IB MYP Integrated Sciences e-Assessment : Chemical bonding
Exam Style Practice Questions - New Syllabus
Question (9 marks)
Phosphoric acid (H3PO4) is added to cola to provide a sour taste that balances the sweetness of sucrose.
Question a (2 marks)
Calculate the molar mass of phosphoric acid (H3PO4) with units.
▶️Answer/Explanation
Answer:
98.00 g mol-1
Explanation:
Calculation:
3(H) = 3 × 1.01 = 3.03
1(P) = 30.97
4(O) = 4 × 16.00 = 64.00
Total = 3.03 + 30.97 + 64.00 = 98.00 g mol-1
Question b (2 marks)
Calculate the moles of sucrose (C12H22O11) in 39g (molar mass = 342.3 g mol-1) to 3 significant figures.
▶️Answer/Explanation
Answer:
0.114 mol
Explanation:
n = m/M = 39 g / 342.3 g mol-1 = 0.1139 mol ≈ 0.114 mol
Question c (3 marks)
Determine the molecular formula of xylitol (artificial sweetener) from its structural formula.

▶️Answer/Explanation
Answer:
C5H12O5
Explanation:
From the structural diagram (not shown here):
• 5 carbon atoms
• 12 hydrogen atoms
• 5 hydroxyl (OH) groups → 5 oxygen atoms
Question d (2 marks)
Identify the functional groups in aspartame (artificial sweetener):


▶️Answer/Explanation
Answer:
• Carboxylic acid (-COOH)
• Ester (-COOCH3)
Explanation:
From the structural diagram:
1. The -COOH group is characteristic of carboxylic acids
2. The -COOCH3 group shows an ester linkage
Question:
Find out each compound’s chemical formula and classify it as ionic or covalent. Are the results with respect to conductivity what you predicted given your knowledge of the properties of these compounds?
▶️Answer/Explanation
Ans: 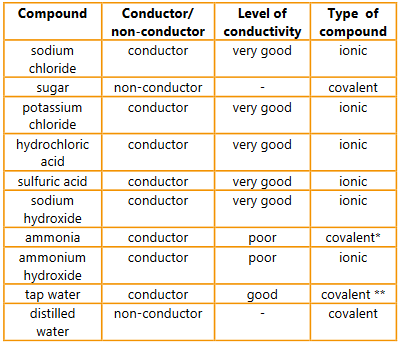
* NH3 + H2O ↔ NH4+ + OH–
** The presence of a wide range of dissolved ions makes tap water a conductor
