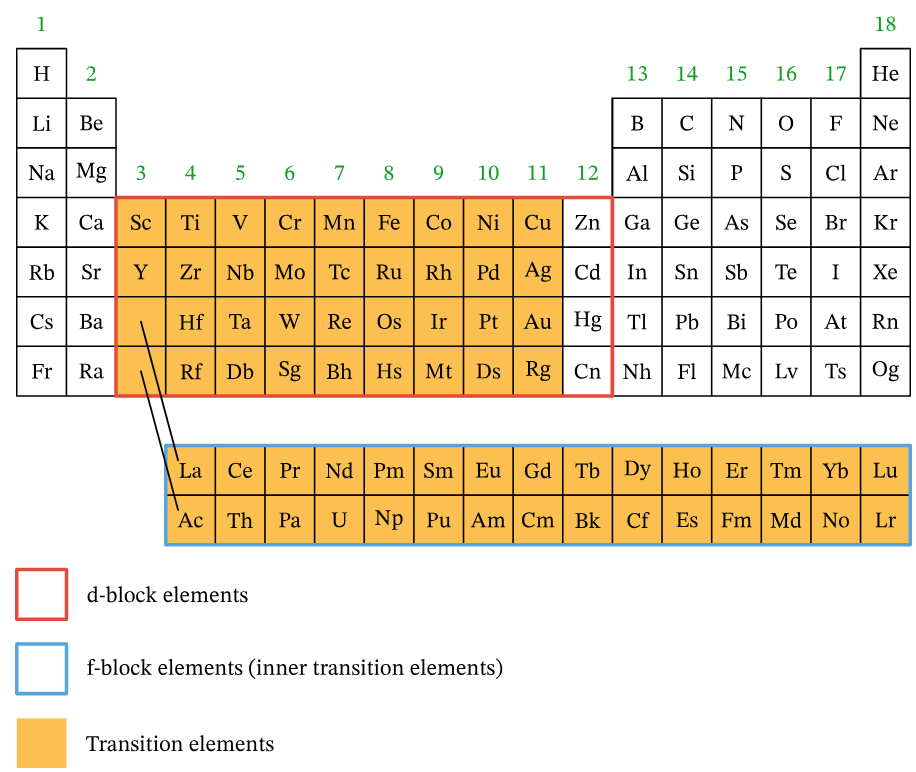
The Periodic Table S3.1.8 Properties of Transition Elements IB DP Chemistry Study Notes - New Syllabus 2026, 2027 & 2028
The periodic table: Classification of elements- IB DP Chemistry- Study Notes
IITian Academy excellent Introduction to the Particulate Nature of Matter – Study Notes and effective strategies will help you prepare for your IB DP Chemistry 2025 exam.
- IB DP Chemistry 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Chemistry 2026, 2027 & 2028 SL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
- IB DP Chemistry 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 1
- IB DP Chemistry 2026, 2027 & 2028 HL- IB Style Practice Questions with Answer-Topic Wise-Paper 2
Structure 3.1.8 – Transition Elements
Structure 3.1.8 – Transition Elements
What Are Transition Elements?
Transition elements are found in the d-block of the periodic table, typically from Groups 3 to 12. According to the IUPAC definition, a transition element is an element that forms at least one stable ion with an incomplete d-subshell.
Electron Configuration and d-Shell
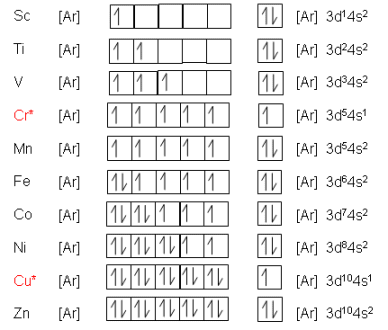
- Transition elements have the general electronic configuration: \( (n-1)d^{1-9}ns^2 \) or \( (n-1)d^{1-10}ns^1 \).
