IB DP Chemistry - R3.4.1 Nucleophiles - Study Notes - New Syllabus - 2026, 2027 & 2028
IB DP Chemistry – R3.4.1 Nucleophiles – Study Notes – New Syllabus
IITian Academy excellent Introduction to the Proton transfer reactions – Study Notes and effective strategies will help you prepare for your IB DP Chemistry exam.
- IB DP Chemistry 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Chemistry 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
- IB DP Chemistry 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Chemistry 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
Reactivity 3.4.1 — Nucleophiles
Reactivity 3.4.1 — Nucleophiles
A species that donates a pair of electrons to form a covalent bond with an electrophile. Nucleophiles are electron-rich and are attracted to electron-deficient species (electrophiles). By definition, nucleophiles are also Lewis bases, since they act as electron-pair donors.
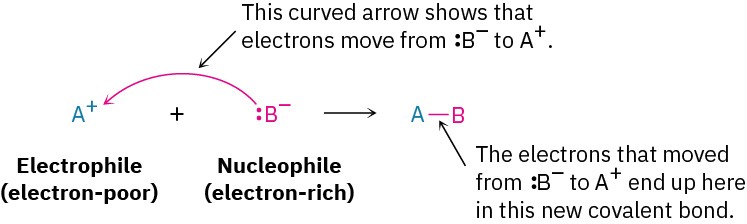
Key characteristics of nucleophiles:
- Possess a lone pair of electrons or a π-bond
- Are either negatively charged or neutral with a region of high electron density
- Can initiate a reaction by attacking a positively charged or electron-deficient center
Common nucleophiles:
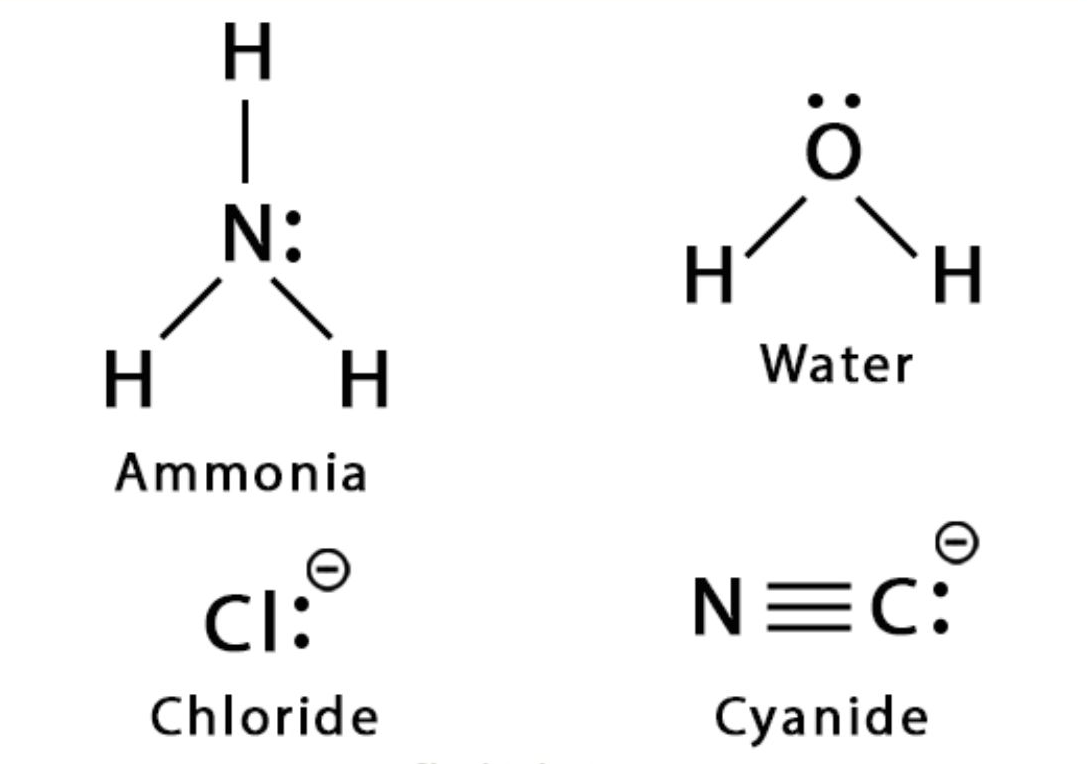
- \( \text{OH}^- \) (hydroxide ion)
- \( \text{CN}^- \) (cyanide ion)
- \( \text{NH}_3 \) (ammonia)
- \( \text{H}_2\text{O} \) (water)
- \( \text{Cl}^- \), \( \text{Br}^- \), \( \text{I}^- \) (halide ions)
Types of nucleophiles:
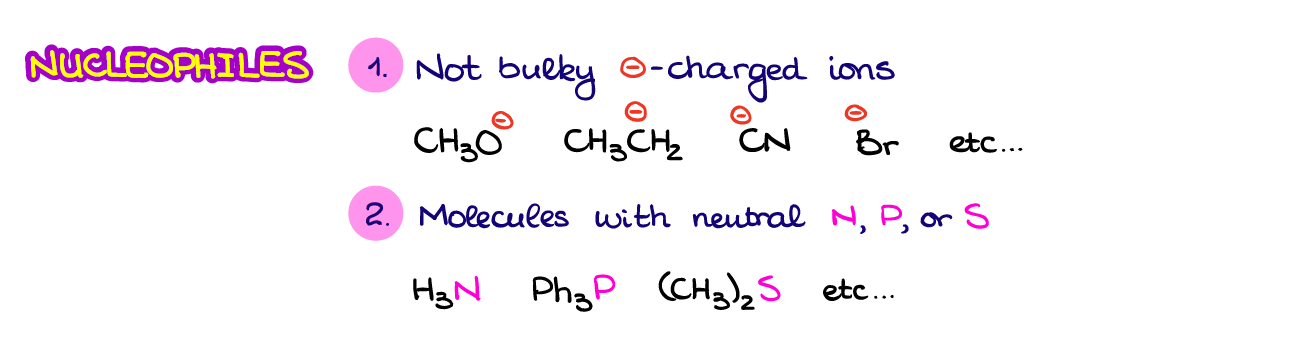
- Negatively charged: These include anions with lone pairs. For example, \( \text{OH}^- \), \( \text{CN}^- \), \( \text{Cl}^- \)
- Neutral: Molecules like \( \text{NH}_3 \), \( \text{H}_2\text{O} \), or alcohols where lone pairs are present on atoms like O or N
Mechanism Note:
