The structure of the atom IB DP Physics Study Notes - 2026, 2027 & 2028 Syllabus
The structure of the atom IB DP Physics Study Notes
The structure of the atom IB DP Physics Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on IB Physics syllabus with Students should understand
the Geiger–Marsden–Rutherford experiment and the discovery of the nucleus
nuclear notation ZAX where A is the nucleon number Z is the proton number and X is the chemical symbol
- that emission and absorption spectra provide evidence for discrete atomic energy levels
- that photons are emitted and absorbed during atomic transitions
- that the frequency of the photon released during an atomic transition depends on the difference in energy level as given by E = hƒ
- that emission and absorption spectra provide information on the chemical composition.
Standard level and higher level: 6 hours
Additional higher level: 3 hours
- IB DP Physics 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Physics 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Physics 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
- IB DP Physics 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
The Geiger–Marsden–Rutherford experiment and the discovery of the nucleus
In 1897, J. J. Thomson discovered the electron and proposed the plum pudding model of the atom, in which negatively charged electrons were embedded in a diffuse sphere of positive charge.
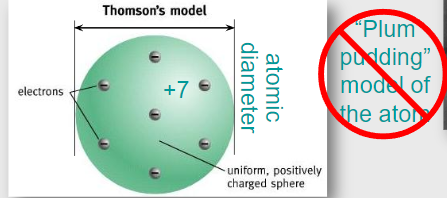
According to this model, alpha particles passing through an atom were expected to experience only small deflections.
In 1911, Ernest Rutherford, together with Geiger and Marsden, investigated atomic structure by directing a beam of alpha particles at a thin sheet of gold foil.
- Gold foil was used because it could be made extremely thin.
- This ensured alpha particles passed through only a few layers of atoms.
FYI
- An alpha particle is a positively charged particle with charge \( +2e \).
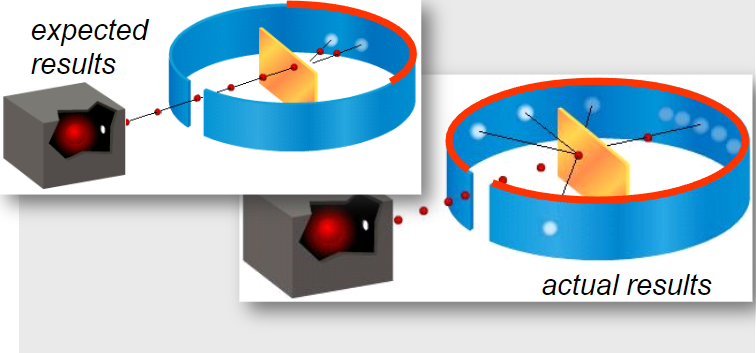
If the plum pudding model were correct, most alpha particles would pass straight through with minimal deflection.

However, the actual observations were very different.
Key observations:
- Most alpha particles passed straight through the foil.
- Some were deflected through small angles.
- A very small number were deflected through large angles, with some rebounding.
These large-angle deflections could not be explained by the plum pudding model.
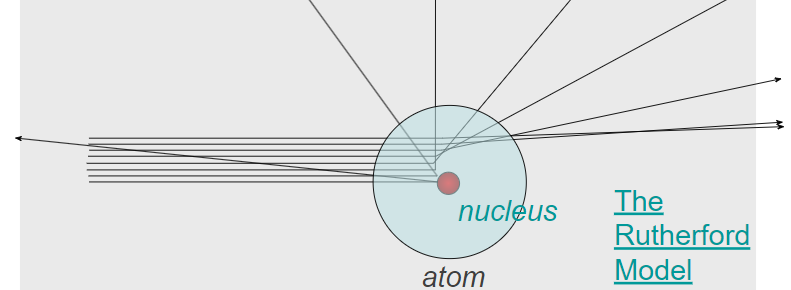
Rutherford proposed that all of the positive charge and most of the mass of the atom were concentrated in a tiny central region called the nucleus.