- Their distinguishing feature is the presence of an incomplete d-subshell in at least one of their oxidation states.
- For example, iron (Fe) has the ground state configuration: \( [\text{Ar}]\,3d^6\,4s^2 \), and its ions Fe²⁺ and Fe³⁺ have configurations with incomplete 3d sublevels.
Definition of a Transition Element
- To qualify as a transition element, the element must form an ion in which the d-subshell is not full (i.e., not d¹⁰).
- Examples:
- Iron (Fe): Fe²⁺ → 3d⁶, Fe³⁺ → 3d⁵ → both are transition ions.
- Copper (Cu): Cu⁺ → 3d¹⁰ → not considered a transition ion, but Cu²⁺ → 3d⁹ → is a transition ion.
- Zinc (Zn): Not a transition element. Although it’s in the d-block, Zn²⁺ has the electron configuration 3d¹⁰ — a complete d-subshell.
Example
Which of the following is not considered a transition element?
A. Fe B. Cu C. Zn D. Cr
▶️Answer/Explanation
- Correct answer: C. Zn
- Reason: Zinc forms Zn²⁺ with a 3d¹⁰ configuration (full d-subshell), so it does not meet the definition of a transition element.
- Fe, Cu, and Cr all form ions with incomplete d-subshells, so they are transition elements.
Properties of Transition Elements
Transition metals are well-known for exhibiting a range of distinctive chemical and physical properties due to their partially filled d orbitals. Below is a breakdown of each property with explanations and examples.
1. Variable Oxidation States
- Transition metals can form multiple stable ions by losing different numbers of d and s electrons.
- This leads to a variety of oxidation states that depend on the specific compound and ligands involved.
- Examples:
- Iron forms Fe²⁺ and Fe³⁺
- Manganese forms Mn²⁺ to Mn⁷⁺
- Chromium forms Cr²⁺, Cr³⁺, and Cr⁶⁺

