The diagram represents an atom of an element.
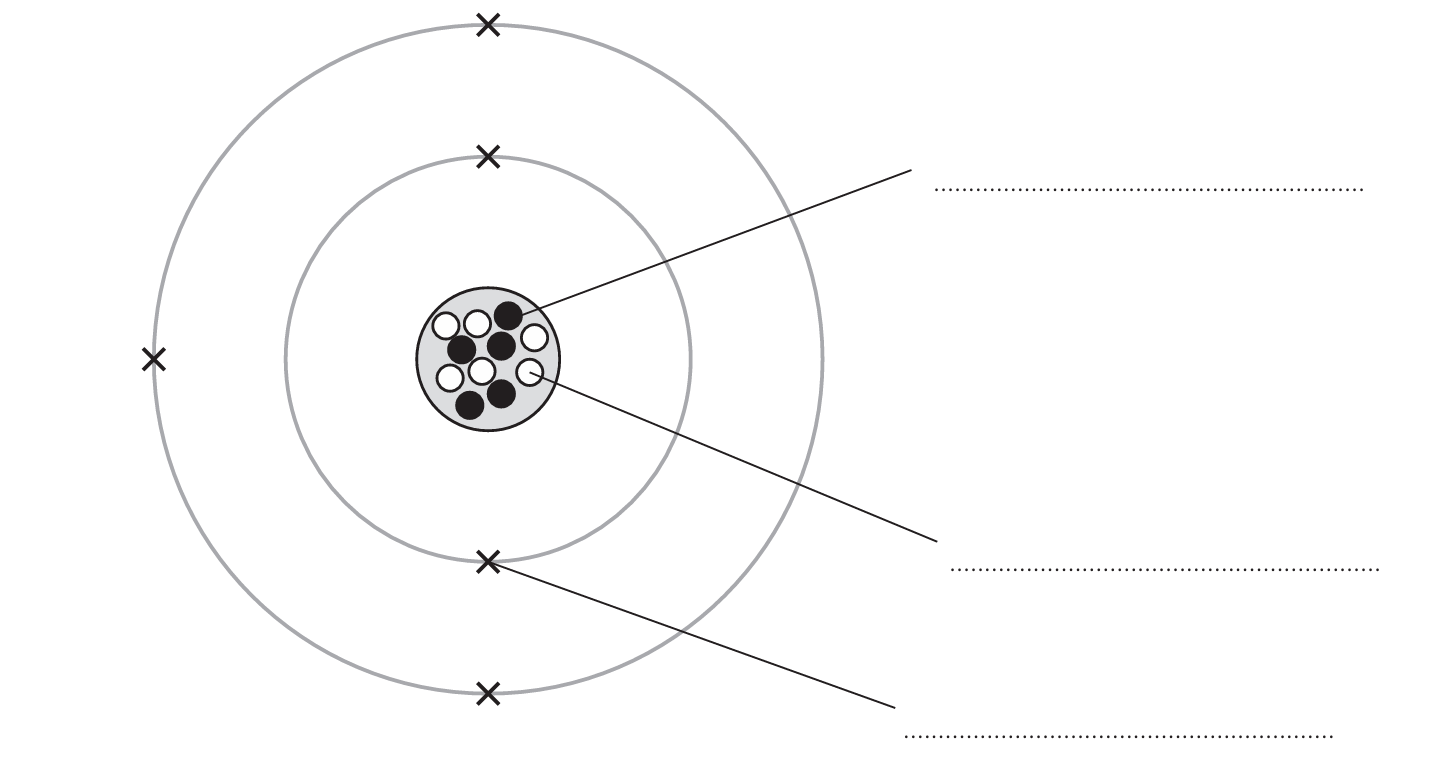
(a) Label the three subatomic particles on the diagram.
(b) Use information from the diagram to complete the table for this element.

(c) Give the name of this element.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(d): The Periodic Table — parts (b) and (c), determining group, period, and element identity from atomic structure.
▶️ Answer/Explanation
(a) Labels on the diagram should be:
- In the nucleus: proton and neutron (must be in the correct order).
- In the electron shells/orbiting: electron.
Marking Note: If proton and neutron are in the wrong order, allow one mark for M1 & M2.
(b) Completed table:
| mass number | 11 |
| group number | 3 |
| period number | 2 |
| electronic configuration | 2,3 |
Explanation:
• Mass number = protons + neutrons. From diagram: 5 protons + 6 neutrons = 11.
• Electronic configuration: 5 electrons total. First shell holds 2, second shell holds the remaining 3: 2,3.
• Group number: For main group elements, the group number is equal to the number of electrons in the outer shell. Outer shell has 3 electrons, so Group 3.
• Period number: The period number is equal to the number of occupied electron shells. This atom has electrons in two shells (first and second), so Period 2.
(c) boron (ALLOW B)
Explanation: The atom has 5 protons. The atomic number (number of protons) defines the element. The element with atomic number 5 is boron (symbol B).
A small piece of lithium is added to a trough of water.
(a) State two observations made when lithium reacts with water.
(b) What are the products of the reaction?
- A) lithium hydroxide and hydrogen
- B) lithium hydroxide and oxygen
- C) lithium oxide and hydrogen
- D) lithium oxide and oxygen
(c) After the reaction is complete, a few drops of universal indicator are added to the solution in the trough.
