IB DP Chemistry Mock Exam HL Paper 1B Set 3 - 2025 Syllabus
IB DP Chemistry Mock Exam HL Paper 1B Set 3
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam HL Paper 1B Set 3. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
▶️ Answer/Explanation
(a)
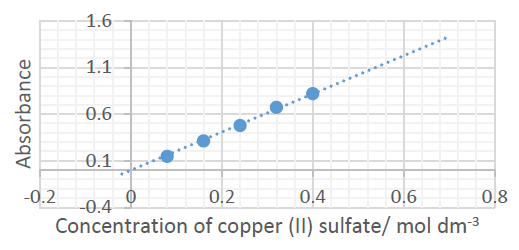
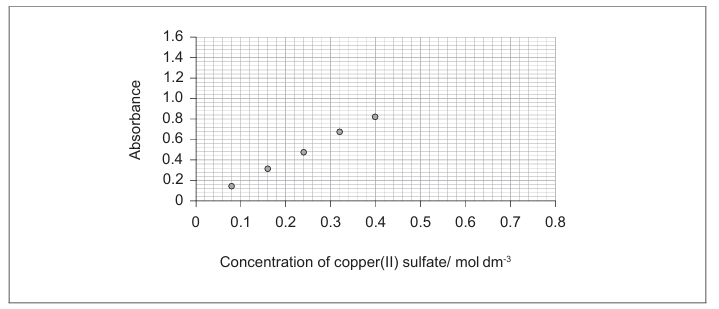
• A reasonable line of best fit that passes through or near all data points ✓
• The line should be extended beyond the range of the measured data (beyond 0.08 AND 0.32 mol dm⁻³) ✓
Appropriate line drawn with extrapolation.
(b)
The graph shows that absorbance is directly proportional to concentration.
\(\boxed{\text{Directly proportional}}\)
(c)
A specific wavelength is used because absorbance varies with wavelength, and maximum absorbance occurs at a characteristic wavelength for each substance, allowing for more accurate concentration determination.
\(\boxed{\text{For maximum accuracy in concentration measurement}}\)
(d)
• Gradient = 2.0 (accept range 1.9-2.3) ✓
• Equation: Absorbance = 2.0 × Concentration (y = 2.0x) ✓
\(\boxed{2.0}\) and \(\boxed{A = 2.0C}\)
(e)
Using the equation A = 2.0C: 2.44 = 2.0 × C → C = 1.22 mol dm⁻³
\(\boxed{1.22}\) mol dm⁻³ (accept 1.20-1.30)
(f)
There is no significant difference between using this calibration curve and a theoretical one for determining concentrations.
\(\boxed{\text{No significant difference}}\)
(g)
The wavelength of maximum absorbance for copper(II) sulfate is approximately 635 nm.
\(\boxed{635}\) nm (accept 585-647 nm)