Energy from fuels: R1.3.5 Fuel cells IB DP Chemistry Study Notes - New Syllabus 2026, 2027 & 2028
Energy from fuels – IB DP Chemistry- Study Notes
IITian Academy excellent Introduction to the Particulate Nature of Matter – Study Notes and effective strategies will help you prepare for your IB DP Chemistry 2025 exam.
- IB DP Chemistry 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Chemistry 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
- IB DP Chemistry 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Chemistry 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
Reactivity 1.3.5 — Fuel Cells
Reactivity 1.3.5 — Fuel Cells
Fuel cells are electrochemical devices that convert the chemical energy of a fuel (typically hydrogen or methanol) and an oxidizing agent (usually oxygen) directly into electrical energy through redox reactions. Unlike combustion engines, fuel cells do not involve thermal energy conversion; hence, they offer higher efficiency and lower emissions.
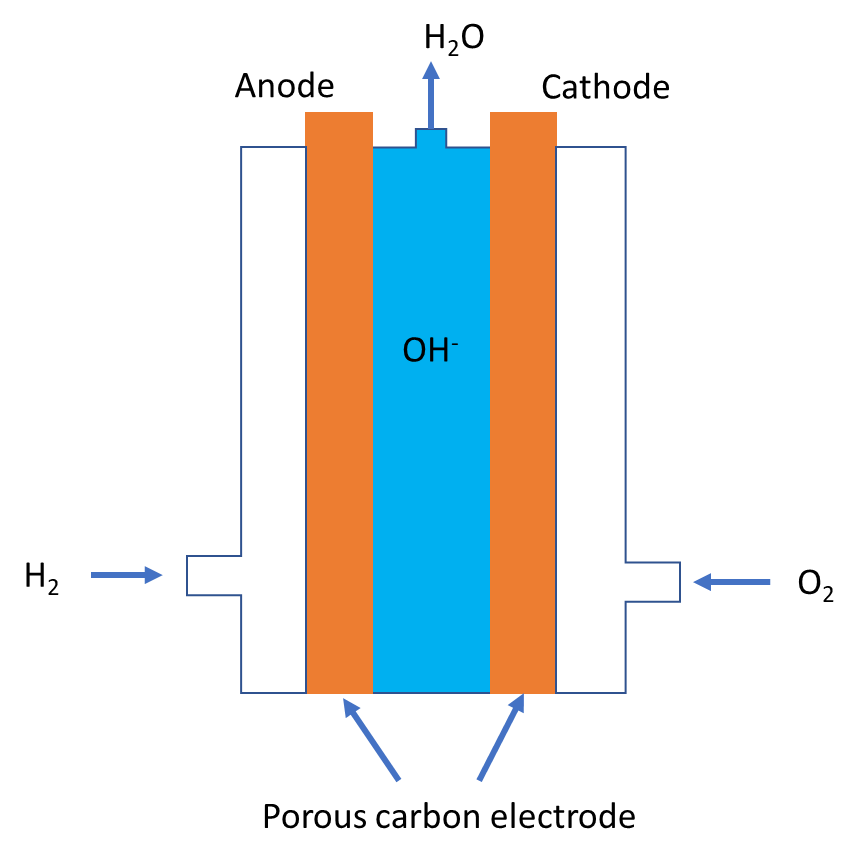
Key Features of a Fuel Cell:
- Continuous supply of fuel and oxidant maintains energy output as long as they are provided.
- Operates under relatively low temperatures compared to combustion systems.
- Produces electricity, water, and sometimes heat as by-products.
- No combustion involved — therefore, no greenhouse gases are emitted from the process itself (especially in hydrogen fuel cells).
Main Components:
- Anode: Site of oxidation (fuel loses electrons).
- Cathode: Site of reduction (oxidant gains electrons).
- Electrolyte: Allows ion transfer between the electrodes but blocks electron flow.
- External Circuit: Electrons flow through this, producing electric current.