(c) (i) Explain the colour and pH of the solution in the trough.
(c) (ii) Give the formula of the ion responsible for this pH.
(d) Lithium ions can be identified using a flame test.
What flame colour indicates the presence of lithium ions?
- A) lilac
- B) orange
- C) red
- D) yellow
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(f): Acids, alkalis and titrations — part (c)(i), (c)(ii)
• 2(h): Chemical tests — part (d)
• 1(e): Chemical formulae, equations and calculations — part (b)
▶️ Answer/Explanation
(a) Any two from:
- Effervescence / fizzing / bubbles
- Lithium moves (on the surface)
- Lithium floats
- Lithium gets smaller / disappears
The reaction is vigorous. Lithium, being less dense than water, floats and darts around on the surface as hydrogen gas is produced. The solid lithium gradually reacts away.
(b) A (lithium hydroxide and hydrogen)
The general reaction for an alkali metal (M) with water is: \(2M(s) + 2H_2O(l) \rightarrow 2MOH(aq) + H_2(g)\). For lithium: \(2Li(s) + 2H_2O(l) \rightarrow 2LiOH(aq) + H_2(g)\). Lithium oxide (\(Li_2O\)) is not the product in this aqueous reaction.
(c) (i) An explanation that links the following three points:
- (Colour of solution is) blue / purple / indigo / violet.
- (pH value or range between) 10 and 14.
- (As the solution is) alkaline / an alkali / basic.
The universal indicator turns blue-purple because lithium hydroxide (\(LiOH\)) is a strong alkali. It dissociates completely in water to produce hydroxide ions (\(OH^-\)), resulting in a high pH (strongly alkaline).
(c) (ii) \(OH^-\) (Hydroxide ion)
Accept \(HO^-\). This ion is responsible for the alkaline properties and high pH of the solution.
(d) C (red)
A is incorrect as lilac is the flame colour for potassium ions (\(K^+\)).
B is incorrect as orange-red is the flame colour for calcium ions (\(Ca^{2+}\)).
D is incorrect as yellow is the flame colour for sodium ions (\(Na^+\)).
The characteristic red flame for lithium ions (\(Li^+\)) is a key identification test, as specified in the syllabus (2.46).
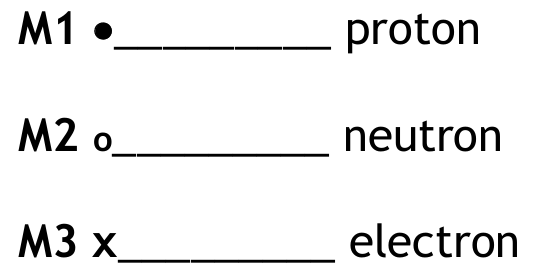
A student uses paper chromatography to investigate the dyes in five different inks, V, W, X, Y and Z.
The chromatogram shows the results of the investigation.
(a) Explain why the start line on the paper is drawn in pencil rather than in ink.
(b) Explain which two inks contain the dye that is most soluble in the solvent.
(c) Explain how the chromatogram shows that:
- W contains only one dye
- V may contain more than one dye
(d) Calculate the \( R_f \) value for the dye in W. Give your answer to two significant figures.
Most-appropriate topic codes (Edexcel IGCSE Chemistry – 4CH1):
▶️ Answer/Explanation
(a) An explanation linking the following two points:
- The ink (dyes) would dissolve in the solvent used in chromatography / the pencil mark (graphite) will not dissolve in the solvent.
- If ink were used, it would travel/smudge/run with the solvent, producing spots that would interfere with the results / make it impossible to identify the dyes in the test inks or calculate accurate \( R_f \) values. The pencil mark remains fixed at the start line.
(b) An explanation linking the following two points:
- Inks X and Z.
- Both contain a dye that has traveled the furthest distance from the start line towards the solvent front. In chromatography, a substance that is more soluble in the solvent will be carried further up the paper.
(c) An explanation linking the following points:
- For W: It produces only one distinct spot on the chromatogram. Since each dye in a mixture should separate into its own spot, a single spot indicates the presence of only one dye.
- For V: It has not moved from the start line (it is insoluble in the solvent used). Because it has not separated at all, it is impossible to tell from this chromatogram whether it contains one or more dyes. All components remain together at the origin, so it may contain more than one dye.
(d) Calculation of the \( R_f \) value:
The \( R_f \) value is calculated using the formula: \[ R_f = \frac{\text{distance moved by substance (spot)}}{\text{distance moved by solvent front}} \]
- Measure distance from start line to centre of spot W: \( 2.2 \, \text{cm} \) to \( 2.3 \, \text{cm} \) (allow within this range).
- Measure distance from start line to solvent front: \( 6.9 \, \text{cm} \).
- Calculation: \( R_f = \frac{2.2}{6.9} \) or \( \frac{2.3}{6.9} \).
- Result: \( \frac{2.2}{6.9} = 0.3188… \) or \( \frac{2.3}{6.9} = 0.3333… \).
- Answer to two significant figures: \( \mathbf{0.32} \) or \( \mathbf{0.33} \).
Note: Values are based on typical measurements from the chromatogram. In an exam, actual measurements would be taken from the diagram provided.
