IB DP Chemistry Mock Exam SL Paper 1B Set 2 - 2025 Syllabus
IB DP Chemistry Mock Exam SL Paper 1B Set 2
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam SL Paper 1B Set 2. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
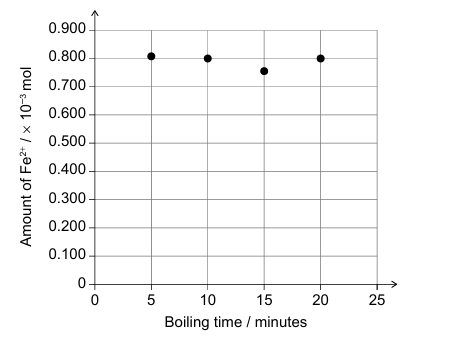
(b) The mean values from part (a) were plotted on a graph of amount of \(\text{Fe}^{2+}\) released (\(\times 10^{-3}\ \text{mol}\)) against boiling time (\(\text{minutes}\)).
▶️ Answer/Explanation
(a)(i)
Independent variable: boiling time of the spinach in water (for example \(5\), \(10\), \(15\), \(20\ \text{minutes}\)).
Dependent variable: volume (titre) of \(\text{KMnO}_{4}(\text{aq})\) added in the titration.
(a)(ii)
Any two appropriate controlled conditions, e.g.:
• Keep the final volume of solution the same for each trial by adding water after filtration to a fixed volume.
• Use spinach leaves of the same freshness/colour/size or surface area (similar samples).
• Boil the water before adding spinach and ensure the temperature while boiling is the same for every trial.
• Use distilled or deionized water instead of tap water.
• Store all solutions in the refrigerator for the same length of time before titration.
• Use the same size storage bottles for all samples.
• Ensure all samples are titrated at the same temperature (allow to reach room temperature).
• Maintain the same acidic conditions of the titration solution (constant \(\text{pH}\)).
• Protect \(\text{KMnO}_{4}(\text{aq})\) from light because it is photosensitive.
(Any two suitable controls gain credit.)
(a)(iii)
Perform a trial using boiled water with no spinach leaves (a blank).
This measures how much \(\text{Fe}^{2+}\), if any, is contributed by the water or equipment, so the amount from the spinach alone can be found by comparison.
(a)(iv)
Any two valid difficulties, e.g.:
• \(\text{KMnO}_{4}(\text{aq})\) is dark purple, so it can be hard to read the burette scale accurately; the reading must be taken from the top of the meniscus.
• The spinach solutions are green/coloured, so the colour change at the end point can be difficult to judge.
• The end point is a mixture of green and faint pink, so the exact moment of the first permanent pink colour can be subjective.
• The reaction may be slow, so the colour takes a long time to disappear, but titration must be done quickly to prevent organic substances reducing \(\text{Fe}^{3+}\) back to \(\text{Fe}^{2+}\).
(Any two suitable points gain credit.)
(b)(i)
The amount (or concentration) of \(\text{Fe}^{2+}\) extracted from the spinach does not show a clear increase as boiling time rises from \(5\) to \(20\ \text{minutes}\); the points are roughly constant (or show no consistent trend).
So there is essentially no obvious relationship/correlation shown in the graph.
(b)(ii)
Range:
• The boiling times (from \(5\) to \(20\ \text{minutes}\)) are realistic cooking times, but the overall range is quite narrow and may not be sufficient to reveal a clear trend. It is likely most \(\text{Fe}^{2+}\) was already extracted before \(5\ \text{minutes}\). More boiling times, including values below \(5\ \text{minutes}\), are needed to see a pattern.
Quantity of measurements:
• There are several measurements for each boiling time (replicates), which is good; however, more replicates would allow calculation of a standard deviation and give better information about variation between trials.
• A measurement at \(t = 0\ \text{minutes}\) (no boiling) is missing, so it is hard to judge whether boiling actually changes the amount of \(\text{Fe}^{2+}\).
• There is only one interval between \(0\) and \(5\ \text{minutes}\); this is not enough to see how fast \(\text{Fe}^{2+}\) is extracted at the beginning.
(c)(i)
The hypothesis is not supported by the data.
Reason: the results do not show an increase in the amount of \(\text{Fe}^{2+}\) with longer boiling times; in fact, the amount at \(5\ \text{minutes}\) is similar to or even slightly higher than at later times. Because no measurement at \(t = 0\ \text{minutes}\) was taken, we also cannot say whether boiling makes any difference compared with no boiling.
(c)(ii)
One suitable modification is to repeat the investigation using a wider and more detailed set of boiling times, for example at \(1\), \(2\), \(3\), \(4\), and \(5\ \text{minutes}\) (or over a much longer range, depending on focus).
Other acceptable changes include: repeating the investigation without boiling (to provide a \(0\ \text{minute}\) point), using a spectrophotometer/atomic absorption spectrophotometer to measure \(\text{Fe}^{2+}\) more precisely, or using a larger mass of spinach to produce more measurable amounts of \(\text{Fe}^{2+}\).
