This question is about elements, compounds and mixtures.
(a) The box gives the names of some substances.

Choose a substance from the box to answer each of these questions.
(i) Give the name of a compound.
(ii) Give the name of a mixture.
(iii) Give the name of a diatomic molecule.
(iv) Give the name of a metal element.
(b) (i) Name the technique used to separate crude oil into fractions.
(ii) Give one use of the kerosene fraction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(a): States of matter — part (a)(i), (a)(ii)
• 1(c): Atomic structure — part (a)(iii)
• 1(d): The Periodic Table — part (a)(iv)
• 4(b): Crude oil — part (b)
▶️ Answer/Explanation
(a)(i) water
(a)(ii) petrol
(a)(iii) fluorine
(a)(iv) calcium
(b)(i) fractional distillation
ALLOW: fractionating / fractionation
IGNORE: distillation alone
(b)(ii) aircraft fuel / jet fuel / (aero)plane fuel / aviation fuel / fuel for lamps / fuel for heaters / fuel for stoves
MUST mention fuels, not just aircraft etc.
Magnesium reacts with dilute hydrochloric acid to form magnesium chloride and hydrogen.
The hydrogen is collected in a gas syringe.
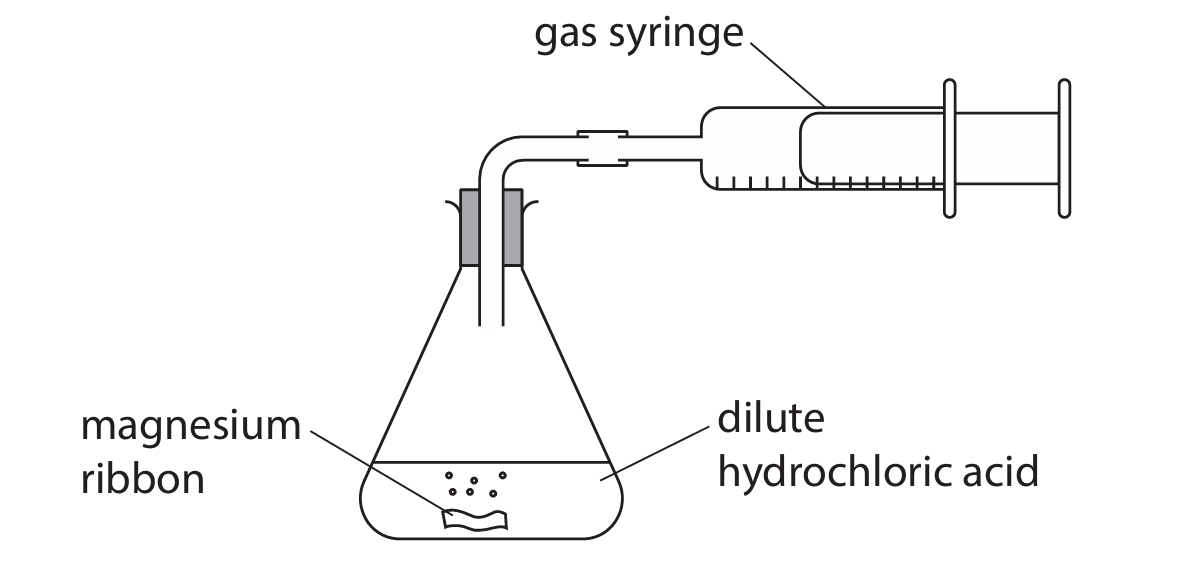
The reaction is exothermic and magnesium is in excess.
(a) State what is meant by the term exothermic.
(b) Complete the chemical equation for the reaction.
