This question is about the three states of matter, solid, liquid and gas.
(a) Solids, liquids and gases can be changed from one state to another. The box gives the names of some changes of state.

Use words from the box to answer the questions.
(i) Give the name of the change from liquid to solid.
(ii) Give the name of the change from liquid to gas.
(iii) Give the name of the change from solid to gas.
(b) Describe the arrangement, movement and energy of the particles in a gas.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry):
This section requires students to understand the three states of matter in terms of particle arrangement, movement, and energy, and the interconversions between them.
▶️ Answer/Explanation
(a)(i) freezing
Explanation: The process where a liquid loses thermal energy, particles slow down and arrange into a fixed pattern, forming a solid.
(a)(ii) evaporation
Explanation: The process where a liquid gains thermal energy, particles at the surface gain enough energy to overcome intermolecular forces and escape into the gas phase. (Note: ‘Evaporation’ specifically occurs at the surface and below the boiling point, whereas ‘boiling’ is the rapid change throughout the liquid at its boiling point. The term provided in the box is ‘evaporation’).
(a)(iii) sublimation
Explanation: The direct change from solid to gas without passing through the liquid state. This requires particles in the solid to gain sufficient energy to break all intermolecular forces directly.
(b) A description covering three key points about gas particles:
• Arrangement: The particles are far apart / have large spaces between them / are randomly / irregularly arranged.
• Movement: The particles move rapidly in all directions / have (almost) total freedom of movement / move freely.
• Energy: The particles have high (kinetic) energy / more energy than in solids or liquids.
Full context: In a gas, the particles are widely separated with negligible forces of attraction between them except during collisions. They move randomly and rapidly, colliding with each other and the container walls (which creates gas pressure). They possess the highest kinetic energy of the three states of matter at a given temperature.
This question is about the rusting of iron.
(a) Oxygen is needed for iron to rust.
(i) Name one other substance needed for iron to rust.
(ii) Give the chemical name for rust.
(b) The diagram shows method A and method B, which are both used to prevent iron from rusting.
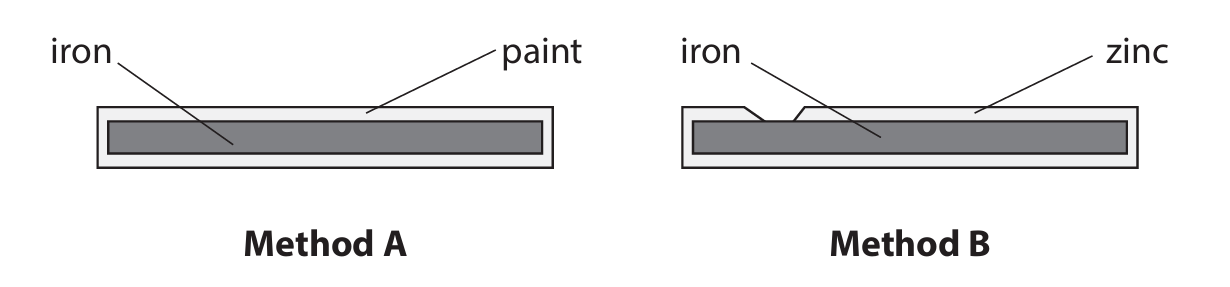
(i) Describe how method A prevents iron from rusting.
(ii) Name method B.
(iii) Explain how method B prevents iron from rusting even when the zinc layer is damaged.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(d) Specification Point 2.18: Know the conditions under which iron rusts.
• 2(d) Specification Point 2.19: Understand how the rusting of iron may be prevented by: barrier methods, galvanising, sacrificial protection.
• 2(d) Specification Point 2.20: Understand the terms oxidation, reduction, redox, oxidising agent, reducing agent in terms of gain or loss of oxygen and loss or gain of electrons. — part (b)(iii)
▶️ Answer/Explanation
(a)(i) Water
Also acceptable: moisture / water vapour / steam.
Rusting is an electrochemical process that requires both oxygen and water (or water vapour) to occur.
(a)(ii) Hydrated iron(III) oxide
Also acceptable: ferric oxide / \(\text{Fe}_2\text{O}_3\).x\(\text{H}_2\text{O}\) (commonly simplified to \(\text{Fe}_2\text{O}_3\)).
Rust is a hydrated form of iron(III) oxide, which forms when iron reacts with oxygen and water.
(b)(i) The paint acts as a barrier / protective layer which prevents oxygen/air and water from reaching the surface of the iron.
(b)(ii) Galvanising / Galvanisation
Also acceptable: sacrificial protection.
Method B involves coating the iron with a layer of zinc, a process known as galvanising.
(b)(iii)
• Zinc is more reactive than iron (it is higher in the reactivity series).
• Because zinc is more reactive, it oxidises / loses electrons / corrodes in preference to iron. The zinc acts as a sacrificial anode.
• Even if the zinc layer is scratched and the iron is exposed, the zinc will still oxidise first, protecting the iron. The electrons lost by zinc prevent the iron from losing electrons (being oxidised).
This is an application of sacrificial protection, a form of cathodic protection.
Copper(II) sulfate and sodium hydroxide react to form the insoluble solid copper(II) hydroxide.
Crystals of copper(II) sulfate and sodium hydroxide are placed at opposite ends of a container of water.
Solid copper(II) hydroxide forms after several minutes.
The diagram shows the container at the start and after several minutes.
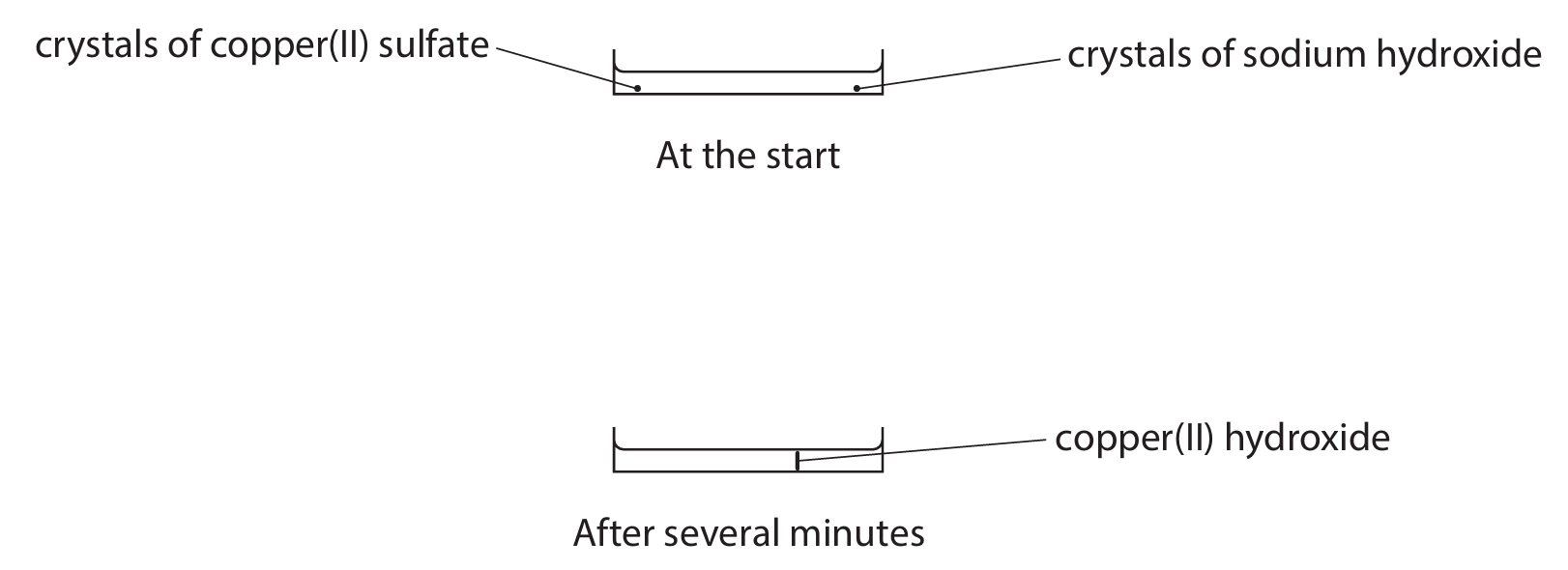
(a) Name the two processes that must occur before the copper(II) hydroxide forms.