Calcium phosphate is an ionic compound with the formula \( \text{Ca}_3(\text{PO}_4)_2 \).
(a) (i) What is the total number of atoms in the formula \( \text{Ca}_3(\text{PO}_4)_2 \)?
- A) 8
- B) 11
- C) 13
- D) 19
(a) (ii) What are the correct charges on the ions in calcium phosphate?
- A) \( \text{Ca}^{2+} \) and \( \text{PO}_4^{2-} \)
- B) \( \text{Ca}^{2+} \) and \( \text{PO}_4^{3-} \)
- C) \( \text{Ca}^{3+} \) and \( \text{PO}_4^{2-} \)
- D) \( \text{Ca}^{3+} \) and \( \text{PO}_4^{3-} \)
(b) (i) Calculate the relative formula mass (\( M_r \)) of \( \text{Ca}_3(\text{PO}_4)_2 \).
(b) (ii) Calculate the percentage by mass of calcium in calcium phosphate.
(c) Calcium hydroxide can react with phosphoric acid to form calcium phosphate.
Complete the equation for this reaction.
______ \( \text{Ca(OH)}_2 \) + ______ \( \text{H}_3\text{PO}_4 \) → ______ \( \text{Ca}_3(\text{PO}_4)_2 \) + ______ \( \text{H}_2\text{O} \)
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(f): Ionic bonding — part (a)(ii)
• 2(g): Acids, bases and salt preparations — part (c)
▶️ Answer/Explanation
(a)(i) C (13)
Explanation: The formula \( \text{Ca}_3(\text{PO}_4)_2 \) contains:
3 Ca atoms
Inside the phosphate group \( \text{PO}_4 \): 1 P and 4 O atoms. Since the subscript ‘2’ is outside the bracket, we multiply the atoms inside by 2: 2 P and 8 O atoms.
Total atoms = 3 (Ca) + 2 (P) + 8 (O) = 13.
A is incorrect as there are not 8 atoms.
B is incorrect as there are not 11 atoms.
D is incorrect as there are not 19 atoms.
(a)(ii) B (\( \text{Ca}^{2+} \) and \( \text{PO}_4^{3-} \))
Explanation: Calcium is in Group 2, so it forms a \( \text{Ca}^{2+} \) ion. The phosphate ion \( \text{PO}_4^{3-} \) has a 3- charge. For the compound to be neutral, three \( \text{Ca}^{2+} \) ions (total charge +6) must balance two \( \text{PO}_4^{3-} \) ions (total charge -6).
A is incorrect as the charges are incorrect.
C is incorrect as the charges are incorrect.
D is incorrect as the charges are incorrect.
(b)(i) \( M_r = 310 \)
Working:
Using \( A_r \) values: Ca = 40, P = 31, O = 16.
\( M_r = (3 \times 40) + (2 \times 31) + (8 \times 16) \)
\( = 120 + 62 + 128 \)
\( = 310 \)
(b)(ii) Percentage by mass of calcium = \( 38.7\% \) (or \( 39\% \))
Working:
Mass of calcium in formula = \( 3 \times 40 = 120 \).
Total \( M_r = 310 \).
Percentage = \( \frac{120}{310} \times 100 \approx 38.7\% \).
Allow ecf for \(M_r\) incorrectly calculated. Allow any number of significant figures (e.g., 38.7 for 2 marks or 12.9 for 1 mark if \(M_r\) was incorrectly calculated as 930).
(c) \( 3\text{Ca(OH)}_2 + 2\text{H}_3\text{PO}_4 \rightarrow 1\text{Ca}_3(\text{PO}_4)_2 + 6\text{H}_2\text{O} \)
Explanation: The balanced equation ensures the number of atoms of each element is equal on both sides. The coefficients are derived by balancing Ca, P, O, and H atoms. The ‘1’ in front of \( \text{Ca}_3(\text{PO}_4)_2 \) is often omitted.
Allow multiples and fractions.
Butane (\(C_4H_{10}\)) is an alkane.
(a) Explain why butane is a saturated hydrocarbon.
(b) (i) When butane burns completely in oxygen, carbon dioxide and water are produced. Give a balanced chemical equation for this combustion reaction.
(ii) Incomplete combustion can occur when the oxygen supply is limited. One product of the incomplete combustion of butane is carbon monoxide. Give the name of another product of this incomplete combustion.
(iii) State why carbon monoxide is poisonous to humans.
(c) \(C_4H_{10}\) exists as two isomers.
(i) State what is meant by the term isomers.
(ii) Draw the displayed formula of each isomer.

(d) Explain why hexane (\(C_6H_{14}\)) has a higher boiling point than butane (\(C_4H_{10}\)).
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(a): Introduction — part (c)(i)
▶️ Answer/Explanation
(a)
- Butane contains only hydrogen and carbon atoms (it is a hydrocarbon).
- It contains only carbon–carbon single bonds (and carbon–hydrogen single bonds).
- Therefore, it is saturated (it contains the maximum possible number of hydrogen atoms per carbon, with no double or triple bonds).
Mark scheme note: M1 – contains H and C only. M2 – “only”. M3 – contains only single bonds.
(b)(i)
\(2C_4H_{10} + 13O_2 \rightarrow 8CO_2 + 10H_2O\)
Balanced equation showing complete combustion. Accept multiples or fractions for balancing.