\[ \text{Mg} + 2\text{HCl} \rightarrow \_\_\_\_\_\_\_\_\_\_ + \_\_\_\_\_\_\_\_\_\_ \]
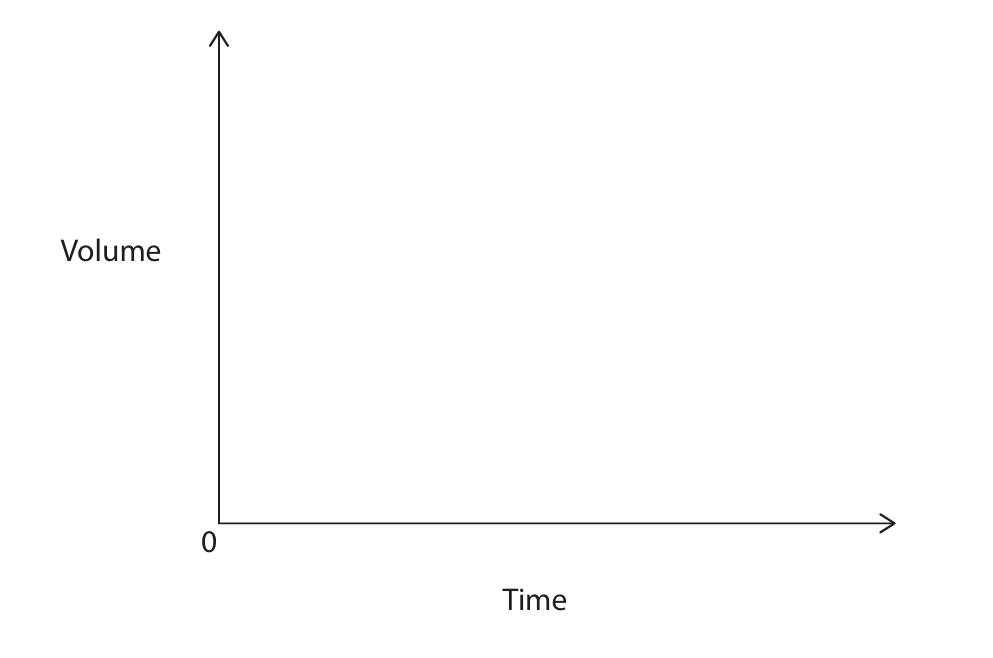
(c) Draw a sketch graph to show how the volume of hydrogen changes with time until the reaction has finished.
(d) Explain why increasing the temperature of the hydrochloric acid affects the rate of the reaction.
Refer to collision theory in your answer.
(e) Draw a reaction profile diagram for the reaction, showing \(\Delta H\) and activation energy.
Label the reactants and products.

Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(e) Chemical formulae, equations and calculations: Writing balanced equations — part (b)
• 3(b) Rates of reaction: Effect of temperature on rate, collision theory, graphing volume vs time — parts (c), (d)
• 2(d) Reactivity series: Reactions of metals with acids — context of the reaction
▶️ Answer/Explanation
(a) A reaction in which heat (energy)/thermal energy is given out/released to the surroundings.
(b) \[ \text{Mg} + 2\text{HCl} \rightarrow \text{MgCl}_2 + \text{H}_2 \]
(c) Sketch graph: Smooth curve starting at the origin, rising steeply at first, then gradually levelling off to a horizontal plateau (constant volume).
(d) • Increasing temperature increases the rate of reaction.
• Particles (molecules) gain more kinetic energy/move faster.
• This leads to more collisions per unit time/more frequent collisions.
• More particles have the (necessary) activation energy, leading to more successful collisions.
(e) Reaction profile diagram:
• Axes: Energy (y) vs Reaction progress (x).
• Reactants labelled at a higher energy level.
• Products labelled at a lower energy level (exothermic).
• An activation energy hump shown between reactants and products.
• \(E_a\) labelled from reactants to the top of the hump.
• \(\Delta H\) labelled from reactants to products (downwards arrow, negative value).
Mark scheme references from images:
(a) Heat/energy is given out/released/lost to the surroundings.
(b) Subscripts, superscripts, and case must be correct: \(\text{MgCl}_2\) and \(\text{H}_2\).
(c) M1: smooth curve starting at origin. M2: levels off horizontally.
(d) M1: rate increases. M2: more KE. M3: more collisions. M4: more successful collisions.
(e) M1: label reactants/products. M2: product line below reactant line. M3: activation hump. M4: \(E_a\) labelled. M5: \(\Delta H\) shown.
This question is about Group 2 elements and their compounds.
(a) A sample of magnesium contains three isotopes.