(b) The experiment is repeated with the water at a higher temperature.
Explain, in terms of particles, why the copper(II) hydroxide takes less time to form.
(c) The formula of copper(II) hydroxide is \(\text{Cu(OH)}_2\).
(i) Give the number of different elements in copper(II) hydroxide.
(ii) Give the charge on the copper ion in \(\text{Cu(OH)}_2\).
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 3(b): Rates of reaction — part (b)
• 1(e): Chemical formulae, equations and calculations — part (c)(i)
• 1(f): Ionic bonding — part (c)(ii)
▶️ Answer/Explanation
(a)
M1: dissolving / dissolves / dissolution
M2: diffusion / diffuses
Note: The order of the two answers can be either way round.
Details: The solid crystals must first dissolve in the water to form mobile ions (\(\text{Cu}^{2+}, \text{SO}_4^{2-}, \text{Na}^+, \text{OH}^-\)). These ions then diffuse through the water from areas of high concentration (near the crystals) to areas of lower concentration. When \(\text{Cu}^{2+}\) and \(\text{OH}^-\) ions meet, they react to form the insoluble precipitate \(\text{Cu(OH)}_2\).
(b)
M1: particles move faster / particles have more kinetic energy
M2: so particles collide and react after a shorter period of time / diffuse faster / more frequent collisions
Details: At a higher temperature, the particles (ions) gain more kinetic energy. This causes them to move faster. As a result, the dissolved ions diffuse through the solution more rapidly, leading to more frequent and more energetic collisions between \(\text{Cu}^{2+}\) and \(\text{OH}^-\) ions per unit time. This increases the rate of the reaction, so the solid \(\text{Cu(OH)}_2\) forms in less time.
(c)(i)
3 / three
Details: The formula \(\text{Cu(OH)}_2\) contains copper (\(\text{Cu}\)), oxygen (\(\text{O}\)), and hydrogen (\(\text{H}\)). These are three different chemical elements.
(c)(ii)
2+ / \(\text{Cu}^{2+}\)
Allow: +2 / \(\text{Cu}^{+2}\)
Details: In ionic compounds, the sum of the charges must be zero. The hydroxide ion has a charge of \(1-\) (\(\text{OH}^-\)). Since there are two hydroxide ions, their total charge is \(2-\). To balance this, the copper ion must have a charge of \(2+\).
This question is about atoms and ions.
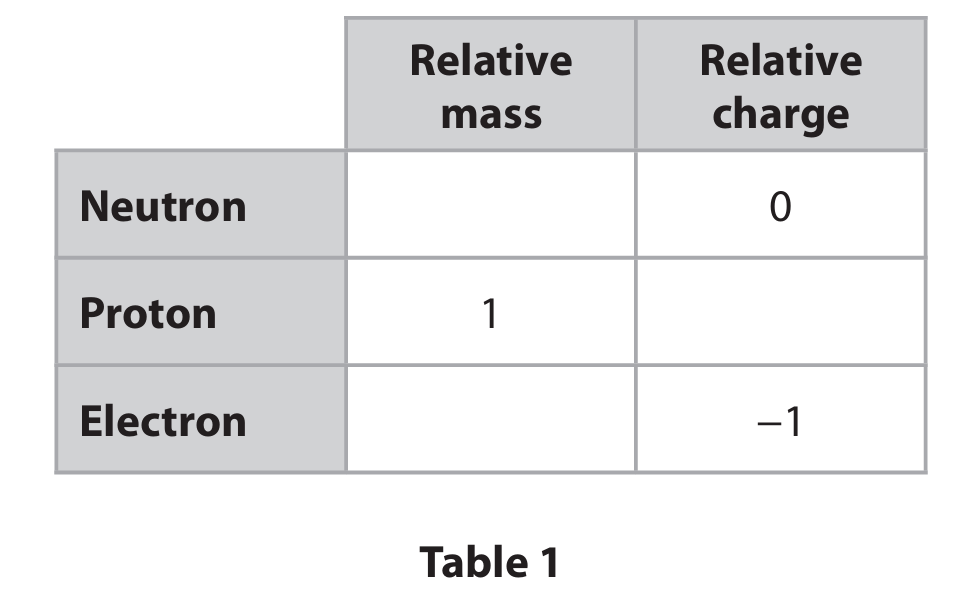
(a) Table 1 shows some information about sub-atomic particles.
Complete Table 1 by giving the missing information.
(b) Table 2 shows the numbers of protons, neutrons and electrons in species A, B, C, D, E and F.
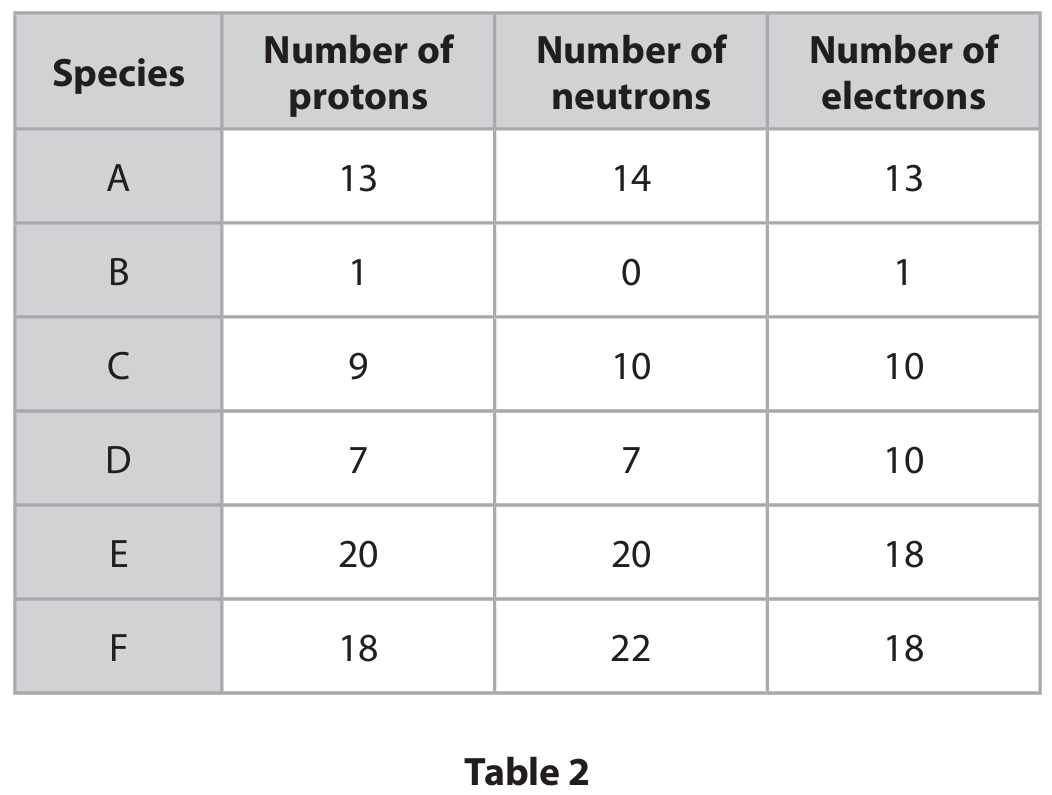
Use the information in Table 2 to answer these questions.
Each species may be used once, more than once or not at all.
(b)(i) Give the letter of the species that has 3 electrons in its outer shell.
(b)(ii) Give the letter of the species that is an ion with a 3- charge.