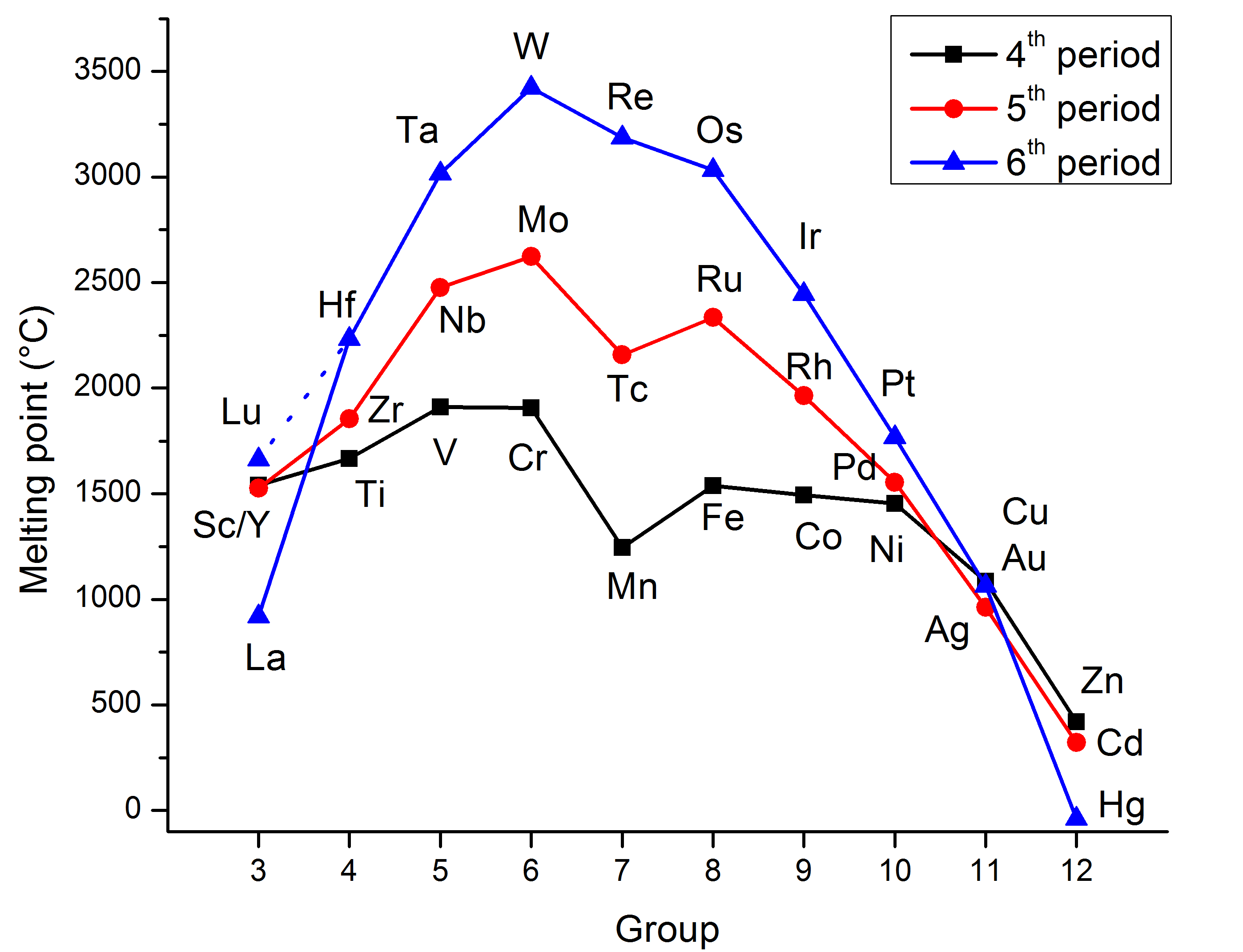
2. High Melting Points
- Transition metals generally have high melting points due to strong metallic bonding.
- The presence of delocalised d electrons enhances metallic bonding strength across the lattice.
- Example: Iron (Fe) has a melting point of 1538°C.
3. Magnetic Properties
- These arise from unpaired electrons in d orbitals.
- The more unpaired electrons, the stronger the magnetic moment.
- Magnetic behaviour depends on electron configuration:
- Fe, Co, Ni show ferromagnetism due to domain alignment.
- Paramagnetism (weak attraction) results from unpaired electrons (not assessed in IB).
- Example: Fe (3d⁶) has four unpaired electrons → strongly magnetic.
4. Catalytic Properties
- Transition metals act as excellent catalysts due to:
- Variable oxidation states — allow them to gain/lose electrons easily in redox reactions.
- Ability to adsorb reactants onto their surface, weakening bonds and lowering activation energy.
- Examples:
- Iron in the Haber Process: \( \text{N}_2 + 3\text{H}_2 \rightleftharpoons 2\text{NH}_3 \)
- Vanadium(V) oxide in the Contact Process: \( \text{SO}_2 + \tfrac{1}{2}\text{O}_2 \rightarrow \text{SO}_3 \)
- Nickel in hydrogenation of alkenes
Types of Catalysis
- Heterogeneous: Catalyst is in a different phase from the reactants.
- Example: Fe in the Haber Process for ammonia synthesis.
- Homogeneous: Catalyst is in the same phase as the reactants.
- Example: Fe²⁺ in redox reactions in biological systems (e.g., enzymes).
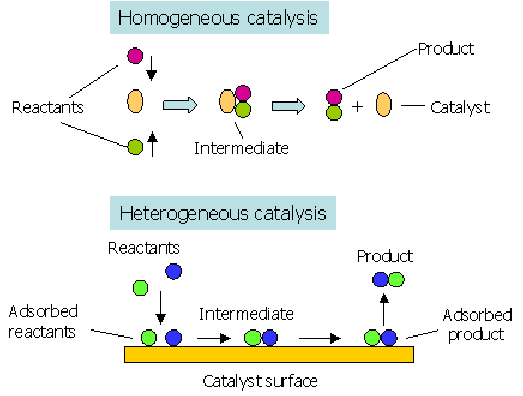
Catalytic Converters
- Use platinum and rhodium metals to convert toxic gases (CO, NOₓ, hydrocarbons) into less harmful products.