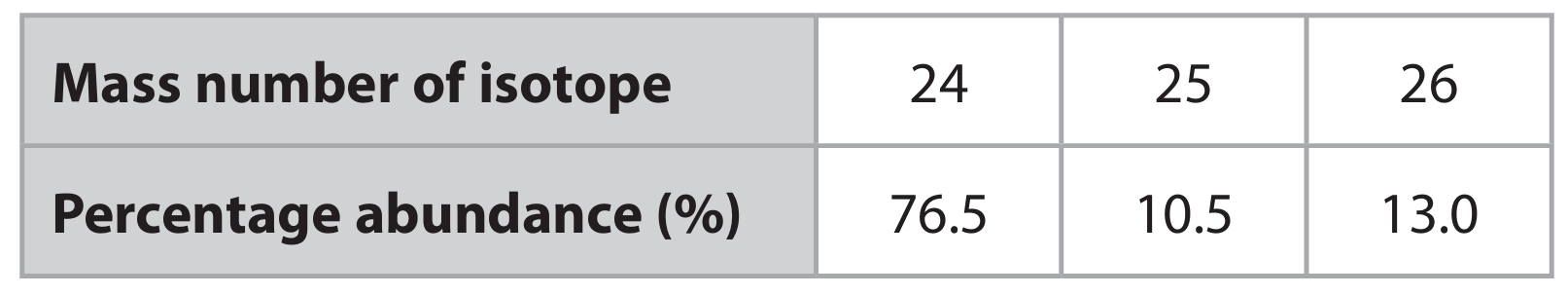
(i) State, in terms of subatomic particles, what is meant by the term isotopes.
(ii) Calculate the relative atomic mass (\(A_r\)) of magnesium. Give your answer to one decimal place.
(b) Which of these compounds is an insoluble solid?
- A) calcium nitrate
- B) calcium sulfate
- C) magnesium nitrate
- D) magnesium sulfate
(c) Magnesium oxide is an ionic compound. The diagrams show the atoms of magnesium and oxygen.
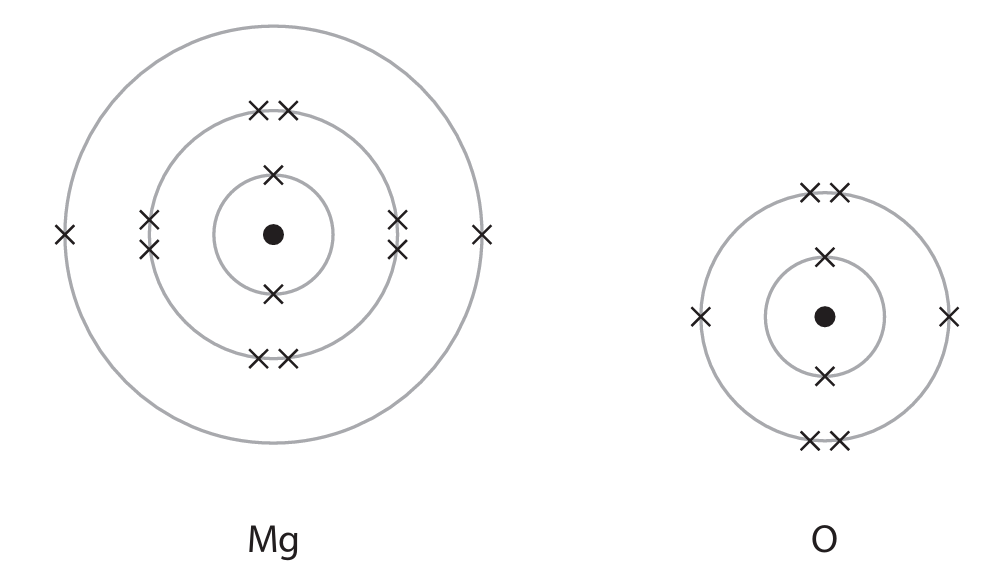
Draw diagrams to show the structure of each ion in magnesium oxide. Include the charge on each ion.
(d) The table lists some substances and shows which substance conducts electricity.
