(aq) + H_2(g)} \)
Explanation: Coefficients are adjusted to balance hydrogen and oxygen atoms.
Conclusion: Balanced equation follows conservation of mass.
Types of Chemical Reactions
Chemical reactions can be classified into different types based on how reactants are transformed into products. Understanding these types helps predict products and recognise reaction patterns.
Combination (Synthesis) Reactions
In a combination reaction, two or more substances combine to form a single product.

\( \mathrm{A + B \rightarrow AB} \)
Example:
\( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)
Decomposition Reactions
In a decomposition reaction, one compound breaks down into simpler substances, often using heat or electricity.

\( \mathrm{AB \rightarrow A + B} \)
Example:
\( \mathrm{CaCO_3 \rightarrow CaO + CO_2} \)
Displacement Reactions
In a displacement reaction, a more reactive element replaces a less reactive element in a compound.
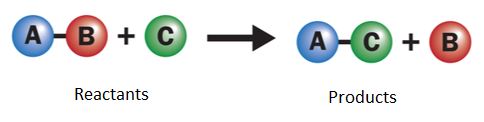
\( \mathrm{A + BC \rightarrow AC + B} \)
Example:
\( \mathrm{Zn + CuSO_4 \rightarrow ZnSO_4 + Cu} \)
Double Displacement (Precipitation) Reactions
In this reaction, ions in two compounds exchange partners to form two new compounds. Often, a solid (precipitate) is formed.
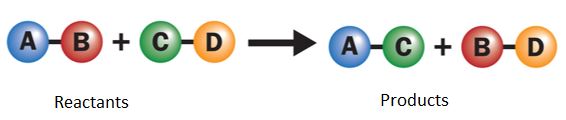
\( \mathrm{AB + CD \rightarrow AD + CB} \)
Example:
\( \mathrm{AgNO_3 + NaCl \rightarrow AgCl(s) + NaNO_3} \)
Combustion Reactions
Combustion reactions occur when a substance reacts with oxygen, releasing energy (usually heat and light).