(b)(iii) Give the letter of the species with a mass number of 19.
(c) A sample of lithium contains two isotopes, \( ^{6}\text{Li} \) and \( ^{7}\text{Li} \).
In terms of sub-atomic particles, give one similarity and one difference between these isotopes.
- similarity:
- difference:
(d) A sample of magnesium contains 79.0% of \( ^{24}\text{Mg} \), 10.0% of \( ^{25}\text{Mg} \) and 11.0% of \( ^{26}\text{Mg} \).
Calculate the relative atomic mass, \( A_r \), of this sample of magnesium.
Give your answer to one decimal place.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(c): Isotopes — part (c)
• 1(e): Chemical formulae, equations and calculations — part (d) calculation of \(A_r\)
• 1(f): Ionic bonding — part (b)(ii) ion identification
▶️ Answer/Explanation
(a)
Completed Table:
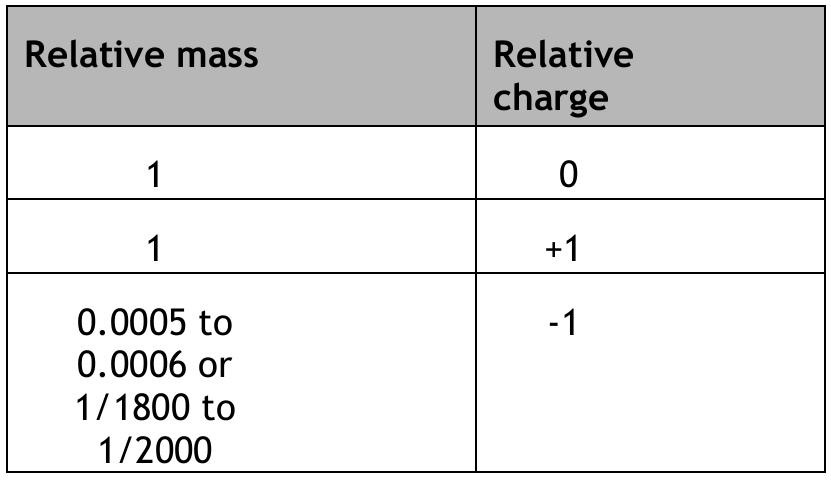
Explanation: The relative mass of a neutron and proton is 1. The relative charge of a proton is +1. The relative mass of an electron is approximately \( \frac{1}{1836} \) (or 0.0005).
(b)(i) A
Explanation: Species A (Aluminium atom) has 13 electrons. Its electron configuration is 2,8,3, so it has 3 electrons in its outer shell.
(b)(ii) D
Explanation: Species D has 7 protons (Nitrogen) but 10 electrons, meaning it has gained 3 electrons, giving it a 3- charge (\( \text{N}^{3-} \)).
(b)(iii) C
Explanation: Species C has 9 protons and 10 neutrons. Mass number = protons + neutrons = 9 + 10 = 19.
(c)
Similarity: same number of protons / same atomic number / same electron configuration / 3 protons
Difference: different number of neutrons / \( ^{6}\text{Li} \) has 3 neutrons, \( ^{7}\text{Li} \) has 4 neutrons
Note: Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
(d)
Calculation:
\( A_r = \frac{(79.0 \times 24) + (10.0 \times 25) + (11.0 \times 26)}{100} \)
\( A_r = \frac{1896 + 250 + 286}{100} \)
\( A_r = \frac{2432}{100} = 24.32 \)
Rounded to one decimal place: \( \mathbf{24.3} \)
Explanation: The relative atomic mass is the weighted average of the isotopic masses, based on their percentage abundances.
This question is about elements in Group 7 of the Periodic Table.
(a) Complete the table by giving the missing information.
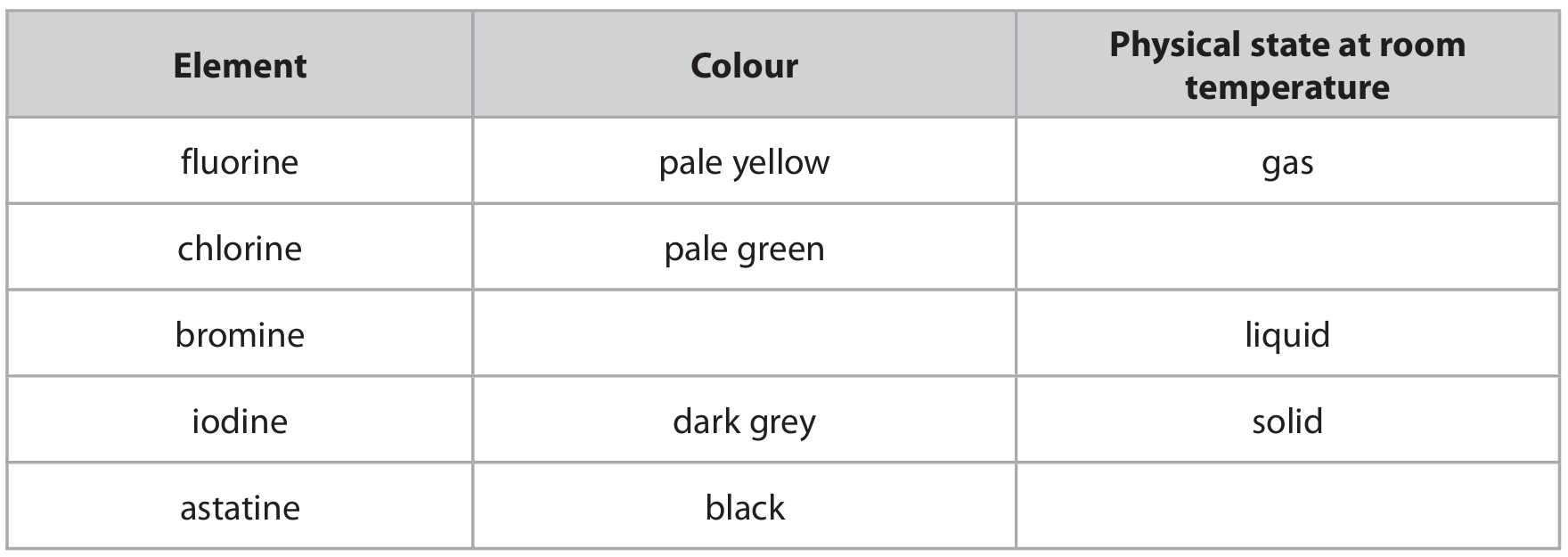
(b) The order of reactivity of the Group 7 elements can be shown by using displacement reactions.
(i) When chlorine is added to sodium bromide solution, chlorine displaces bromine. Give a chemical equation for this reaction.
(ii) Explain whether a reaction takes place when bromine water is added to sodium chloride solution.