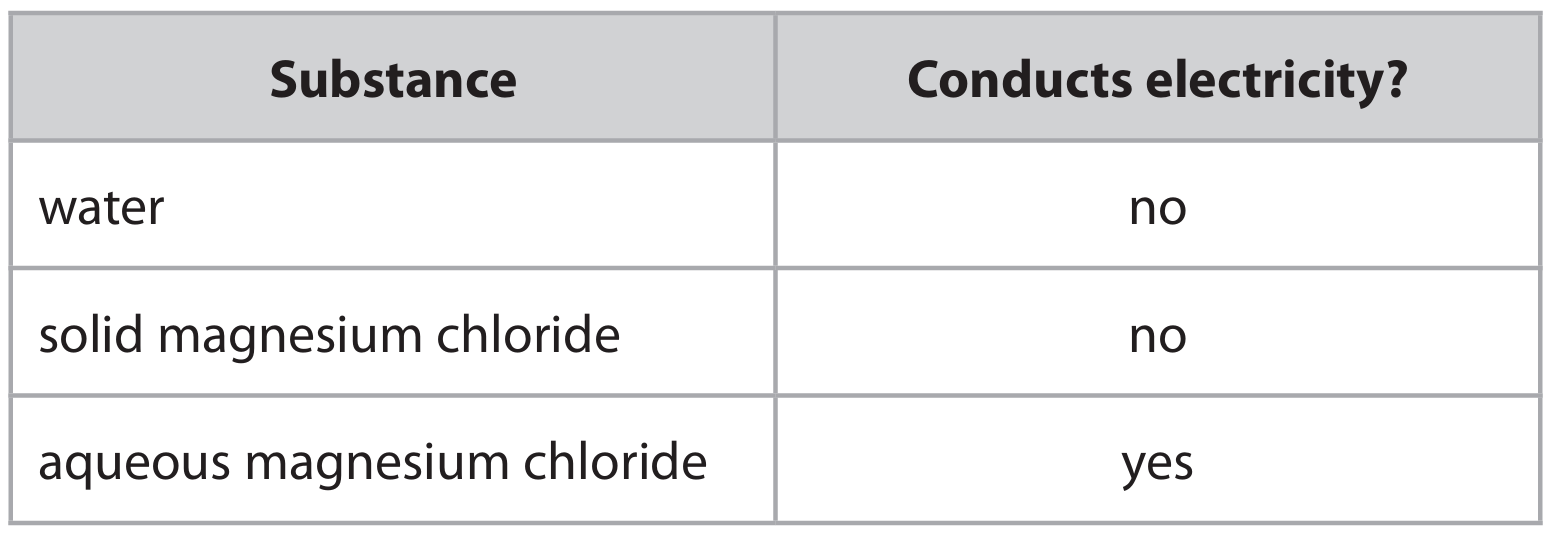
Explain these observations. Refer to structure and bonding in your answer.
(e) A concentrated aqueous solution of magnesium chloride is electrolysed. Chlorine forms at the positive electrode.
(i) Give the half-equation for the reaction at the positive electrode.
(ii) Give a reason why this reaction is classified as oxidation.
Most-appropriate topic codes (Edexcel IGCSE Chemistry – 4CH1):
• 2(f)(g): Acids, bases and salt preparations – solubility rules (part b)
• 1(f): Ionic bonding – formation of ions, dot-and-cross diagrams (part c)
• 1(f) & 1(i): Ionic bonding & Electrolysis – electrical conductivity of ionic compounds (part d)
• 1(i): Electrolysis – half-equations, oxidation (part e)
• 2(a)(d): Group 1 & Reactivity series – properties of Group 2 metals (context) (question context)
▶️ Answer/Explanation
(a)(i) Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
(a)(ii) \[ A_r = \frac{(76.5 \times 24) + (10.5 \times 25) + (13.0 \times 26)}{100} = \frac{1836 + 262.5 + 338}{100} = \frac{2436.5}{100} = 24.365 \] To one decimal place: \( \mathbf{24.4} \)
(b) B (calcium sulfate)
• A is incorrect as calcium nitrate is soluble.
• C is incorrect as magnesium nitrate is soluble.
• D is incorrect as magnesium sulfate is soluble.
(c)
• Magnesium ion (\( \text{Mg}^{2+} \)): Diagram showing electron configuration 2,8.
• Oxide ion (\( \text{O}^{2-} \)): Diagram showing electron configuration 2,8.
Charges must be clearly indicated on each ion.
(d) Explanation linking the following points:
• Water is a covalent molecule with no free ions or delocalised electrons, so it cannot conduct electricity.
• Solid magnesium chloride is an ionic lattice; its ions are fixed in position and cannot move, so it cannot conduct.
• In aqueous solution, magnesium chloride dissociates into free-moving ions (\( \text{Mg}^{2+} \) and \( \text{Cl}^- \)), which can carry charge and conduct electricity.