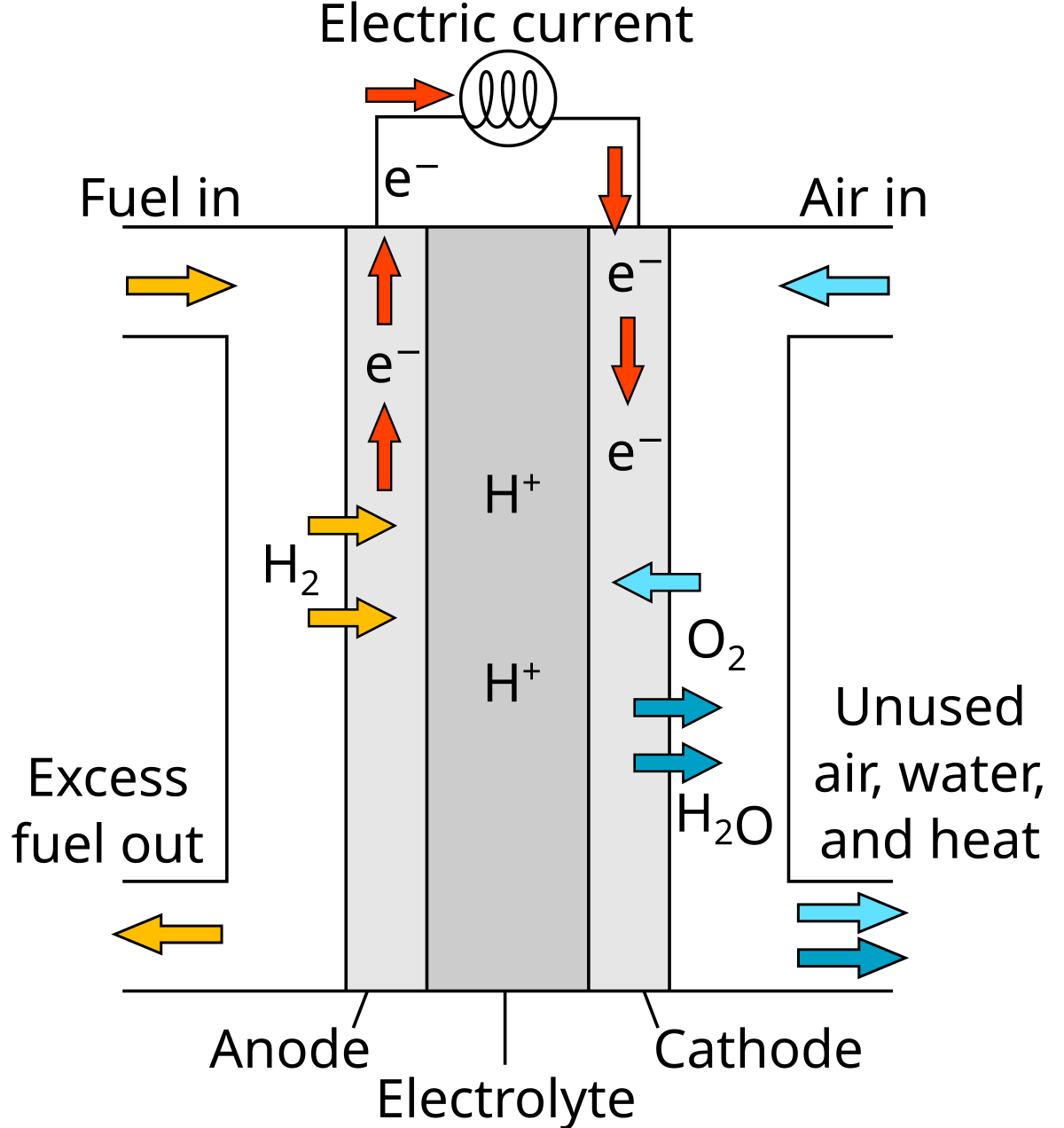
Electrode Half-Equations in Fuel Cells
Fuel cells rely on redox reactions at electrodes to convert chemical energy into electrical energy.
- Oxidation occurs at the anode (fuel loses electrons).
- Reduction occurs at the cathode (oxidant gains electrons).
- Electrons flow through the external circuit, powering electrical devices.
Types of Fuel Cells
1. Hydrogen-Oxygen Fuel Cell (in Acidic Medium)
Uses hydrogen gas as the fuel and oxygen as the oxidant. Water is the only product, making this a clean energy source.
Half-Equations of Hydrogen-Oxygen Fuel Cell:
- Anode (oxidation): $ \text{H}_2(g) \rightarrow 2\text{H}^+(aq) + 2e^- $
- Cathode (reduction): $ \frac{1}{2}\text{O}_2(g) + 2\text{H}^+(aq) + 2e^- \rightarrow \text{H}_2\text{O}(l) $
Overall Reaction:
$ \text{H}_2(g) + \frac{1}{2}\text{O}_2(g) \rightarrow \text{H}_2\text{O}(l) $
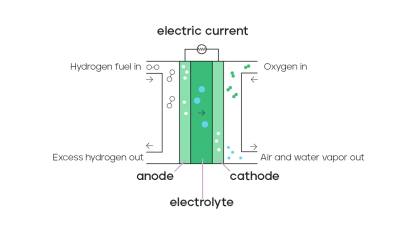
Key Notes:
- Only water is produced — no CO2.
- Efficient and environmentally friendly when hydrogen is sourced sustainably.
Advantages
- Produces only water — no greenhouse gases if hydrogen is sustainably sourced.
- High efficiency (~60%) compared to internal combustion engines.
- Quiet operation — minimal moving parts.
- Suitable for clean environments (e.g., space, hospitals).
Disadvantages
- Hydrogen is difficult to store — requires high pressure or cryogenic tanks.
- Currently limited infrastructure for hydrogen refueling.
- Hydrogen production may still rely on fossil fuels (e.g., steam reforming).
- Platinum catalysts increase cost.
2. Methanol Fuel Cell (Direct Methanol Fuel Cell – DMFC)(in Acidic Medium)
Uses liquid methanol (CH<sub>3</sub>OH) as the fuel. These are easier to store and transport than hydrogen.