(b)(ii)
Carbon (soot/particulates) OR Water (vapour).
Incomplete combustion produces carbon (soot), carbon monoxide, and water vapour.
(b)(iii)
Carbon monoxide binds irreversibly to haemoglobin in red blood cells, forming carboxyhaemoglobin. This reduces the blood’s capacity to carry oxygen around the body, leading to oxygen deprivation in tissues (asphyxiation).
Mark scheme: “carbon monoxide reduces the capacity of blood to transport oxygen”.
(c)(i)
- Isomers are molecules that have the same molecular formula (same number and type of atoms).
- But they have different structural formulae / different arrangements of atoms.
(c)(ii)
Displayed formulae for the two isomers of butane:
1. Butane (n-butane):
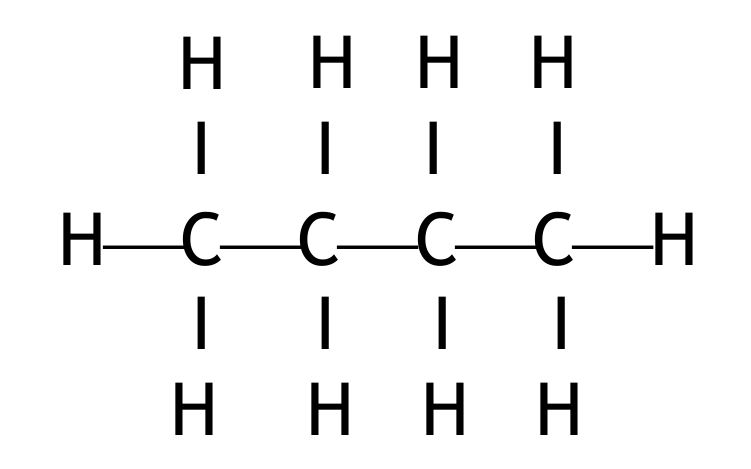
2. Methylpropane (isobutane):
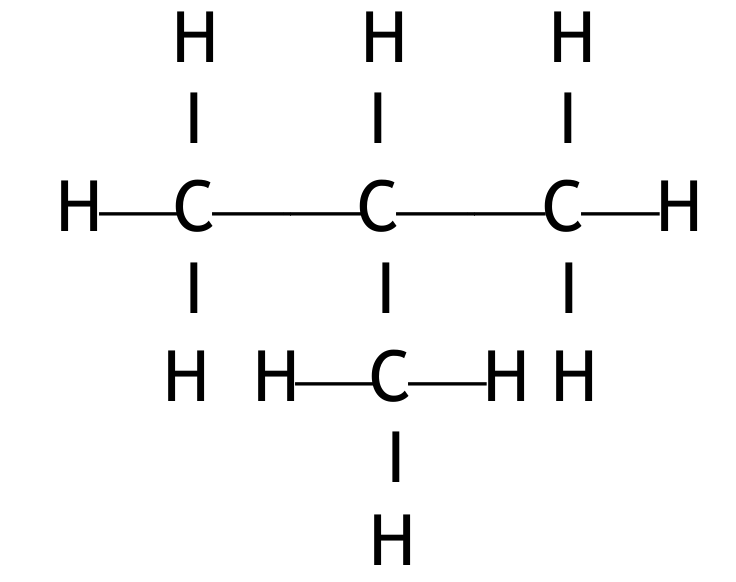
Both must show all atoms and all bonds.
(d)
- Hexane (\(C_6H_{14}\)) has a longer carbon chain/more atoms/higher molecular mass than butane (\(C_4H_{10}\)).
- This results in stronger intermolecular forces (specifically London dispersion/van der Waals forces) between hexane molecules.
- Therefore, more energy is required to overcome these stronger forces during boiling.
- Hence, hexane has a higher boiling point than butane.
Mark scheme note: M1 – hexane is larger/longer chain. M2 – stronger intermolecular forces. M3 – more energy needed to overcome forces. DO NOT mention breaking covalent bonds.
(a) When iron rusts, iron(III) oxide forms. State two conditions needed for iron to rust.
(b) What is the name of the process used to coat iron with zinc?
(c) What is the correct order of reactivity of these four metals?

(d) Describe a test to show that a solution contains Fe\(^{3+}\) ions.
(e) A sample of iron(III) oxide reacts with excess carbon monoxide. This is the equation for the reaction.
\[ \text{Fe}_2\text{O}_3 + 3\text{CO} \rightarrow 2\text{Fe} + 3\text{CO}_2 \]
(i) Explain why this is a redox reaction.
(ii) Calculate the minimum mass of iron(III) oxide needed to produce a theoretical yield of 28 g of iron.
\[ [\text{for } \text{Fe}_2\text{O}_3 \, M_r = 160] \]
(iii) The actual yield of iron from this sample is 21 g. Calculate the percentage yield of iron.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(h): Chemical tests — part (d)
• 1(e): Chemical formulae, equations and calculations — part (e)(ii), (e)(iii)
• 2(d): Reactivity series (redox definitions) — part (e)(i)
▶️ Answer/Explanation
(a) Two conditions needed for iron to rust:
1. Oxygen (or air) (ALLOW: air)
2. Water (or moisture/water vapour/steam) (ALLOW: moisture, water vapour, steam)
(b) A (galvanisation)
B is not the answer because oxidation is a type of reaction, not the name of a coating process.