Nucleophiles are the reactants that attack the electrophilic center (usually a carbon bonded to a leaving group) in substitution or addition reactions.
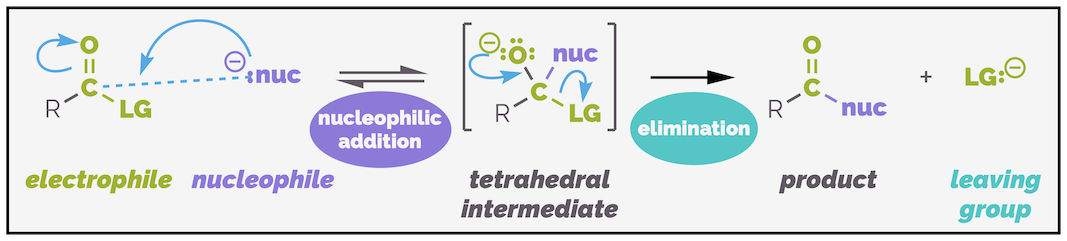
Identifying a nucleophile in a chemical reaction:
Look for species that:
- Have lone pairs of electrons (e.g., N, O atoms)
- Are negatively charged or polarised
- Are drawn to partial positive charges (especially δ+ C atoms in polar bonds)
Example
Explain: The reaction of hydroxide ion with bromoethane:
\( \text{CH}_3\text{CH}_2\text{Br} + \text{OH}^- \rightarrow \text{CH}_3\text{CH}_2\text{OH} + \text{Br}^- \)
▶️Answer/Explanation
The hydroxide ion acts as the nucleophile and attacks the δ+ carbon bonded to Br.
This is a nucleophilic substitution (SN2) reaction where the leaving group is Br−.
Example
Explain: The reaction of ammonia with chloroethane:
\( \text{CH}_3\text{CH}_2\text{Cl} + \text{NH}_3 \rightarrow \text{CH}_3\text{CH}_2\text{NH}_2 + \text{HCl} \)
▶️Answer/Explanation
Ammonia acts as a neutral nucleophile. It donates its lone pair on nitrogen to the electrophilic carbon, displacing Cl− and forming ethylamine.
Example
Explain: Reaction of cyanide ion with 2-bromopropane:
\( (\text{CH}_3)_2\text{CHBr} + \text{CN}^- \rightarrow (\text{CH}_3)_2\text{CHCN} + \text{Br}^- \)
▶️Answer/Explanation
The cyanide ion is a strong nucleophile and displaces the bromide ion via SN2 mechanism, forming a new carbon-carbon bond and producing a nitrile.
Example
Explain: Hydration of ethene using water as a nucleophile under acidic conditions:
\( \text{CH}_2=CH_2 + \text{H}_2\text{O} \xrightarrow{\text{H}^+} \text{CH}_3\text{CH}_2\text{OH} \)
▶️Answer/Explanation
Water, although neutral, acts as a nucleophile. In the presence of an acid catalyst, it adds across the double bond of ethene, forming ethanol.