- Structured on a ceramic honeycomb to maximise surface area and reduce cost.
Biological Catalysts
- Transition metals like Fe play a role in enzymes and oxygen transport.
- Example: Haemoglobin contains an Fe²⁺ ion in a porphyrin ring and binds O₂ reversibly. Each haemoglobin molecule can transport 4 oxygen molecules via coordinate bonding.
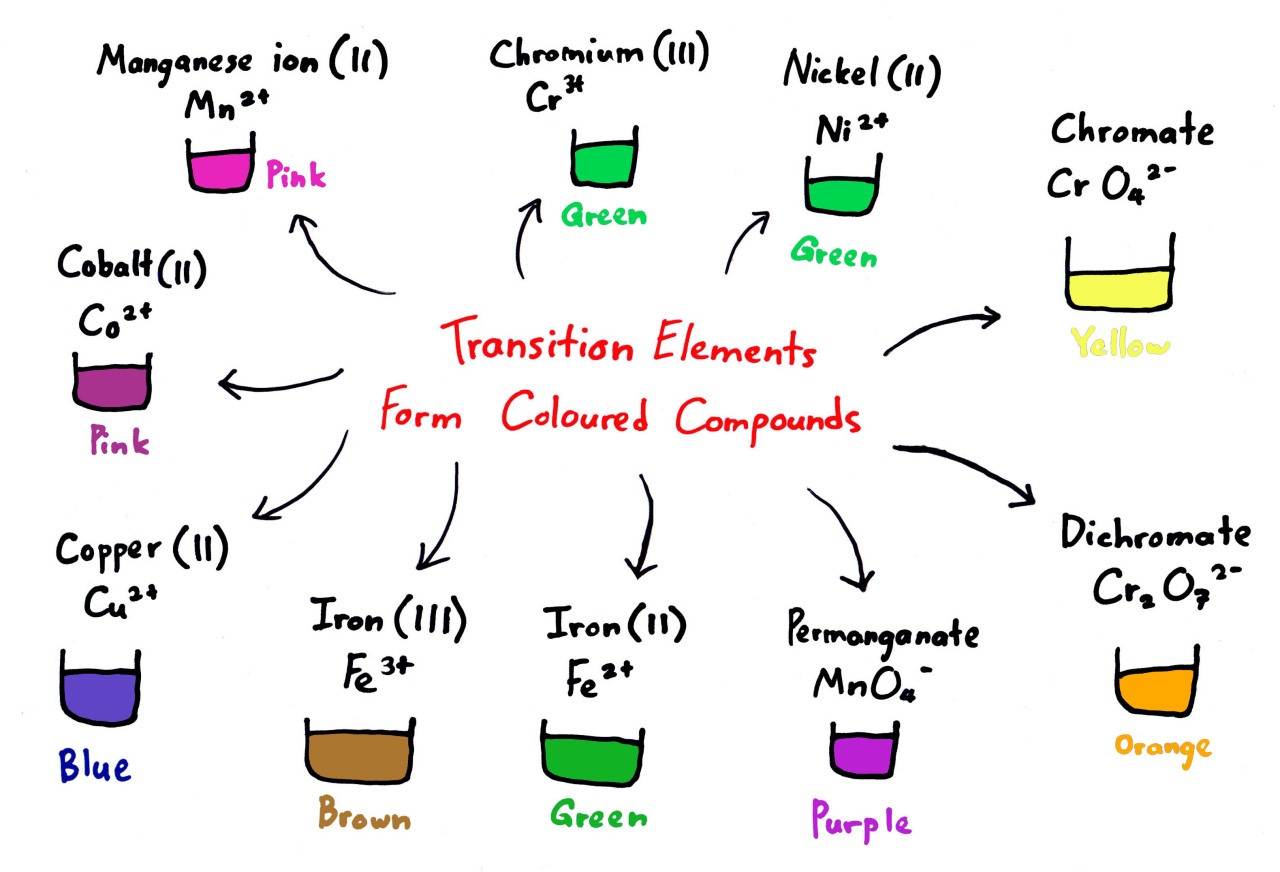
5. Formation of Coloured Compounds
- Transition metal ions absorb certain wavelengths of visible light.