C is not the answer because reduction is a type of reaction, not the name of a coating process.
D is not the answer because sacrificial protection is a principle/mechanism, not the name of the coating process itself.
(c) C (aluminium zinc iron copper)
A is not the correct order of reactivity.
B is not the correct order of reactivity.
D is not the correct order of reactivity.
Knowledge: Aluminium is more reactive than zinc, which is more reactive than iron, which is more reactive than copper.
(d) Test: Add sodium hydroxide solution.
Result: A brown precipitate forms. (ACCEPT: red-brown, orange-brown)
Note: The brown precipitate is iron(III) hydroxide, Fe(OH)\(_3\). This is a standard test for Fe\(^{3+}\) ions.
(e)(i) An explanation that links the following points:
• Iron(III) oxide / Fe\(_2\)O\(_3\) loses oxygen, so it is reduced.
• Carbon monoxide gains oxygen (to form CO\(_2\)), so it is oxidised.
ALTERNATIVE: Fe\(^{3+}\) gains electrons (to become Fe) and is reduced, while carbon in CO loses electrons (or gains oxygen) and is oxidised.
(e)(ii) Calculation:
M1: Amount (moles) of Fe = \( \frac{28}{56} = 0.5 \) mol (M\(_r\) of Fe = 56)
M2: From equation: Fe\(_2\)O\(_3\) → 2Fe, so moles of Fe\(_2\)O\(_3\) = \( \frac{0.5}{2} = 0.25 \) mol
M3: Mass of Fe\(_2\)O\(_3\) = \( 0.25 \times 160 = 40 \) g
Answer: 40 g (correct answer without working scores 3 marks)
(e)(iii) Calculation:
M1: Percentage yield = \( \frac{\text{actual yield}}{\text{theoretical yield}} \times 100 = \frac{21}{28} \times 100 \)
M2: \( = 75\% \) (0.75 scores 1 mark only)
Answer: 75% (correct answer without working scores 2 marks)
A student does an experiment to determine the maximum temperature change when sulfuric acid reacts with sodium hydroxide.
(a) Complete the chemical equation for this neutralisation reaction. Include the state symbols.
\[ 2\mathrm{NaOH(aq)} + \mathrm{H_2SO_4(aq)} \rightarrow \] ______ + ______
(b) This is the student’s method.
- Step 1: add \(50\ \mathrm{cm^3}\) of sodium hydroxide solution to a glass beaker
- Step 2: record the initial temperature of the sodium hydroxide solution
- Step 3: add \(5\ \mathrm{cm^3}\) of dilute sulfuric acid to the beaker
- Step 4: stir the mixture and record the highest temperature reached
The student repeats steps 3 and 4 until a total of \(40\ \mathrm{cm^3}\) of acid has been added.
Explain one way the student could improve the accuracy of the experiment.
(c) The table shows the student’s results.
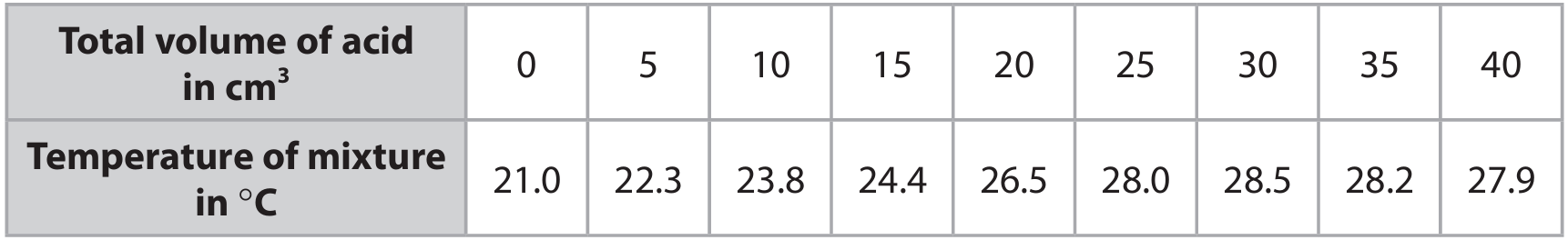
(i) Plot the results on the grid.
(ii) Draw a circle around the anomalous result.
(iii) Draw a straight line of best fit through the first six points, ignoring the anomalous result. Draw another straight line of best fit through the last three points. Make sure that the two lines cross.
(d) The point where the lines cross shows the volume of acid needed to exactly neutralise the alkali and the maximum temperature reached. Use your graph to determine these values.
(e) Explain what might have caused the anomalous result.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 3(a): Energetics — parts (b), (c), (d) (temperature changes in neutralisation)
• Practical Skills (Section 2: Chemistry content, experimental skills) — parts (c), (e) (graph plotting, identifying anomalies, suggesting improvements)
▶️ Answer/Explanation
(a)
\[ 2\mathrm{NaOH(aq)} + \mathrm{H_2SO_4(aq)} \rightarrow \mathrm{Na_2SO_4(aq)} + 2\mathrm{H_2O(l)} \]
Explanation: A balanced neutralisation reaction producing sodium sulfate and water. State symbols indicate aqueous solutions and liquid water.