(iii) The displacement reaction between potassium astatide and bromine can be represented by this ionic equation:
\[ 2At^- + Br_2 \rightarrow At_2 + 2Br^- \]
Explain why this is described as a redox reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(d) The Periodic Table — relating properties to group trends
• 2(d) Reactivity series — part (b)(iii) definitions of oxidation, reduction, and redox in terms of electron transfer
▶️ Answer/Explanation
(a) Completed Table
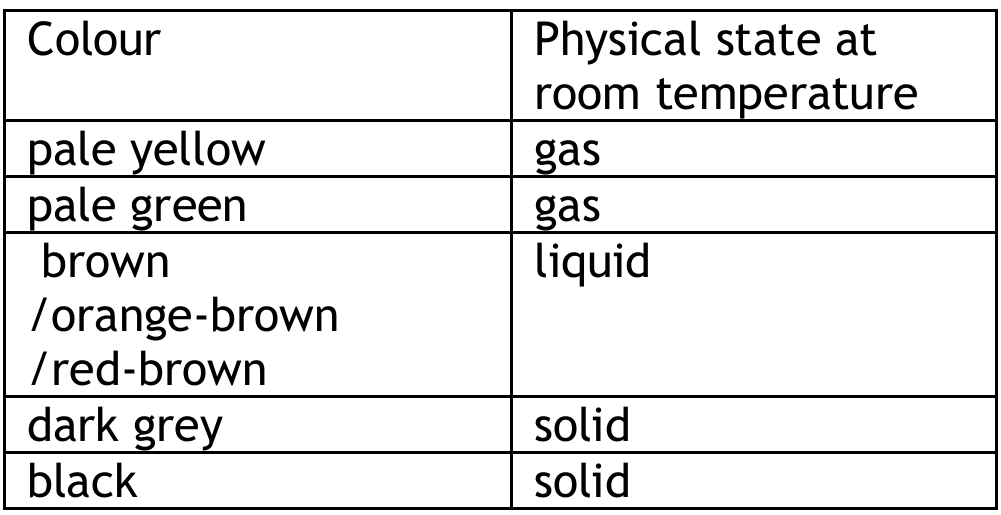
Explanation: The properties of halogens show trends down the group. As the atomic number increases, the elements become darker in colour and their physical state at room temperature changes from gas (F\(_2\), Cl\(_2\)) to liquid (Br\(_2\)) to solid (I\(_2\), At). Astatine, being the last naturally occurring halogen, is predicted to be a dark-colored solid.
(b)(i) The balanced chemical equation is:
\[ Cl_2 + 2NaBr \rightarrow Br_2 + 2NaCl \] Explanation: Chlorine is more reactive than bromine. It displaces bromide ions from solution, oxidizing them to bromine molecules (Br\(_2\)), while itself being reduced to chloride ions (Cl\(^-\)).
(b)(ii) No reaction occurs.
Explanation: Bromine is less reactive than chlorine. Since chlorine is above bromine in Group 7, it is a stronger oxidizing agent. Bromine cannot oxidize chloride ions (Cl\(^-\)) to chlorine (Cl\(_2\)) because chloride ions are more stable (harder to oxidize) than bromide ions. Therefore, adding bromine water to sodium chloride solution results in no observable change.
(b)(iii) This is a redox reaction because:
- Oxidation occurs: Astatide ions (At\(^-\)) lose electrons (their oxidation state increases from -1 to 0) to form astatine molecules (At\(_2\)). At\(^-\) is the reducing agent.
- Reduction occurs: Bromine molecules (Br\(_2\)) gain electrons (their oxidation state decreases from 0 to -1) to form bromide ions (Br\(^-\)). Br\(_2\) is the oxidizing agent.
Since there is simultaneous electron loss (oxidation) and electron gain (reduction), the reaction is classified as a redox reaction. This also demonstrates the reactivity trend: bromine is higher in the group than astatine and is therefore a stronger oxidizing agent, capable of oxidizing astatide ions.
A student uses the reaction between magnesium and dilute hydrochloric acid to prepare crystals of hydrated magnesium chloride.
The student adds excess magnesium powder to hydrochloric acid. This is the equation for the reaction.
\[ \text{Mg(s) + 2HCl(aq) → MgCl}_2\text{(aq) + H}_2\text{(g)} \]
(a) How does hydrochloric acid behave when acting as an acid?
(b) State one observation made during this reaction.
(c) Give a reason why the student adds an excess of magnesium to the dilute hydrochloric acid.
(d) Describe what the student should do to the mixture formed in (c) to produce pure, dry crystals of hydrated magnesium chloride.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(e) Chemical formulae, equations and calculations: part (a) – acid behaviour (proton donor)
• 2(f) Acids, alkalis and titrations: part (a) – acid definition
• 2(d) Reactivity series: part (b) – reaction observation
• Specification point 2.39/2.40C: part (d) – preparation of a pure, dry soluble salt
▶️ Answer/Explanation
(a) D (as a proton donor)
Explanation: According to the Brønsted–Lowry theory (specification 2.35/2.36), an acid is a proton (\( \text{H}^+ \)) donor. Hydrochloric acid (\( \text{HCl} \)) donates a proton in aqueous solution to form \( \text{H}_3\text{O}^+ \) and \( \text{Cl}^- \).
- A is not correct because acids are not electron acceptors (that describes Lewis acids, not required here).
- B is not correct because acids are not electron donors.
- C is not correct because a proton acceptor is a base, not an acid.
(b) Effervescence / bubbles / fizzing / a colourless solution forms / magnesium dissolves / magnesium gets smaller / reaction gets warm.
Explanation: The reaction produces hydrogen gas (\( \text{H}_2 \)), which is seen as effervescence. Magnesium metal dissolves as it reacts, and the reaction is exothermic, so a temperature increase may be felt.
(c) To ensure all the (hydrochloric) acid has reacted / to ensure the acid is completely neutralised / to prevent hydrochloric acid remaining in the product mixture.
Explanation: Adding excess magnesium ensures that the limiting reactant is the acid, so no acid remains after the reaction. This is important because any leftover acid would contaminate the salt crystals and could prevent crystallisation or cause impurities.
(d) A description connecting any 5 of the following steps:
- Filter (the excess magnesium).
- Heat the filtrate.
- Heat until crystals first start to form (or until a saturated solution forms).
- Allow to cool and crystallise.
- Pour off (decant) the excess liquid (mother liquor).
- Leave in a warm place to dry (or dry with a paper towel / in a warm oven / in a desiccator).
Notes:
• “Heat to evaporate some of the water” is acceptable.
• If the sample is heated to dryness, only the first two marks are awarded, as this would produce anhydrous or impure crystals.
• Washing the crystals is not required here (and might redissolve them).
Specification link: This practical procedure relates to specification points 2.39 and 2.40C (preparing a pure, dry sample of a soluble salt).
A student reacts small chips of calcium carbonate with an excess of dilute hydrochloric acid. This is the student’s method.
- Add 5.00 g of small calcium carbonate chips to a conical flask.
- Add 30 cm³ of dilute hydrochloric acid which is in excess.
- Quickly add a cotton wool plug and place the conical flask on a balance.
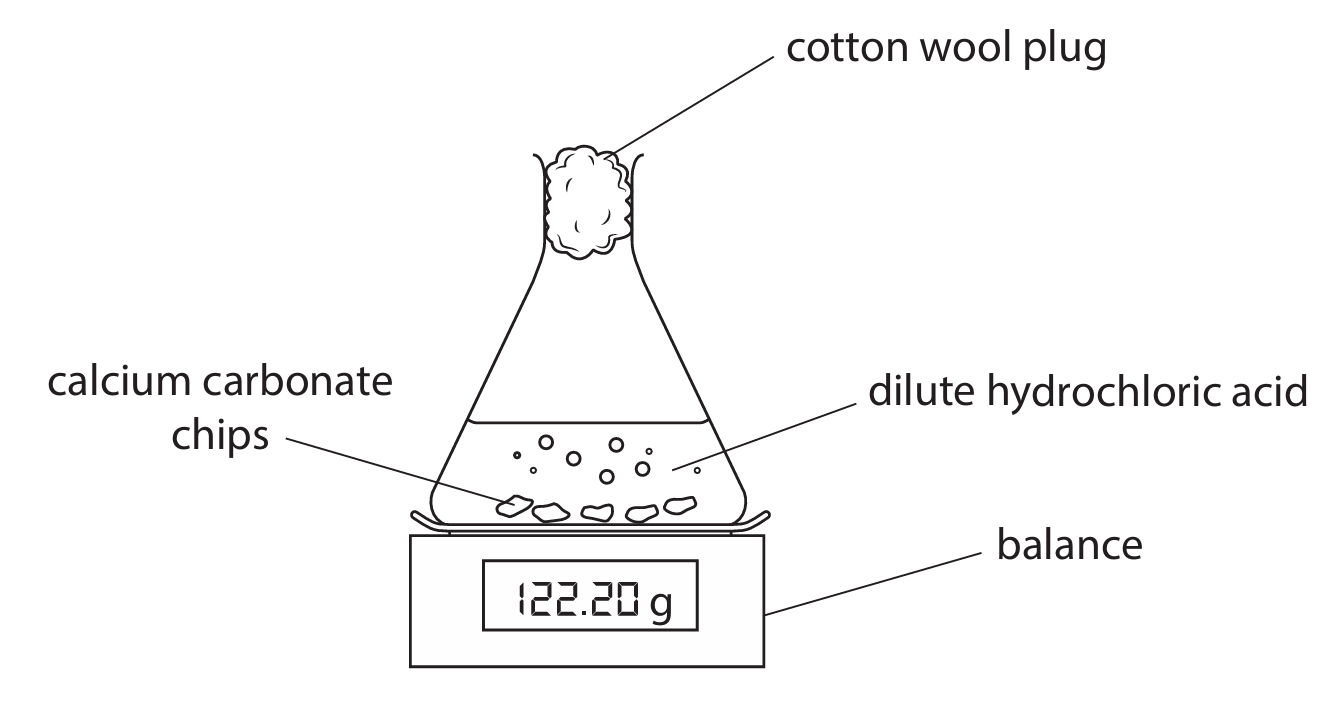
This is the equation for the reaction:
\(\text{CaCO}_3(s) + 2\text{HCl}(aq) \rightarrow \text{CaCl}_2(aq) + \text{H}_2\text{O}(l) + \text{CO}_2(g)\)
The reading on the balance decreases as carbon dioxide gas is lost through the cotton wool plug.