(e)(i) Half-equation at the positive electrode (anode): \[ 2\text{Cl}^- \rightarrow \text{Cl}_2 + 2e^- \]
(e)(ii) This is oxidation because chloride ions lose electrons.
This question is about the reactions of ethanol and ethanoic acid.
(a) (i) Ethanol reacts with potassium dichromate(VI) to form ethanoic acid.
Name the other reagent needed in this reaction.
(a) (ii) State the colour change that occurs when ethanol reacts with potassium dichromate(VI).
(b) (i) Ethanoic acid reacts with solid sodium carbonate.
Complete the chemical equation for this reaction.
\[ 2CH_3COOH + Na_2CO_3 \rightarrow \]
\[ + \]
\[ + \]
(b) (ii) State two observations seen in this reaction.
(c) (i) A student prepares the ester, ethyl ethanoate, by reacting ethanol with ethanoic acid.
This is the student’s method:
- mix equal amounts of ethanol and ethanoic acid in a boiling tube
- add a few drops of concentrated sulfuric acid
- place the boiling tube in a hot water bath for several minutes
State the role of sulfuric acid in this reaction.
(c) (ii) State why the student heats the mixture in a water bath instead of directly with a Bunsen burner flame.
(c) (iii) State how the student would know that ethyl ethanoate has formed.
(c) (iv) Draw the displayed formula of ethyl ethanoate.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 4(f): Carboxylic acids — parts (b)(i), (b)(ii)
• 4(g): Esters — parts (c)(i), (c)(ii), (c)(iii), (c)(iv)
• 2(f): Acids, alkalis and titrations / 2(g): Acids, bases and salt preparations — part (b)(i), (b)(ii)
▶️ Answer/Explanation
(a)(i) sulfuric acid / phosphoric acid
ALLOW \( H_2SO_4 \) or \( H_3PO_4 \)
(a)(ii) M1 (from) orange
M2 (to) green
*Must be in the correct order.*
(b)(i)
\[ 2CH_3COOH + Na_2CO_3 \rightarrow 2CH_3COONa + CO_2 + H_2O \]
M1: \( 2CH_3COONa \)
M2: \( CO_2 + H_2O \)
ALLOW \( 2NaCH_3COO \) or \( 2NaOOCCH_3 \)
(b)(ii) Any two from:
• bubbles / fizzing / effervescence
• sodium carbonate / solid disappears / gets smaller
• loss of vinegar smell
ALLOW “dissolves”
(c)(i) catalyst / to speed up the reaction
(c)(ii) ethanol is (in)flammable / can ignite
ALLOW “mixture is flammable”
(c)(iii) distinctive / fruity / sweet smell
ACCEPT “oily liquid”
(c)(iv)
M1: correct ester functional group
M2: rest of molecule correct
Displayed formula: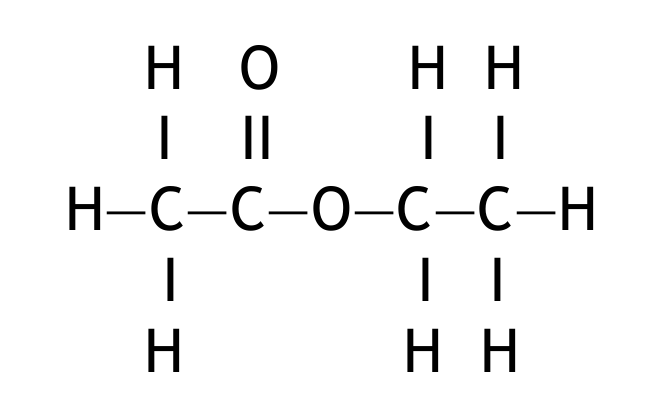
*M2 is dependent on M1.*
A student does a titration to find the concentration of a solution of sulfuric acid.
This is the student’s method.
- fill a burette with sulfuric acid and record the reading
- transfer 25.0 cm\(^3\) of potassium hydroxide solution to a conical flask
- add a few drops of methyl orange to the conical flask
- place the conical flask on a white tile
- swirl the flask continuously while adding sulfuric acid from the burette
- keep adding sulfuric acid until the end-point has been reached
- record the rough reading at the end-point
(a) (i) Name the most suitable piece of apparatus for measuring 25.0 cm\(^3\) of potassium hydroxide solution.