- This occurs due to d–d transitions — electrons are excited between split d orbitals in the presence of ligands.
- The complementary colour of the absorbed light is observed.
- Colour depends on:
- Identity of the metal
- Oxidation state
- Type of ligand
- Examples:
- \( \text{Cu}^{2+} \) → Blue solution
- \( \text{Fe}^{3+} \) → Yellow-brown solution
- \( \text{Cr}_2\text{O}_7^{2-} \) → Orange
- \( \text{MnO}_4^- \) → Purple
6. Formation of Complex Ions with Ligands
- Transition metal ions form complex ions by accepting lone pairs from ligands.
- Ligands form coordinate covalent bonds with the central metal ion.
- Complex ion properties depend on:
- Coordination number (number of ligand bonds)
- Ligand type (e.g., H₂O, NH₃, Cl⁻, CN⁻)
- Examples:
- \([ \text{Cu(H}_2\text{O)}_6 ]^{2+}\) — Blue octahedral complex
- \([ \text{Cr(NH}_3)_6 ]^{3+}\) — Purple octahedral complex
- \([ \text{Fe(CN)}_6 ]^{3-} \) — Yellow complex
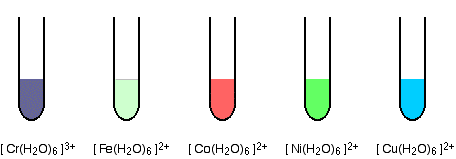
Summary Table: Recognizing Transition Element Properties
| Property | Description | Examples |
|---|---|---|
| Variable Oxidation States | Forms ions with different charges | Fe²⁺, Fe³⁺, Cr²⁺, Cr⁶⁺ |
| High Melting Points | Strong metallic bonding with delocalized d electrons | Fe, Ni, Cr |
| Magnetic Properties | Unpaired d electrons generate magnetism | Fe, Co, Ni |
| Catalytic Properties | Enable redox or surface reactions | Fe (Haber), V₂O₅ (Contact), Ni (hydrogenation) |
| Coloured Compounds | d-d transitions absorb visible light | \( \text{Cr}_2\text{O}_7^{2-} \), \( \text{Cu}^{2+} \) |
| Complex Ion Formation | Coordinate bonds with ligands | \([ \text{Fe(CN)}_6 ]^{3-} \), \([ \text{Cr(H}_2\text{O)}_6 ]^{3+} \) |
Example
An unknown metal forms the following ions: X²⁺ (green), X³⁺ (purple). It also acts as a catalyst in the hydrogenation of alkenes and forms a hexaaqua complex in water. Which type of element is X most likely to be?
▶️ Answer/Explanation
- The element shows variable oxidation states (X²⁺ and X³⁺).
- It forms coloured ions (green and purple).
- It catalyzes chemical reactions (hydrogenation).
- It forms a complex ion with water — [X(H₂O)₆]²⁺ or [X(H₂O)₆]³⁺.
- Conclusion: X is a transition metal.
Example
Write the formula and coordination number of the complex ion formed when copper(II) ions are surrounded by four ammonia molecules and two water molecules.
▶️ Answer/Explanation
- Copper(II) ion: Cu²⁺
- Ligands: 4 NH₃ and 2 H₂O
- Total number of ligands = 6 → Coordination number = 6
- Complex ion formula: [Cu(NH₃)₄(H₂O)₂]²⁺