(a) Calculate the mass reading on the balance when all of the calcium carbonate has reacted. Assume that all of the carbon dioxide gas is lost through the cotton wool plug.
[For \(\text{CaCO}_3\), \(M_r = 100\); for \(\text{CO}_2\), \(M_r = 44\)]
(b) The student repeats the experiment with a different mass of calcium carbonate and a different volume of dilute hydrochloric acid. The hydrochloric acid is in excess. The student records the total mass of carbon dioxide produced at 1-minute intervals for 8 minutes. The table shows the student’s results.
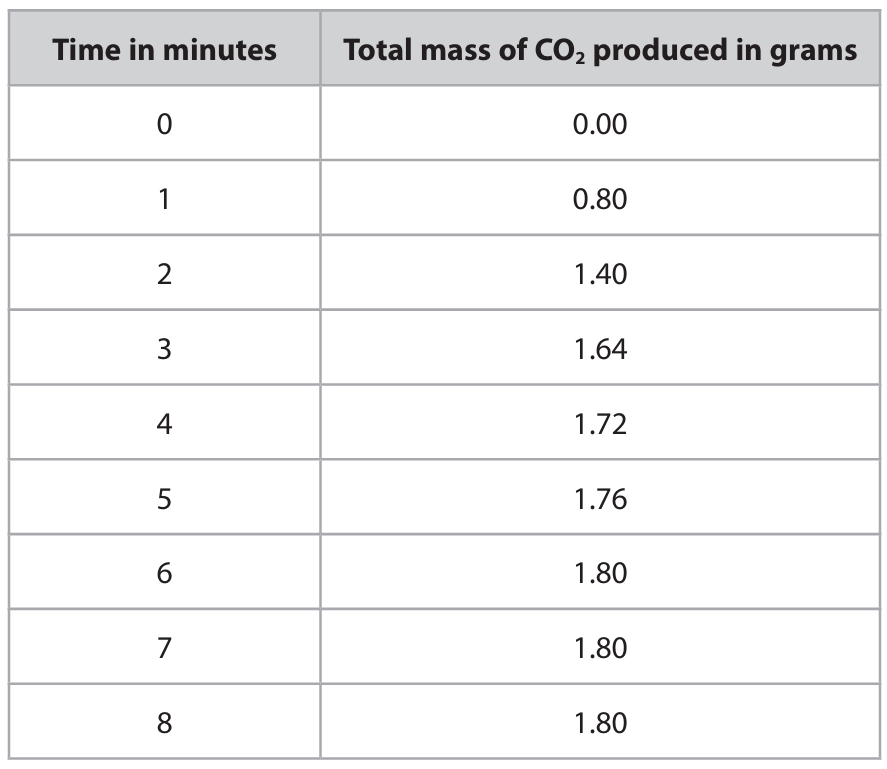
(b)(i) Plot the student’s results. The first three have been plotted for you.
(b)(ii) Draw a curve of best fit through the points.
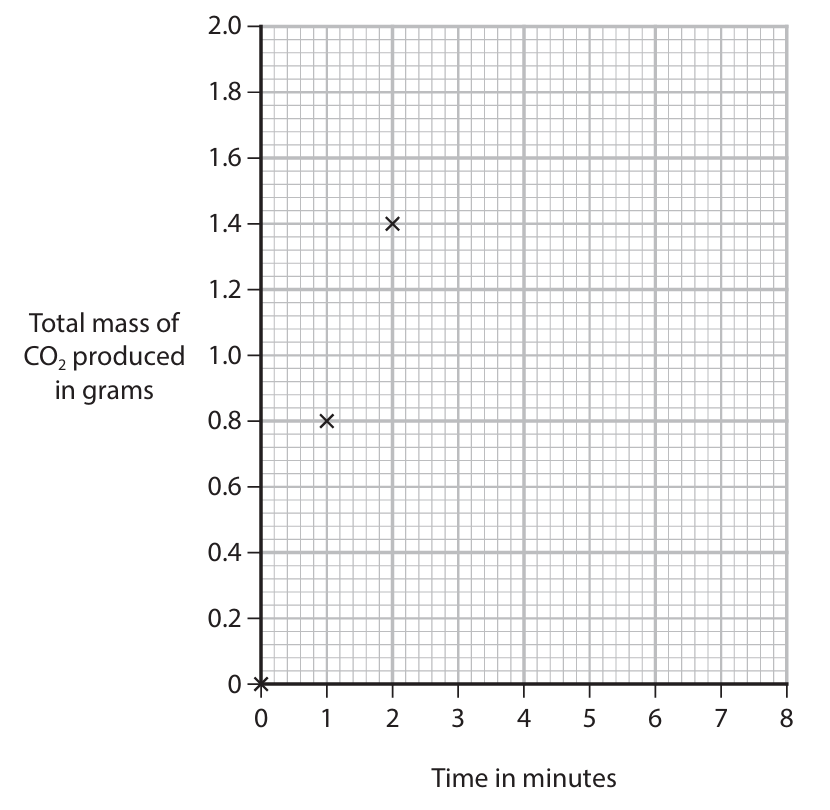
(b)(iii) Calculate the rate of reaction at 2 minutes by drawing a tangent to the curve and calculating the gradient of the tangent. Give the units for the rate of reaction.
(b)(iv) Explain why the shape of the graph changes during the 8 minutes.
(c) The student repeats the experiment using the same mass of calcium carbonate but as powder rather than chips, keeping all other conditions the same. Explain the effect of this change on the rate of reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 3(b): Rates of reaction — parts (b)(iii), (b)(iv), (c)
• Appendix 4 (Mathematical Skills): Graphs — part (b)(iii) tangent/gradient
▶️ Answer/Explanation
(a) Mass reading on the balance
- Moles of \(\text{CaCO}_3 = \frac{5.00}{100} = 0.0500 \text{ mol}\)
- From the equation, 1 mol \(\text{CaCO}_3\) produces 1 mol \(\text{CO}_2\).
- Mass of \(\text{CO}_2\) lost = \(0.0500 \times 44 = 2.20 \text{ g}\).
- Initial total mass = mass of flask + contents. The balance reading decreases by the mass of \(\text{CO}_2\) lost. The final reading = initial reading \( – 2.20 \text{ g}\).
