IB MYP Integrated Science- Physics- Energy transfer and transformation-Study Notes - New Syllabus
IB MYP Integrated Science- Physics – Link -Study Notes – New syllabus
IB MYP Integrated Science- Physics – Link -Study Notes -As per latest Syllabus.
Key Concepts:
Energy transfer and transformation (including heat)
IB MYP Integrated Science -Concise Summary Notes- All Topics
Types of Energy
Energy is the ability to do work or cause change.
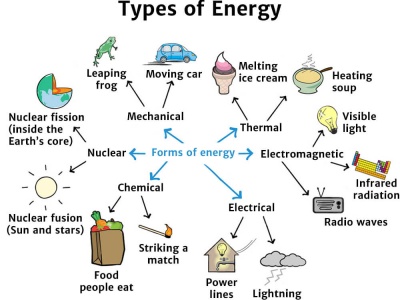
Key Types of Energy
| Type of Energy | Description | Example |
|---|---|---|
| Kinetic Energy | Energy of motion | Moving car |
| Potential Energy | Stored energy due to position | Object at height |
| Thermal Energy | Energy due to heat | Hot water |
| Chemical Energy | Energy stored in chemical bonds | Batteries, food |
| Electrical Energy | Energy carried by moving charges | Electric current |
| Light Energy | Energy from light waves | Sunlight |
| Sound Energy | Energy carried by sound waves | Speaker |
| Nuclear Energy | Energy from atomic nucleus | Nuclear power plants |
Key Concepts
- Energy can exist in different forms
- Energy can be transferred from one form to another
- Total energy is always conserved (cannot be created or destroyed)
Example:
What type of energy does a moving car have?
▶️ Answer/Explanation
A moving object has energy due to motion.
Final Answer: \( \boxed{\text{Kinetic Energy}} \)
Example:
A stretched spring stores energy. What type is it?
▶️ Answer/Explanation
Energy stored due to position or shape is potential energy.
Final Answer: \( \boxed{\text{Potential Energy}} \)
Energy Changes (Energy Transfer & Transformation)
Definition
Energy changes refer to the transfer or transformation of energy from one form to another.
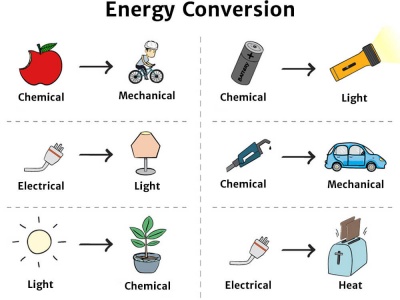
Key Concepts
- Energy can be transferred between objects
- Energy can be transformed from one form to another
- Total energy is always conserved
- Some energy is often lost as heat to the surroundings
Common Energy Transformations
| Device / System | Energy Input | Energy Output |
|---|---|---|
| Electric bulb | Electrical | Light + Heat |
| Motor | Electrical | Kinetic |
| Generator | Kinetic | Electrical |
| Falling object | Potential | Kinetic |
| Battery | Chemical | Electrical |
Energy Transfer Methods
- Mechanically → force doing work
- Electrically → moving charges
- Heating → due to temperature difference
- Radiation → energy transfer by waves (e.g., light)
Conservation of Energy
The total energy before and after a transformation remains constant.
\( \text{Total Energy Input} = \text{Total Energy Output} \)
Example:
What energy change occurs in a torch (flashlight)?
▶️ Answer/Explanation
Chemical energy from battery is converted to electrical energy, then to light and heat.
Final Answer: \( \boxed{\text{Chemical → Electrical → Light + Heat}} \)
Example:
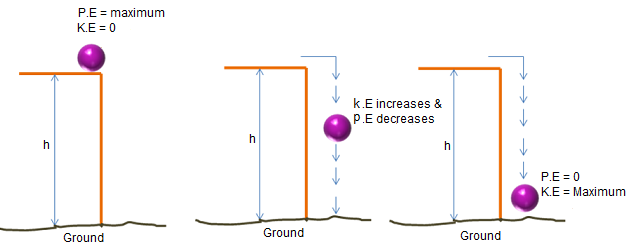
A ball is dropped from a height. Describe the energy changes.
▶️ Answer/Explanation
Gravitational potential energy converts into kinetic energy as it falls.