Conclusions of the experiment:
- The atom is mostly empty space.
- The nucleus is small, dense, and positively charged.
- Electrons occupy the space surrounding the nucleus.
Only by assuming a concentrated positive nucleus could Rutherford and his team explain the observed scattering pattern.
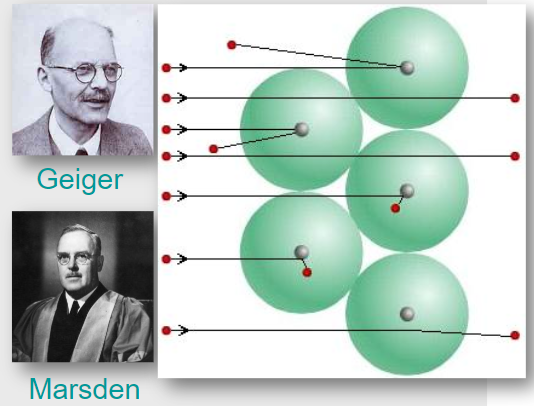
The Geiger–Marsden experiment therefore provided direct experimental evidence for the existence of the atomic nucleus and led to the nuclear model of the atom.
Example:
Explain how the results of the Geiger–Marsden experiment led Rutherford to propose the nuclear model of the atom.
▶️ Answer / Explanation
Most alpha particles passed straight through the foil, showing that atoms are mostly empty space.
The few large-angle deflections indicated that a small, dense, positively charged region existed within the atom.
Rutherford concluded that this region was the nucleus, containing most of the atom’s mass and positive charge.
Nuclear Notation \( ^A_Z X \)
Nuclei are represented using the nuclear notation \( ^A_Z X \), which gives information about the composition of an atom.
\( ^A_Z X \)
Meaning of the symbols:
- \( X \): Chemical symbol of the element
- \( Z \): Proton number (number of protons in the nucleus)
- \( A \): Nucleon number (total number of protons and neutrons)
Relationship between nucleons:
The number of neutrons \( N \) in the nucleus is given by:
![]()
Important points:
- The element is identified by the proton number \( Z \).
- Atoms of the same element always have the same \( Z \) but may have different \( A \) (isotopes).
- Nuclear notation applies to the nucleus, not the electrons.
Example:
For the nucleus \( ^{14}_{6}\text{C} \), determine the number of protons and neutrons.
▶️ Answer / Explanation
From the notation \( ^{14}_{6}\text{C} \):
Number of protons \( = Z = 6 \)
Number of neutrons \( = A – Z = 14 – 6 = 8 \)
This nucleus is an isotope of carbon with 8 neutrons.
Emission and Absorption Spectra and Discrete Energy Levels
When a low-pressure gas is subjected to a high voltage, the gas becomes excited and emits light.

This emitted light is analysed using a spectroscope, which separates the light into its component wavelengths.

The observed emission spectrum consists of discrete bright lines, not a continuous spectrum.
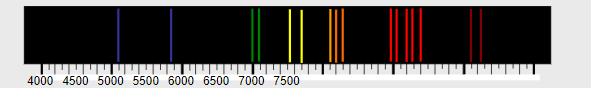
This discontinuous spectrum proves that electrons can only occupy fixed (quantised) energy levels.
Example:
Explain how a line emission spectrum proves that atomic energy levels are discrete.
▶️ Answer / Explanation
A line spectrum shows only specific wavelengths, not a continuous range.
This means electrons can only change energy by fixed amounts.
Therefore, atomic energy levels must be discrete (quantised).
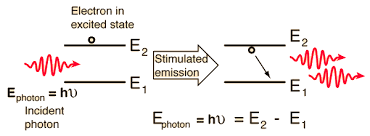
Photon Emission and Absorption in Atomic Transitions
Light energy is carried by particles called photons.
When a photon is absorbed, an electron jumps to a higher energy level (excitation).