(b)
• Use a polystyrene cup instead of a glass beaker.
• Reason: Polystyrene is a better thermal insulator, reducing heat loss to the surroundings, leading to a more accurate measurement of the temperature change.
Alternative answer: Place a lid on the beaker to minimise heat loss by evaporation/convection.
(c) (i), (ii), (iii)
Graph plotted with the following key features:
• Points plotted correctly from the table.
• The point at 15 cm³ (temperature 24.4 °C) is circled as anomalous.
• First line of best fit drawn through points at 0, 5, 10, 20, 25, 30 cm³ (rising trend).
• Second line of best fit drawn through points at 30, 35, 40 cm³ (slight downward trend).
• The two lines intersect.
(d)
Volume of acid ≈ 27.5 cm³
Maximum temperature ≈ 28.6 °C
Note: Values are read from the intersection point on the graph, as per the mark scheme (approximately 27.5 cm³ and 28.6 °C).
(e)
• The temperature was recorded too early (before the mixture was fully mixed or before it reached its maximum temperature).
OR
• The mixture was not stirred properly before the temperature was taken, leading to an uneven temperature distribution and a lower reading.
Both explanations result in the recorded temperature being lower than the true maximum for that volume of acid.
Diamond and graphite are two naturally-occurring forms of carbon. They both have giant covalent structures.
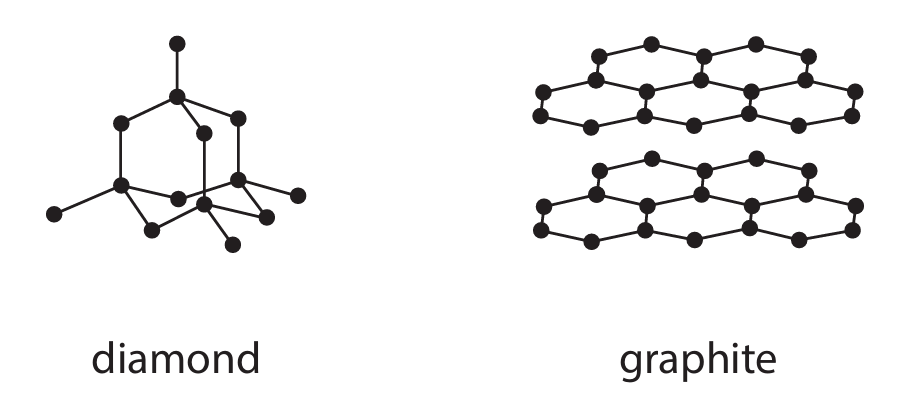
(a) Explain, with reference to its bonding, why diamond has a high melting point.
(b) Explain why graphite is soft and is a conductor of electricity. Refer to structure and bonding in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1 Specification):
▶️ Answer/Explanation
(a) Why diamond has a high melting point:
• M1: The structure of diamond is a giant covalent lattice held together by strong covalent bonds between carbon atoms.
• M2: To melt diamond, a very large number of these strong covalent bonds must be broken.
• M3: Breaking these numerous, strong bonds requires a large amount of thermal energy, which corresponds to a very high melting point.
Key Chemistry: Diamond’s tetrahedral network, where each carbon atom is bonded to four others via covalent bonds, results in a rigid, three-dimensional structure that is extremely resistant to heat.
(b) Why graphite is soft and conducts electricity:
Why graphite is soft:
• M1 & M5: In graphite, each carbon atom forms three strong covalent bonds with other carbon atoms, creating layers of hexagonal rings.
• M2 & M4: These layers are held together by weak intermolecular forces (London dispersion forces), not by covalent bonds.
• M3: Because the forces between the layers are weak, the layers can slide easily over one another when a force is applied, making graphite soft and slippery (useful as a lubricant).
Why graphite conducts electricity:
• M5 & M6: Each carbon atom in graphite uses only three of its four outer electrons for bonding within a layer. The fourth electron from each atom is delocalised and is free to move throughout the entire layer.
• M7: These delocalised electrons are free to move and carry charge. When a voltage is applied, these mobile electrons can flow through the structure, allowing graphite to conduct electricity.
Key Chemistry: The combination of strong covalent bonding within layers and weak forces between them, along with the presence of a ‘sea’ of delocalised electrons, gives graphite its unique set of properties, distinguishing it from diamond.
This question is about organic compounds.
(a) Chloroethene (\(C_2H_3Cl\)) is a covalent molecule that is used to make poly(chloroethene). The displayed formula of chloroethene is:
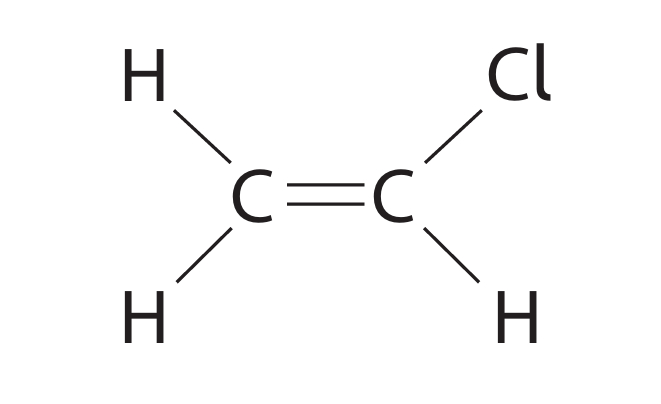
(a) (i) Draw a dot-and-cross diagram of chloroethene. Show only the outer shell electrons.