(a) (ii) State why the student places the conical flask on a white tile.
(b) The diagram shows the final burette reading.
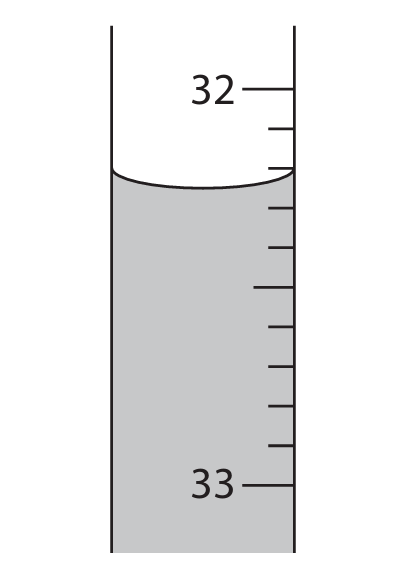
The initial burette reading is given in the table.
Complete the table, giving both values to the nearest 0.05 cm\(^3\).

(c) The student repeats the titration more accurately four times.
The table shows their results.
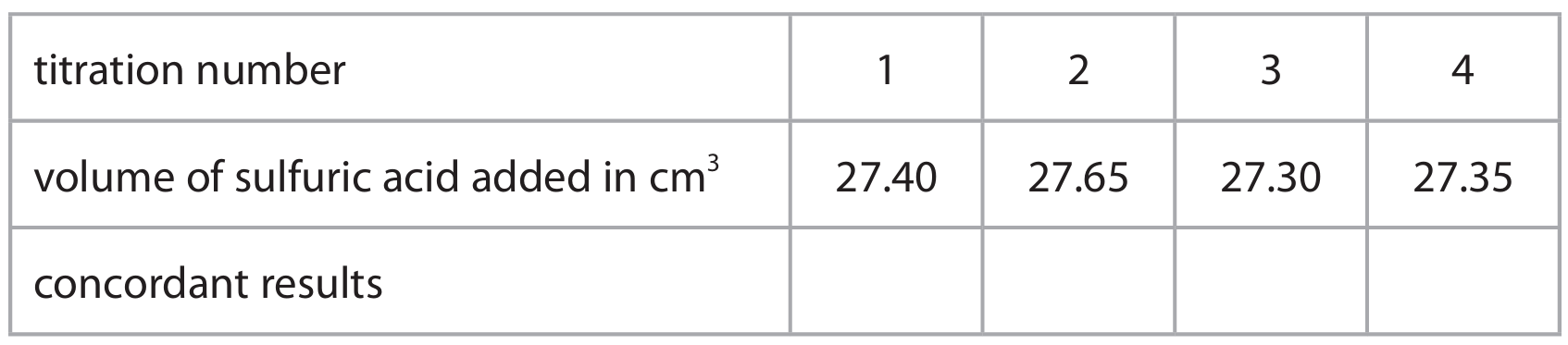
Concordant results are those within \(0.20 \, \text{cm}^3\) of each other.
(i) Place ticks (✓) in the table to show which results are concordant.
(ii) Use the concordant results to calculate the mean volume of sulfuric acid added.
(d) Another student also does the titration.
(i) The student uses 25.0 cm\(^3\) of potassium hydroxide solution with a concentration of 0.500 mol/dm\(^3\).
The mean volume of sulfuric acid added is 26.50 cm\(^3\).
This is the equation for the reaction.
\[2\text{KOH(aq)} + \text{H}_2\text{SO}_4\text{(aq)} \rightarrow \text{K}_2\text{SO}_4\text{(aq)} + 2\text{H}_2\text{O(l)}\]
Calculate the concentration of the sulfuric acid.
(ii) The student repeats the titration without the methyl orange indicator.
They add exactly 26.50 cm\(^3\) of sulfuric acid to the potassium hydroxide solution.