- If the initial reading was, for example, 122.20 g (as implied in the diagram), the final reading would be \(122.20 – 2.20 = 120.00 \text{ g}\).
Answer: 120.00 g.
(b)(i) & (ii) Graph
Plot the points from the table (Time on x-axis, Mass of CO₂ on y-axis) and draw a smooth curve of best fit that starts steep, becomes less steep, and eventually levels off at 1.80 g.
(b)(iii) Rate at 2 minutes
- Method: Draw a tangent to the curve at \(t = 2\) minutes. The gradient of this tangent = \(\frac{\Delta y}{\Delta x} = \frac{\text{change in mass of CO}_2 (\text{g})}{\text{change in time (min)}}\).
- Example Calculation (using typical tangent values): If the tangent passes through (1.0 min, 0.60 g) and (3.0 min, 1.70 g), then:
Gradient = \(\frac{1.70 – 0.60}{3.0 – 1.0} = \frac{1.10}{2.0} = 0.55\) - Units: The rate = gradient = \(0.55 \text{ g min}^{-1}\) (or g/min).
Answer: ~0.55 g min⁻¹ (value depends on tangent drawn).
(b)(iv) Explanation of graph shape
The graph is steepest at the start because the reaction rate is fastest initially. This is due to the highest concentration of HCl. As the reaction proceeds, the concentration of HCl decreases (it is being used up), and the amount of CaCO₃ surface area available also decreases. This reduces the frequency of successful collisions, so the rate slows (curve becomes less steep). After about 6 minutes, the curve levels off (plateaus) because the reaction has finished—all the calcium carbonate has been used up. No more CO₂ is produced, so the total mass of CO₂ becomes constant.
(c) Effect of using powder
The rate of reaction increases significantly. Using powder instead of chips greatly increases the surface area of the calcium carbonate exposed to the acid. A larger surface area provides more sites for collisions between HCl particles and CaCO₃ particles. This leads to more frequent successful collisions per unit time, increasing the rate. The initial steep part of the mass-time graph would be even steeper, and the reaction would finish more quickly (the plateau would be reached sooner).
Key points: Increased surface area → more collisions per second → faster rate.
This question is about the covalent compounds hydrogen chloride and silicon dioxide.
(a) Describe the forces of attraction in a covalent bond.
(b) Hydrogen chloride has the formula HCl
(i) Draw a dot-and-cross diagram to show the bonding in a hydrogen chloride molecule. Show only the outer shell electrons in each atom.
(ii) Explain why the boiling point of hydrogen chloride is low.
(c) The diagram shows part of the structure of silicon dioxide.
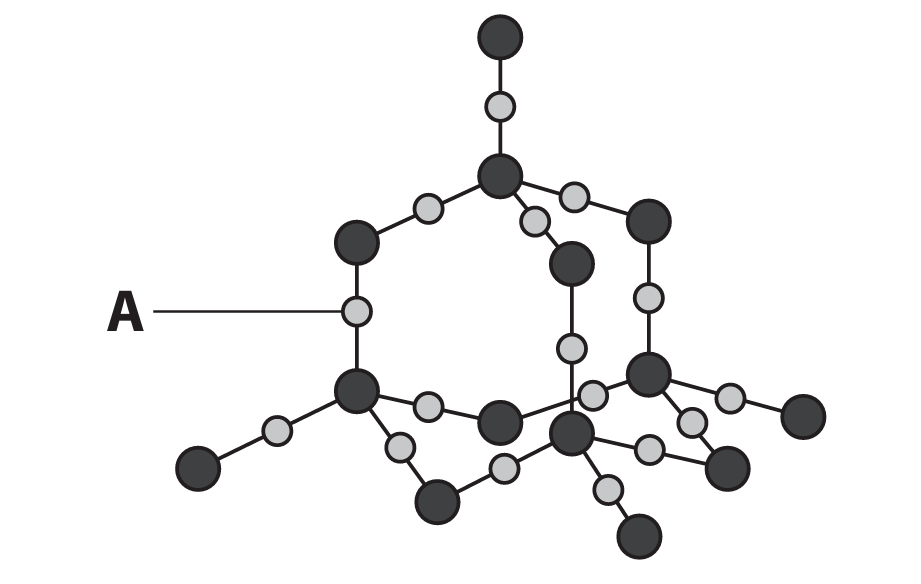
(i) Give a reason why particle A represents an oxygen atom.
(ii) This is the equation for the reaction between silicon dioxide and calcium oxide.
\[ \text{SiO}_2 + \text{CaO} \rightarrow \text{CaSiO}_3 \]
Give a reason why silicon dioxide reacts with calcium oxide.
(iii) Silicon dioxide has the same structure as diamond. Silicon dioxide is very hard and has a very high melting point.
Explain these properties. Refer to the structure and bonding of silicon dioxide in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(g): Simple molecular structures — part (b)(ii)
• 1(g): Giant covalent structures — part (c)(iii)
• 1(f): Ionic bonding — part (c)(ii) context
• 2(g): Acids, bases and salt preparations — part (c)(ii) acid-base reaction
▶️ Answer/Explanation
(a) Any correct description of the electrostatic forces in a covalent bond:
• Attraction between (positively charged) nuclei and the shared pair(s) of electrons. OR
• Attraction between the shared pair(s) of electrons and (positively charged) nuclei.
(Mark scheme: 1 mark for each correct component of the description; “nuclei” must be plural).
(b)(i) A dot-and-cross diagram showing:
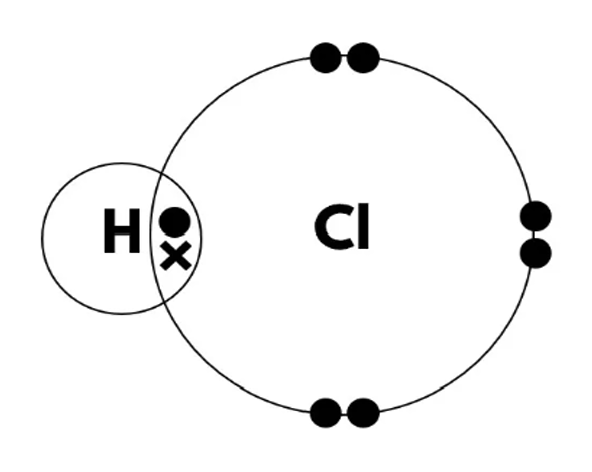
• Hydrogen atom with one electron (dot or cross).
• Chlorine atom with seven outer shell electrons (dots or crosses, using the opposite symbol to hydrogen).
• One shared pair of electrons between them, completing chlorine’s outer shell (8 electrons) and hydrogen’s (2 electrons).
Example: H with one dot, Cl with 7 crosses, one shared pair (dot and cross overlapping).
(b)(ii) Hydrogen chloride has a simple molecular structure. The molecules are held together by weak intermolecular forces. These forces require only a small amount of energy to overcome during boiling, hence the low boiling point.
(Mark scheme: M1 for “weak intermolecular forces”, M2 for “require little energy to overcome”).
(c)(i) Any one of the following:
• Oxygen is a smaller atom than silicon.
• Oxygen has two electron shells, silicon has three.
• Each silicon atom is bonded to four oxygen atoms.
• Each oxygen atom is bonded to two silicon atoms.
(The diagram shows oxygen atoms bridging between silicon atoms).
(c)(ii) Silicon dioxide (\(\text{SiO}_2\)) is an acidic oxide and calcium oxide (\(\text{CaO}\)) is a basic oxide. They react in an acid-base (neutralisation) reaction to form the salt calcium silicate (\(\text{CaSiO}_3\)).
Do not credit “calcium oxide is an alkali” – alkalis are soluble bases.