Half-Equations:
- Anode (oxidation): $ \text{CH}_3\text{OH}(aq) + \text{H}_2\text{O}(l) \rightarrow \text{CO}_2(g) + 6\text{H}^+(aq) + 6e^- $
- Cathode (reduction): $ \frac{3}{2}\text{O}_2(g) + 6\text{H}^+(aq) + 6e^- \rightarrow 3\text{H}_2\text{O}(l) $
Overall Reaction:
$ \text{CH}_3\text{OH}(aq) + \frac{3}{2}\text{O}_2(g) \rightarrow \text{CO}_2(g) + 2\text{H}_2\text{O}(l) $

Key Notes:
- Emits CO2 — not as clean as hydrogen fuel cells.
- More practical storage and transport (liquid fuel).
Advantages
- Liquid methanol is easier and safer to store than hydrogen gas.
- Simple refueling process — ideal for portable devices and backup systems.
- No need for high-pressure tanks or cryogenic systems.
- More compact suited for small-scale and consumer applications.
Disadvantages
- Emits CO₂: Not fully carbon-neutral.
- Lower efficiency (~30–40%) than hydrogen fuel cells.
- Toxicity and flammability: Methanol is poisonous if ingested or inhaled, and spills pose fire hazards, making safe handling and storage important.
- Catalyst reliance: Still requires precious metal catalysts, increasing cost.
Example
Compare the hydrogen-oxygen and methanol fuel cells in terms of environmental impact and practicality.
▶️Answer/Explanation
- Hydrogen fuel cells produce only water as a by-product, making them cleaner than methanol fuel cells, which emit CO₂.
- Methanol is easier to store and transport than hydrogen, which requires special storage conditions.
- Hydrogen fuel cells are more efficient, but methanol fuel cells are more practical for small-scale applications.
Advantages of Fuel Cells:
- High efficiency (up to 60%) compared to internal combustion engines.
- Environmentally friendly — hydrogen fuel cells emit only water vapor.
- Quiet operation — no mechanical engine noise.
- Can run continuously as long as fuel and oxygen are supplied.
Disadvantages of Fuel Cells:
- Hydrogen is difficult to store and transport safely (requires high pressure or cryogenic conditions).
- Hydrogen production may still rely on fossil fuels (unless using electrolysis with renewable energy).
- Fuel cells can be expensive due to precious metal catalysts (e.g., platinum).
- Methanol emits CO2 — though less than fossil fuels, it is not fully carbon-neutral.
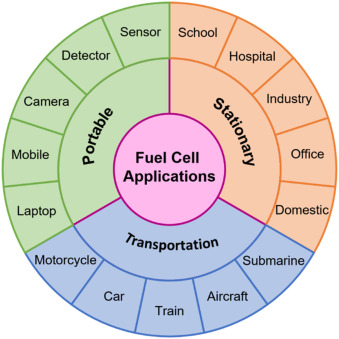
Applications:
- Electric vehicles (e.g., hydrogen-powered cars like the Toyota Mirai)
- Backup power systems (e.g., hospitals, satellites)
- Portable energy sources for electronics and military
Example:
Ethanol Fuel Cell — Write Half-Equations and Overall Reaction
Fuel: Ethanol (\( \text{C}_2\text{H}_5\text{OH} \))
Oxidant: Oxygen gas (\( \text{O}_2 \))
▶️Answer/Explanation
Step 1: Oxidation Half-Equation at the Anode
$ \text{C}_2\text{H}_5\text{OH}(aq) + 3\text{H}_2\text{O}(l) \rightarrow 2\text{CO}_2(g) + 12\text{H}^+(aq) + 12e^- $
Step 2: Reduction Half-Equation at the Cathode
$ 3\text{O}_2(g) + 12\text{H}^+(aq) + 12e^- \rightarrow 6\text{H}_2\text{O}(l) $
Step 3: Overall Cell Reaction
$ \text{C}_2\text{H}_5\text{OH}(aq) + 3\text{O}_2(g) \rightarrow 2\text{CO}_2(g) + 3\text{H}_2\text{O}(l) $
Key Notes:
- This fuel cell has high electron output (12 electrons per ethanol molecule).
- CO2 is produced, so it is not carbon-neutral.
- Ethanol is renewable if produced by fermentation.
Example:
Evaluate which fuel cell — hydrogen-oxygen or methanol — is more suitable for use in electric vehicles (EVs).
▶️Answer/Explanation
- Hydrogen Fuel Cell: Provides higher energy output per gram of fuel; ideal for long-range EVs. Produces only water — no harmful emissions if hydrogen is sourced renewably. However, hydrogen infrastructure (refueling stations, storage) is still limited.
- Methanol Fuel Cell: Easier to store and transport fuel in liquid form. Suitable for short-range or portable vehicles, but emits CO₂ and is less efficient. Infrastructure for liquid fuels is already well established.
- Conclusion: Hydrogen fuel cells are better for long-distance EVs and environmental performance, while methanol fuel cells are more practical for compact systems and early infrastructure use.