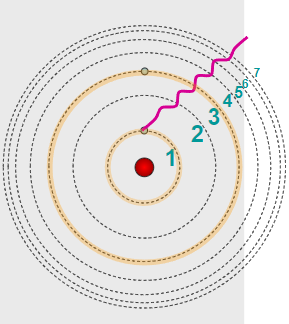
When the electron falls to a lower energy level, a photon is emitted (de-excitation).
Example:
Describe what happens to photons when an atom is excited and de-excited.
▶️ Answer / Explanation
During excitation, a photon is absorbed and an electron moves to a higher energy level.
During de-excitation, the electron falls to a lower level and a photon is emitted.
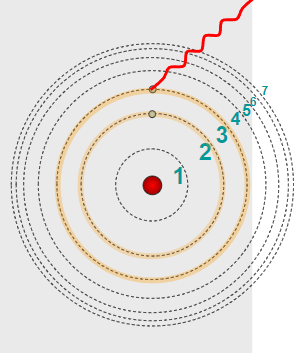
Photon Frequency and Atomic Energy Level Transitions
When an atom undergoes an electronic transition between two energy levels, a photon is either emitted or absorbed.
The frequency of the photon released or absorbed depends on the difference in energy between the two energy levels.
\( E = h f \)
Where:
- \( E \): Energy of the photon (in joules)
- \( h \): Planck constant \( (6.63 \times 10^{-34} \, \text{J s}) \)
- \( f \): Frequency of the photon (in hertz)
Energy level difference:
The energy of the photon is equal to the difference between the initial and final energy levels of the electron:
\( E = \Delta E = E_{\text{initial}} – E_{\text{final}} \)
Emission and absorption:
- If an electron falls to a lower energy level, a photon is emitted.
- If an electron rises to a higher energy level, a photon is absorbed.
Key idea:
Larger energy differences between levels produce photons with higher frequencies.
Example:
An electron in an atom drops to a lower energy level, releasing a photon with energy \( 3.3 \times 10^{-19} \, \text{J} \). Calculate the frequency of the photon.
▶️ Answer / Explanation
Step 1: Use the photon energy equation
\( E = h f \)
Step 2: Rearrange
\( f = \dfrac{E}{h} \)
Step 3: Substitute values
\( f = \dfrac{3.3 \times 10^{-19}}{6.63 \times 10^{-34}} = \boxed{5.0 \times 10^{14} \, \text{Hz}} \)
Interpretation:
The photon lies in the visible region of the electromagnetic spectrum.
Spectra and Chemical Composition
Each element has a unique set of atomic energy levels.
Therefore, each element produces a unique emission and absorption spectrum (spectral fingerprint).
By matching observed spectra with reference spectra, the chemical composition of a source can be identified.

Example:
A star’s spectrum shows hydrogen absorption lines. What does this tell us?
▶️ Answer / Explanation
Hydrogen absorption lines mean hydrogen is present in the star’s atmosphere.
Spectra therefore identify chemical composition.
IB Physics The structure of the atom Exam Style Worked Out Questions
Question
What is the variation of nuclear density $\rho$ with nucleon number A ?
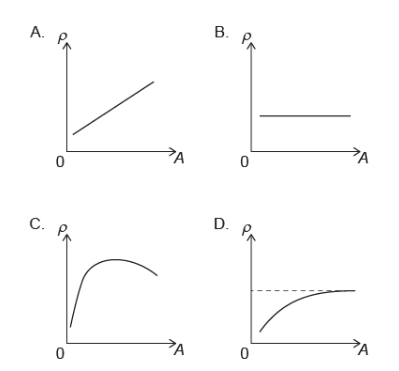
▶️Answer/Explanation
Ans:B
Question
The diameter of a nucleus of a particular nuclide $X$ is $12 \mathrm{fm}$. What is the nucleon number of $X$ ?
A. 5
B. 10
C. 125
D. 155
▶️Answer/Explanation
Ans:C