(c)(iii) An explanation linking structure to properties:
• Silicon dioxide has a giant covalent (macromolecular) structure/lattice. (M1)
• It has a three-dimensional tetrahedral structure where each silicon atom is covalently bonded to four oxygen atoms. (M4, M5)
• There are strong covalent bonds throughout the entire structure. (M2)
• A very large amount of energy is required to break these strong covalent bonds to melt or break the structure. (M3)
• The atoms are fixed in a rigid, non-layered network, so they cannot slide over each other, making it very hard. (M6)
(Mark scheme: 5 marks for any five valid points. Do not credit mention of intermolecular forces or ionic bonding).
This question is about products from crude oil.
(a) Most of the compounds in crude oil are from the same homologous series. Give the name of this homologous series.
(b) Many fuels are obtained from the fractional distillation of crude oil. Explain how the combustion of a common impurity in fuels causes acid rain.
(c) The equation shows the cracking of a hydrocarbon produced from the fractional distillation of crude oil.
\[ C_{10}H_{22} \rightarrow C_8H_{18} + C_2H_4 \]
(i) Give the temperature and the catalyst used in catalytic cracking.
(ii) Explain why cracking is a useful process in the oil industry.
(d) The formula \( C_5H_{12} \) represents three isomers.
(i) Explain what is meant by the term isomers.
(ii) This is the displayed formula of one of the isomers.
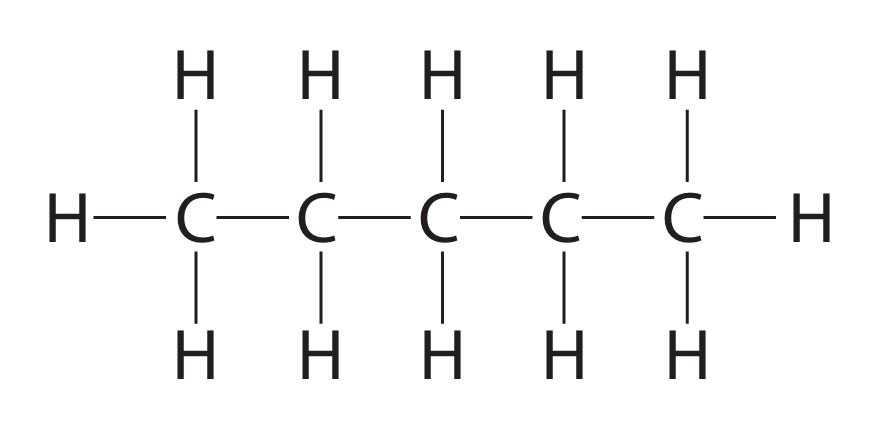
Draw the displayed formula of each of the other two isomers.

Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(b) & 4(c): Crude oil and Alkanes — part (b)
• 4(b): Catalytic Cracking — parts (c)(i), (c)(ii)
• 4(a): Introduction to Organic Chemistry — parts (d)(i), (d)(ii)
• 4(c): Alkanes — part (d)(ii)
▶️ Answer/Explanation
(a) alkanes
Crude oil is predominantly a mixture of hydrocarbons from the alkane homologous series.
(b) A three-mark explanation:
1. (The impurity) sulfur burns/combusts to form sulfur dioxide (\(SO_2\)).
2. Sulfur dioxide dissolves in/reacts with (rain)water.
3. This forms sulfuric acid (\(H_2SO_4\)), causing acid rain.
Nitrogen oxides (from high-temperature combustion) can also cause acid rain, but the question specifies the “common impurity in fuels,” which is sulfur.
(c)(i)
• Temperature: 600–700 °C
• Catalyst: alumina / silica / aluminosilicates / zeolites / \(Al_2O_3\) / \(SiO_2\)
(c)(ii) A three-mark explanation:
1. Long-chain alkanes are less useful/have lower demand.
2. Cracking produces shorter alkanes (e.g., \(C_8H_{18}\)) that are more useful as fuels (e.g., petrol/gasoline).
3. Cracking also produces alkenes (e.g., \(C_2H_4\)) which are used to make polymers/plastics and other chemicals (like alcohols).
This process balances supply (lots of long chains from distillation) with demand (more need for short-chain fuels and feedstocks).
(d)(i) A two-mark explanation:
1. Isomers are molecules with the same molecular formula.
2. But they have different structural/displayed formulae (different arrangements of atoms).
(d)(ii) The other two isomers of \(C_5H_{12}\) (pentane) are:
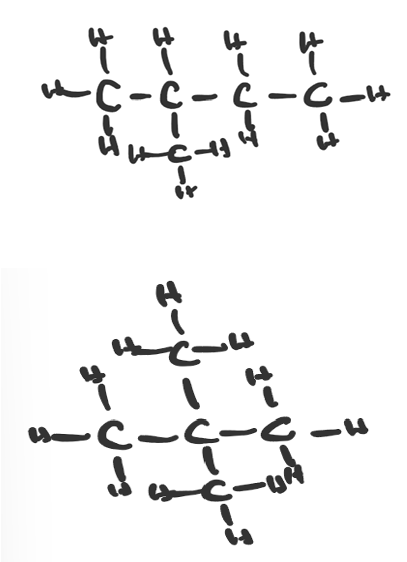
• 2-methylbutane (branched chain with a methyl (\(-CH_3\)) group on the second carbon).
• 2,2-dimethylpropane (neopentane, a central carbon bonded to three methyl groups and one other carbon/hydrogen).
Pentane itself (\(CH_3-CH_2-CH_2-CH_2-CH_3\)) is the straight-chain isomer shown in the question.
A student investigates the reaction between zinc and copper(II) sulfate solution.
This is the equation for the reaction:
\[ \text{Zn(s)} + \text{CuSO}_4(\text{aq}) \rightarrow \text{ZnSO}_4(\text{aq}) + \text{Cu(s)} \]
This is the student’s method:
- Step 1: add 25.0 cm³ of copper(II) sulfate solution to a glass beaker
- Step 2: record the temperature of the solution
- Step 3: add between 5 g and 6 g of zinc powder (an excess) and stir the mixture
- Step 4: record the highest temperature reached
(a) (i) Explain an improvement to step 1 to produce a more accurate temperature rise.
(a) (ii) Give a reason why the student does not need to know the exact mass of zinc powder added in step 3.
(a) (iii) State the colour change in the solution.
(b) (i) The table shows the student’s results.

Calculate the heat energy change (\(Q\)) in joules (\(J\)).
[for the solution, \(c = 4.2 \, \text{J} / \text{g} / ^\circ \text{C}\)]
[mass of 1.0 cm³ of solution = 1.0 g] (3)
(b) (ii) The student repeats the experiment and calculates \(Q\) as 3800 J. The solution used contains 6.38 g of copper(II) sulfate.
Calculate the molar enthalpy change, \(\Delta H\), in kJ/mol. Include a sign in your answer.
[for \(\text{CuSO}_4\), \(M_r = 159.5\)] (4)
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(d): Reactivity series — context of the zinc-copper displacement reaction
• 1(e): Chemical formulae, equations and calculations — mole and enthalpy calculations
▶️ Answer/Explanation
(a) (i) Any two of the following:
- M1: Use a polystyrene cup (instead of a glass beaker).
- M2: This provides better insulation / reduces heat loss to the surroundings.
- M3: Use a lid on the container / leave the mixture to reach a steady temperature before recording it.
Improving the insulation minimizes energy exchange with the environment, leading to a more accurate measurement of the temperature change solely due to the reaction.
(a) (ii) The zinc powder is in excess.
If the zinc is in excess, all of the copper(II) sulfate will react regardless of the exact mass of zinc added. Therefore, the limiting reactant and the amount of heat released depend only on the amount of copper(II) sulfate, not on the zinc.
(a) (iii) From blue to colourless.
The blue colour of the solution is due to aqueous copper(II) ions (\( \text{Cu}^{2+}(\text{aq}) \)). As these ions are displaced and form solid copper, the blue colour fades, leaving a colourless solution of zinc sulfate.
