IB DP Chemistry Mock Exam SL Paper 1B Set 3 - 2025 Syllabus
IB DP Chemistry Mock Exam SL Paper 1B Set 3
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam SL Paper 1B Set 3. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
▶️ Answer/Explanation
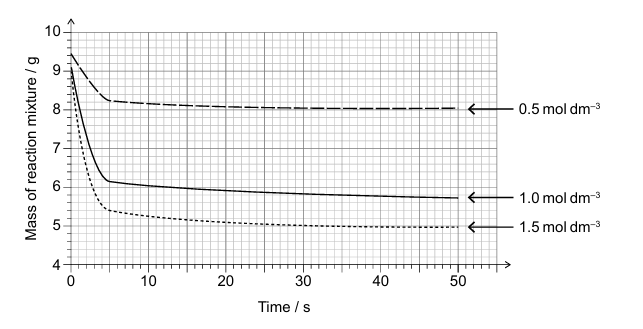
(a)
• Tangent drawn at t = 0 for 1.5 mol dm⁻³ solution ✓
• Gradient calculated AND positive value for rate: ≈2.3 g s⁻¹ ✓
\(\boxed{2.3}\) g s⁻¹
(b)
• Limiting reagent: HCl ✓
• Reason: Same initial mass of solid produces different mass of CO₂/carbon dioxide ✓
HCl is limiting because different acid concentrations produce different CO₂ masses.
(c)(i)
Actual mass of CO₂ produced = 9.1 – 5.8 = 3.3 g ✓
\(\boxed{3.3}\) g
(c)(ii)
Percentage difference = [(4.40 – 3.3)/4.40] × 100 = 25% ✓
\(\boxed{25\%}\)
(c)(iii)
Any one of:
• Recording of mass started after reaction began ✓
• Button on data-logger was pressed too late ✓
• Balance not tared/zeroed ✓
• Use of different beakers/flasks/glassware with slightly different masses ✓
• Glassware was not dried/cleaned from previous trial ✓
Any one valid source of error.