Final Answer: \( \boxed{\text{Potential → Kinetic}} \)
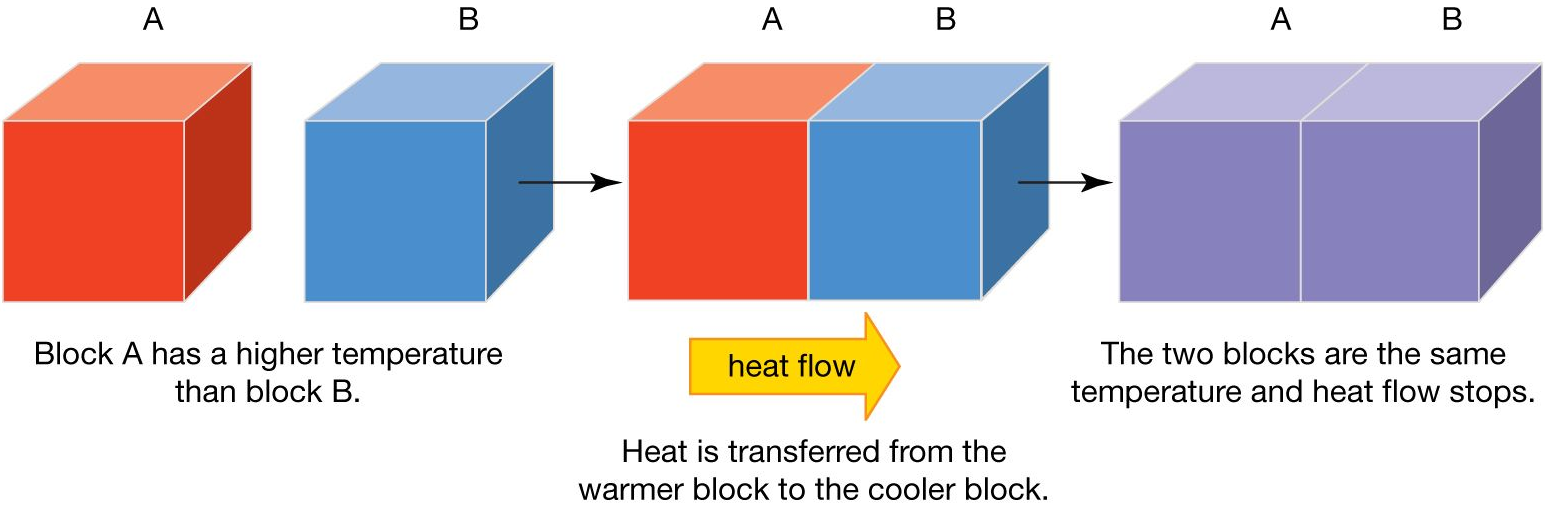
Heat Transfer
Definition
Heat transfer is the movement of thermal energy from a region of higher temperature to a region of lower temperature due to a temperature difference.
Modes of Heat Transfer
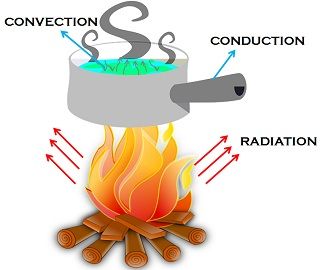
- Conduction
- Convection
- Radiation
Conduction
Conduction is the transfer of heat through a substance without any bulk movement of the substance.

Mechanism (Microscopic Explanation)
- Particles in a solid vibrate about fixed positions
- When heated, particles gain energy and vibrate more
- They transfer energy to neighboring particles through collisions
- In metals, free electrons also carry energy quickly

Key Concepts
- Occurs mainly in solids
- No movement of the material as a whole
- Metals are good conductors (e.g., copper, aluminium)
- Insulators are poor conductors (e.g., wood, plastic, air)
- Rate of conduction depends on:
- Material
- Temperature difference
- Thickness
Applications
- Cooking utensils made of metal
- Handles of utensils made of plastic (insulators)
- Thermal insulation in buildings
Example:
Why does one end of a metal rod become hot when the other end is heated?
▶️ Answer/Explanation
Heat is transferred through particle vibrations and free electrons.
Final Answer: \( \boxed{\text{Heat transfer by conduction}} \)
Example:
Why are house walls made thick?
▶️ Answer/Explanation
Thicker walls reduce heat transfer by conduction.
Final Answer: \( \boxed{\text{Reduces heat loss}} \)
Convection
Convection is the transfer of heat through the bulk movement of fluids (liquids or gases).
Mechanism

- When a fluid is heated, it expands
- Density decreases → fluid becomes lighter
- Hot fluid rises
- Cool fluid (denser) sinks
- This creates a convection current
Key Concepts
- Occurs only in liquids and gases
- Involves movement of the substance
- Heat transfer depends on fluid motion
- Forms convection currents
Applications
- Heating water in a kettle
- Air circulation in rooms (heaters and AC)
- Sea breeze and land breeze
Example:
Why does smoke rise up a chimney?