(b) (i) Calculation of heat energy change \(Q\):
- Mass of solution, \(m = 25.0 \, \text{cm}^3 \times 1.0 \, \text{g cm}^{-3} = 25.0 \, \text{g}\)
- Temperature change, \(\Delta T = 48.6^\circ \text{C} – 18.5^\circ \text{C} = 30.1^\circ \text{C}\)
- Specific heat capacity, \( c = 4.2 \,\mathrm{J\,g^{-1}\,^{\circ}C^{-1}} \)
- Using \(Q = mc\Delta T\):
\[ Q = 25.0 \times 4.2 \times 30.1 \] \[ Q = 3160.5 \, \text{J} \]
Answer: \(3160.5 \, \text{J}\) (Accept \(3160 \, \text{J}\) or \(3161 \, \text{J}\) with appropriate significant figures).
(b) (ii) Calculation of molar enthalpy change \(\Delta H\):
- Calculate moles of \(\text{CuSO}_4\): \[ \text{moles} = \frac{\text{mass}}{M_r} = \frac{6.38}{159.5} = 0.0400 \, \text{mol} \]
- Energy change per mole from experiment: \[ \frac{Q}{\text{moles}} = \frac{3800 \, \text{J}}{0.0400 \, \text{mol}} = 95000 \, \text{J/mol} \]
- Convert to kJ/mol: \[ \frac{95000 \, \text{J/mol}}{1000} = 95 \, \text{kJ/mol} \]
- The reaction is exothermic (temperature increased), so \(\Delta H\) is negative. \[ \Delta H = -95 \, \text{kJ/mol} \]
Answer: \(-95 \, \text{kJ/mol}\)
The negative sign is crucial as it indicates the reaction releases heat (exothermic). The calculation follows the formula: \(\Delta H = \frac{-Q}{\text{moles of limiting reactant}}\).
This question is about sodium sulfate, \(\text{Na}_2\text{SO}_4\).
(a) A teacher finds an unlabelled bottle containing a white solid. Describe tests to show that the white solid is sodium sulfate.
(b) A student has a sample of crystals of hydrated sodium sulfate, \(\text{Na}_2\text{SO}_4 \cdot x\text{H}_2\text{O}\). The student uses this apparatus to find the value of \(x\) in \(\text{Na}_2\text{SO}_4 \cdot x\text{H}_2\text{O}\).
This is the student’s method:
- find the mass of the empty crucible
- add some hydrated sodium sulfate crystals to the crucible
- find the total mass of the crucible and hydrated sodium sulfate crystals
- heat the crucible to remove water from the crystals
- find the mass of the crucible and its contents every 2 minutes for 10 minutes
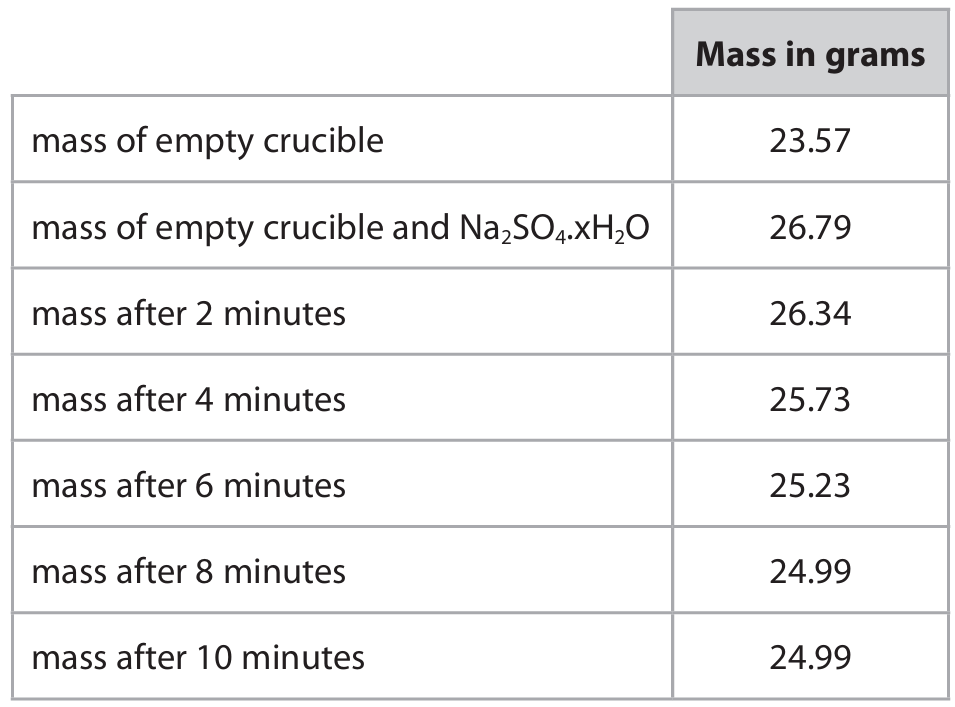
The table shows the student’s results.
(b)(i) State how the results show that all the water has been removed from the crystals.
(b)(ii) Calculate the mass of anhydrous sodium sulfate remaining after heating.
(b)(iii) Calculate the mass of water lost from the sodium sulfate crystals during heating.
(b)(iv) Use the student’s results to calculate \(x\) in the formula \(\text{Na}_2\text{SO}_4 \cdot x\text{H}_2\text{O}\).
\([ \text{For } \text{Na}_2\text{SO}_4, M_r = 142 \quad \text{for } \text{H}_2\text{O}, M_r = 18 ]\)
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(e): Chemical formulae, equations and calculations — parts (b)(ii), (b)(iii), (b)(iv)
• 1(e): Determination of formulae from experimental data (water of crystallisation) — part (b)(iv)
• 2(g): Acids, bases and salt preparations — context of hydrated salts
▶️ Answer/Explanation
(a) Tests to identify the white solid as sodium sulfate:
To identify sodium ions:
• Perform a flame test on a small sample of the solid.
• Observation: A persistent yellow/orange flame confirms the presence of \(\text{Na}^+\).
To identify sulfate ions:
• Dissolve a sample of the solid in distilled water to make a solution.
• Add dilute hydrochloric acid (to remove carbonate or other acid-soluble ions that could interfere).
• Add barium chloride solution (or barium nitrate).
• Observation: A white precipitate (\(\text{BaSO}_4\)) confirms the presence of \(\text{SO}_4^{2-}\).
(b)(i) The results show that all water has been removed because the mass of the crucible and its contents stays constant between the 8-minute and 10-minute readings (both 24.99 g). This indicates no further mass loss, meaning no more water is being driven off.
(b)(ii) Mass of anhydrous sodium sulfate:
The final constant mass (crucible + anhydrous \(\text{Na}_2\text{SO}_4\)) = 24.99 g.
Mass of empty crucible = 23.57 g.
\(\text{Mass of anhydrous } \text{Na}_2\text{SO}_4 = 24.99 – 23.57 = 1.42 \text{ g}\).
(b)(iii) Mass of water lost:
Initial mass (crucible + hydrated salt) = 26.79 g.
Final mass (crucible + anhydrous salt) = 24.99 g.
\(\text{Mass of water lost} = 26.79 – 24.99 = 1.80 \text{ g}\).
(b)(iv) Calculation of \(x\):
• Moles of \(\text{Na}_2\text{SO}_4 = \frac{1.42}{142} = 0.0100 \text{ mol}\).
• Moles of \(\text{H}_2\text{O} = \frac{1.80}{18} = 0.100 \text{ mol}\).
• Ratio \(\text{H}_2\text{O} : \text{Na}_2\text{SO}_4 = \frac{0.100}{0.0100} = 10\).
Therefore, \(x = 10\). The formula is \(\text{Na}_2\text{SO}_4 \cdot 10\text{H}_2\text{O}\).
Note: This is the common hydrated form of sodium sulfate (Glauber’s salt). The experiment relies on the principle of thermal decomposition to remove water of crystallisation and using molar masses to find the mole ratio.