(a) (ii) Describe, in terms of electrostatic attraction, what is meant by a covalent bond.
(a) (iii) Chloroethene is a monomer. Draw the displayed formula of the repeat unit of poly(chloroethene).
(a) (iv) Poly(chloroethene) is non-biodegradable. There are two main methods used to dispose of poly(chloroethene). Describe one problem caused by each method.
(b) An organic compound contains this percentage composition by mass:
\( C = 22.0\% \) \( H = 4.6\% \) \( Br = 73.4\% \)
Calculate the empirical formula of this compound.
(c) Ethane and ethene undergo different types of reaction with bromine. Describe the differences between these two reactions.
Most-appropriate topic codes (Edexcel IGCSE Chemistry – 4CH1):
• 4(h): Synthetic polymers — parts (a)(iii), (a)(iv)
• 1(e): Chemical formulae, equations and calculations — part (b)
• 4(d): Alkenes & 4(c): Alkanes — part (c)
▶️ Answer/Explanation
(a)(i)
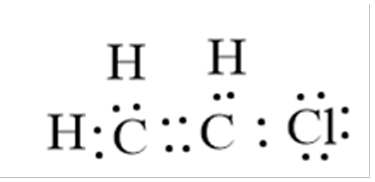
A correct dot-and-cross diagram showing the double bond between the two carbon atoms and single bonds to H and Cl. Only outer shell electrons (valency electrons) are shown. For example, Carbon (Group 4) has 4 outer electrons, Hydrogen has 1, Chlorine (Group 7) has 7. The double bond is represented by two shared pairs of electrons between the carbons.
(a)(ii)
A covalent bond is the electrostatic attraction between a shared pair of electrons and the nuclei of the bonded atoms. The negatively charged electrons are attracted to the positively charged nuclei, holding the atoms together.
(a)(iii)
The repeat unit of poly(chloroethene) (PVC) is derived from the monomer chloroethene by opening the double bond. Its displayed formula is:
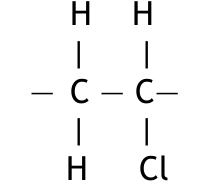
It is shown with extension bonds at each end to indicate it is a repeating unit within a long chain.
(a)(iv)
Method 1: Landfill
Problem: Poly(chloroethene) is inert and non-biodegradable. It takes up valuable space in landfill sites and takes a very long time to decompose (hundreds of years).
Method 2: Burning (Incineration)
Problem: When burned, poly(chloroethene) can produce toxic hydrogen chloride gas (HCl) and other harmful gases, contributing to air pollution and acid rain.
(b)
Step 1: Assume 100g of compound, so masses are C=22.0g, H=4.6g, Br=73.4g.
Step 2: Find moles of each element.
Moles of C = \( \frac{22.0}{12.0} = 1.833 \)
Moles of H = \( \frac{4.6}{1.0} = 4.6 \)
Moles of Br = \( \frac{73.4}{79.9} \approx 0.919 \)
Step 3: Divide by the smallest number of moles (0.919).
C: \( \frac{1.833}{0.919} \approx 2.0 \)
H: \( \frac{4.6}{0.919} \approx 5.0 \)
Br: \( \frac{0.919}{0.919} = 1.0 \)
Step 4: The simplest whole number ratio is C : H : Br = 2 : 5 : 1.
Therefore, the empirical formula is C\(_2\)H\(_5\)Br.
(c)
The reactions differ in type, conditions, observation, and products.
Ethene (alkene):
• Type: Rapid addition reaction.
• Condition: Occurs readily at room temperature.
• Observation: Bromine water is decolorized immediately (orange/brown to colorless).
• Product: Forms 1,2-dibromoethane (C\(_2\)H\(_4\)Br\(_2\)), a single saturated molecule.
Ethane (alkane):
• Type: Slow substitution reaction.
• Condition: Requires ultraviolet (UV) light as a catalyst.
• Observation: Bromine water decolorizes very slowly or not visibly under normal conditions; stays orange.
• Product: Forms bromoethane (C\(_2\)H\(_5\)Br) and hydrogen bromide (HBr) gas.
A student uses this apparatus to investigate the energy content of different fuels.