▶️ Answer/Explanation
Hot air is less dense and rises, carrying smoke upward.
Final Answer: \( \boxed{\text{Convection currents}} \)
Example:
Why are air conditioners placed near the top of a room?
▶️ Answer/Explanation
Cool air sinks and spreads downward, improving cooling.
Final Answer: \( \boxed{\text{Cool air circulates by convection}} \)
Radiation
Radiation is the transfer of heat in the form of electromagnetic waves without requiring a medium.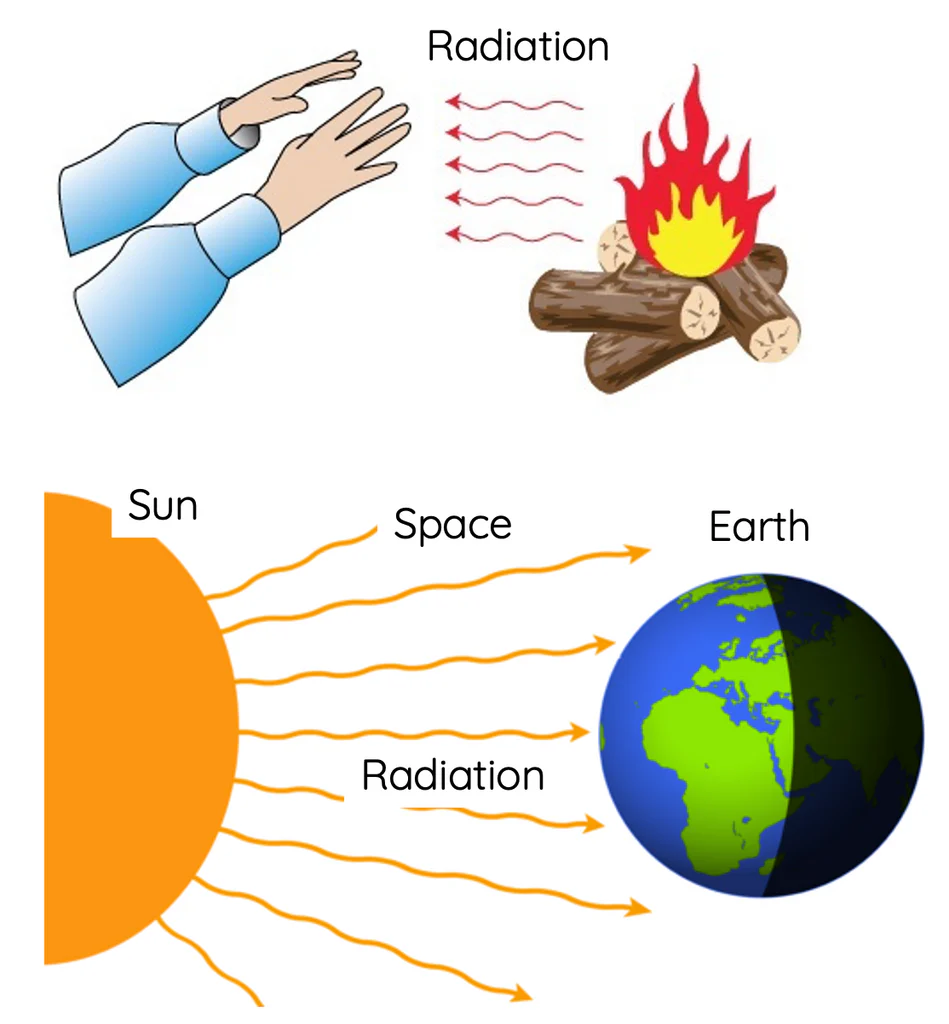
Mechanism
- All objects emit thermal radiation
- Energy travels as electromagnetic waves (infrared)
- No particles are required → can occur in vacuum
Key Concepts
- Does not require a medium
- Travels at the speed of light
- Darker surfaces absorb and emit more radiation
- Shiny/light surfaces reflect radiation
Applications
- Heat from the Sun reaching Earth
- Solar panels
- Thermal flasks (reflect radiation)
Example:
Why can we feel heat from a fire without touching it?
▶️ Answer/Explanation
Heat travels through electromagnetic waves.
Final Answer: \( \boxed{\text{Radiation}} \)
Example:
Why are shiny surfaces used in thermal flasks?
▶️ Answer/Explanation
Shiny surfaces reflect radiation and reduce heat loss.
Final Answer: \( \boxed{\text{Reduces heat transfer by radiation}} \)