For hydrocarbons:
\( \mathrm{Hydrocarbon + O_2 \rightarrow CO_2 + H_2O} \)
Example:
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Neutralisation Reactions
A neutralisation reaction occurs when an acid reacts with a base to form salt and water.
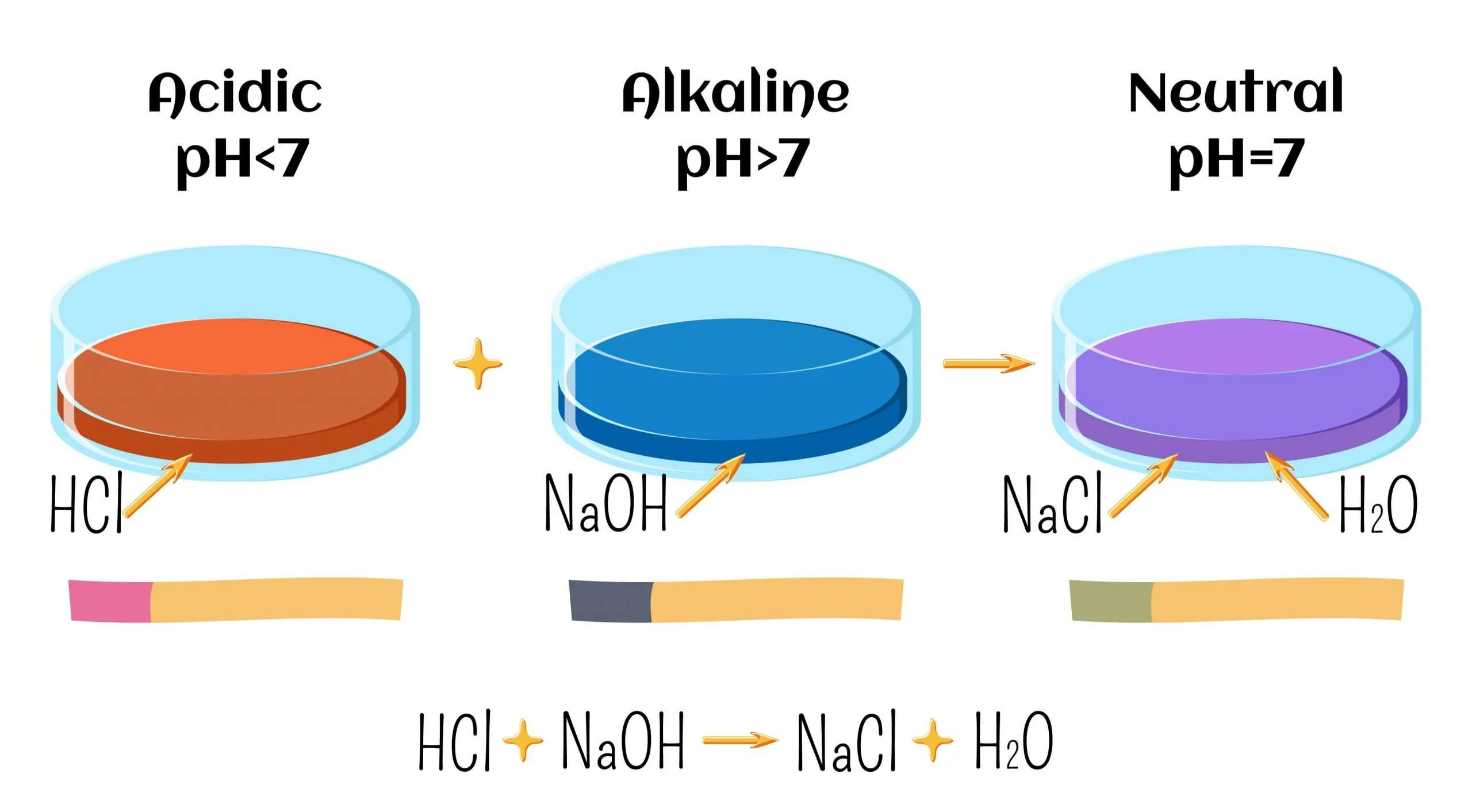
\( \mathrm{Acid + Base \rightarrow Salt + Water} \)
Example:
\( \mathrm{HCl + NaOH \rightarrow NaCl + H_2O} \)
Exothermic and Endothermic Reactions
Reactions can also be classified based on energy changes:
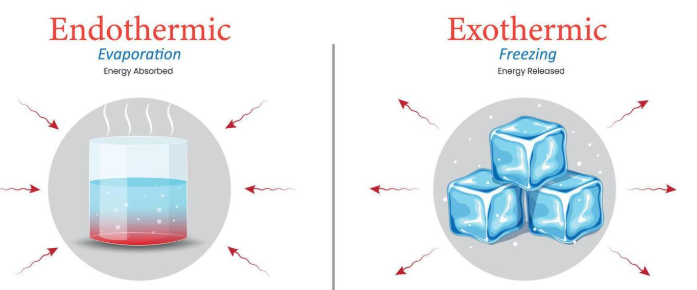
- Exothermic: release energy (temperature increases)
- Endothermic: absorb energy (temperature decreases)
Real-World Importance
- Combustion reactions power vehicles and industries
- Neutralisation is used in agriculture to control soil pH
- Displacement reactions are used in metal extraction
Example 1:
Classify the reaction: \( \mathrm{2K + Cl_2 \rightarrow 2KCl} \)
▶️ Answer/Explanation
Two elements combine to form one compound
Conclusion: Combination (synthesis) reaction.
Example 2:
Identify the type of reaction: \( \mathrm{CuO + H_2 \rightarrow Cu + H_2O} \)
▶️ Answer/Explanation
Hydrogen replaces copper in the compound
Conclusion: Displacement reaction.
Example 3:
A compound \( \mathrm{XCO_3} \) is heated and forms \( \mathrm{XO} \) and \( \mathrm{CO_2} \).
a) Identify the type of reaction
b) Explain the energy change involved
▶️ Answer/Explanation
Step 1: Reaction pattern
One compound breaks into two products
Step 2: Type of reaction
Decomposition reaction
Step 3: Energy change
Heat is required to break bonds → endothermic
Conclusion: Decomposition and endothermic reaction.