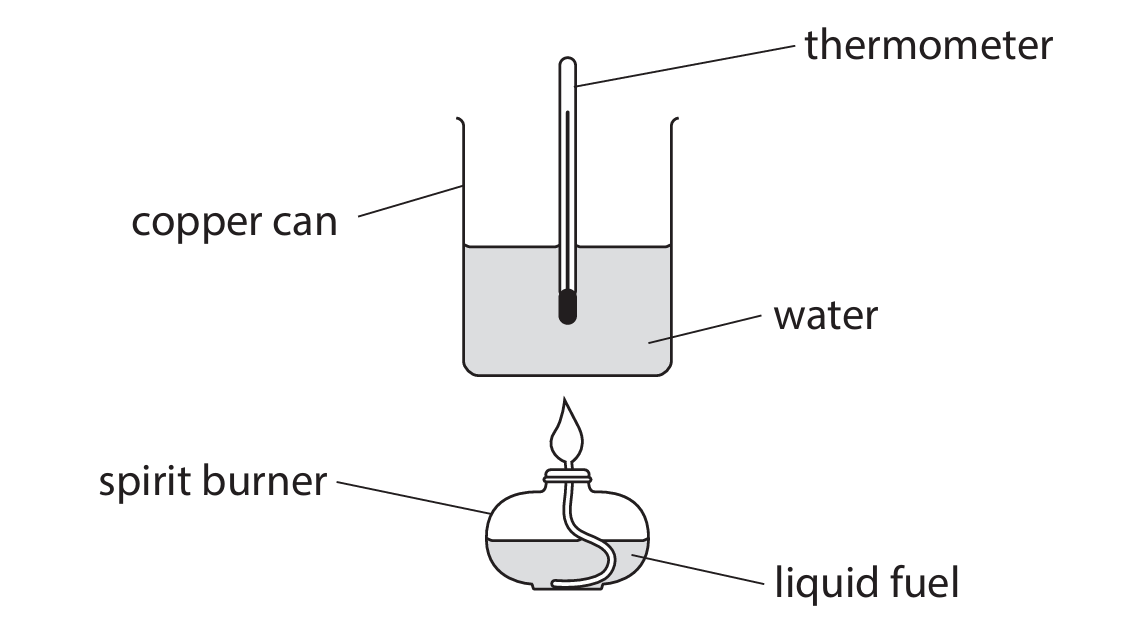
(a) This is the student’s method:
- pour some water into the copper can
- record the mass of the spirit burner and fuel
- measure the initial temperature of the water
- place the spirit burner under the copper can and light the burner
- stop heating the water when the temperature reaches 30°C
- record the new mass of the spirit burner and fuel
The student repeats the experiment with different fuels.
Explain two variables the student should control to make this a valid test.
(b) In one of the experiments, the student uses ethanol as the fuel. These are the student’s results for ethanol.

(b)(i) Calculate the value of heat energy change (\(Q\)) in joules.
[For water, \(c = 4.2 \text{ J g}^{-1} \text{°C}^{-1}\); \(1.0 \text{ cm}^3\) of water has mass = \(1.0 \text{ g}\)]
(b)(ii) Calculate the enthalpy change (\(\Delta H\)) in kJ/mol.
[For ethanol, \(M_r = 46\)]
Include a sign in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(e): Chemical formulae, equations and calculations — Parts (b)(i), (b)(ii) — calculations involving amount of substance and reacting masses.
• 4(e): Alcohols — Part (b)(ii) — ethanol as the fuel.
▶️ Answer/Explanation
(a) An explanation that links any two pairs:
Pair 1:
M1: Use the same volume (or mass) of water.
M2: So that the same amount of heat is required to raise its temperature (makes heat transferred to water comparable).
Pair 2:
M1: Stir the water.
M2: To ensure the temperature is uniform throughout the water (so the thermometer reading is accurate).
Pair 3:
M1: Keep the spirit burner the same distance from the copper can / Keep the wick height the same.
M2: So that the proportion of heat loss to the surroundings is consistent for each fuel.
(b)(i)
Step 1: Calculate the temperature change, \(\Delta T\):
\[ \Delta T = T_{\text{final}} – T_{\text{initial}} = 30 – 18 = 12 \, \text{°C} \]
Step 2: Use the formula \(Q = mc\Delta T\):
Mass of water, \(m = 100 \, \text{g}\) (since \(1 \, \text{cm}^3\) of water has a mass of \(1 \, \text{g}\)).
Specific heat capacity, \(c = 4.2 \, \text{J g}^{-1} \text{°C}^{-1}\).
\[ Q = 100 \times 4.2 \times 12 = 5040 \, \text{J} \]
Answer: \(Q = 5040 \, \text{J}\) (Accept \(5000 \, \text{J}\) to 2 significant figures).
(b)(ii)
Step 1: Calculate the mass of ethanol burned:
\[ \text{Mass of ethanol} = 38.52 – 38.29 = 0.23 \, \text{g} \]
Step 2: Calculate the amount (in moles) of ethanol burned:
Molar mass of ethanol, \(M_r = 46 \, \text{g mol}^{-1}\).
\[ \text{Amount (moles)} = \frac{\text{mass}}{M_r} = \frac{0.23}{46} = 0.0050 \, \text{mol} \]
Step 3: Calculate the enthalpy change per mole (\(\Delta H\)) in J/mol:
The heat energy released (\(Q = 5040 \, \text{J}\)) corresponds to the combustion of 0.0050 mol of ethanol.
\[ \Delta H = \frac{Q}{\text{amount in moles}} = \frac{5040}{0.0050} = 1\,008\,000 \, \text{J mol}^{-1} \]
Step 4: Convert J/mol to kJ/mol:
\[ \Delta H = \frac{1\,008\,000}{1000} = 1008 \, \text{kJ mol}^{-1} \]
Step 5: Include the correct sign (combustion is exothermic):
\[ \Delta H = -1008 \, \text{kJ mol}^{-1} \]
Final Answer: \(\mathbf{-1008 \, kJ \, mol^{-1}}\) (Accept \(-1000 \, \text{kJ mol}^{-1}\) to 2 significant figures).