Describe how the student could obtain pure, dry potassium sulfate crystals from the reaction mixture.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(g): Acids, bases and salt preparations — part (d)(ii)
• 1(e): Chemical formulae, equations and calculations — part (d)(i)
• Appendix 6: Suggested practical investigations — practical context throughout
▶️ Answer/Explanation
(a)(i) pipette
(a)(ii) can see colour change (more) clearly / to make the colour change more visible
(b)
Final burette reading = 32.25 cm\(^3\)
Volume of sulfuric acid added = 32.25 – 4.15 = 28.10 cm\(^3\)
(c)(i)
![]()
(c)(ii) Mean volume = \(\frac{27.40 + 27.30 + 27.35}{3} = 27.35 \, \text{cm}^3\)
(d)(i)
Moles of KOH = \(0.0250 \, \text{dm}^3 \times 0.500 \, \text{mol/dm}^3 = 0.0125 \, \text{mol}\)
From equation: \(2 \text{KOH} \equiv 1 \text{H}_2\text{SO}_4\)
Moles of \(\text{H}_2\text{SO}_4\) = \(0.0125 \div 2 = 0.00625 \, \text{mol}\)
Volume of acid = \(26.50 \, \text{cm}^3 = 0.02650 \, \text{dm}^3\)
Concentration = \(\frac{0.00625}{0.02650} = 0.236 \, \text{mol/dm}^3\)
(d)(ii)
• Heat the solution of potassium sulfate until it is partly evaporated / crystals form / a saturated solution is obtained.
• Allow the solution to cool to crystallise.
• Filter the mixture to obtain the crystals.
• Leave the crystals to dry / dry between filter paper / dry in a warm oven / place in a desiccator.
Hydrogen reacts with iodine vapour to form hydrogen iodide, HI. This is the equation for the reaction:
\[H_2(g) + I_2(g) \rightleftharpoons 2HI(g)\] \[\Delta H = +26 \, \text{kJ/mol}\]
(a) (i) Give the test for hydrogen.
(ii) State what the symbol \(\rightleftharpoons\) represents.
(b) In a sealed container at equilibrium, the reaction conditions are 500°C and 2 atm.
(i) The temperature of the reaction mixture is increased to 600°C, but the pressure is kept at 2 atm.
Explain the effect, if any, on the yield of hydrogen iodide at equilibrium.
(ii) The pressure of the reaction mixture is decreased to 1 atm but the temperature is kept at 500°C.
Explain the effect, if any, on the yield of hydrogen iodide at equilibrium.
(c) An excess of hydrogen reacts with 50.8 g of iodine. At equilibrium, the yield of hydrogen iodide is 80%.
Calculate the volume, in \(\text{cm}^3\), of hydrogen iodide produced at rtp. Give your answer in standard form.
\[ \text{[at rtp, molar volume} = 24 \, 000 \, \text{cm}^3] \]
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 3(c): Reversible reactions and equilibria — parts (a)(ii), (b)(i), (b)(ii)
• 3(a): Energetics — part (b)(i)
• 1(e): Chemical formulae, equations and calculations — part (c)
▶️ Answer/Explanation
(a)(i) A lighted / burning splint produces a “pop” sound.
Reject: glowing splint.
(a)(ii) The symbol \(\rightleftharpoons\) represents a reversible reaction.
(b)(i) Yield of HI increases because:
- the forward reaction is endothermic (\(\Delta H = +26 \, \text{kJ/mol}\))
- increasing temperature favours the endothermic direction (to the right).
(b)(ii) No effect on the yield of HI because:
- there are the same number of gas moles on both sides of the equation (2 moles of gas on each side)
- changing pressure has no effect on the position of equilibrium when the number of gas moles is equal.
(c)
Moles of \(I_2\): \[ n(I_2) = \frac{50.8}{254} = 0.20 \, \text{mol} \]
From the equation: \(H_2 + I_2 \rightarrow 2HI\), 1 mole of \(I_2\) produces 2 moles of HI.
Theoretical moles of HI = \(0.20 \times 2 = 0.40 \, \text{mol}\).
Actual moles of HI (80% yield) = \(0.40 \times \frac{80}{100} = 0.32 \, \text{mol}\).
Volume at rtp (molar volume = \(24\,000 \, \text{cm}^3/\text{mol}\)): \[ V = 0.32 \times 24\,000 = 7\,680 \, \text{cm}^3 \]
In standard form: \[ 7.68 \times 10^3 \, \text{cm}^3 \]
Alternative method:
254 g \(I_2\) → \(2 \times 24\,000 = 48\,000 \, \text{cm}^3\) HI (theoretical).
50.8 g \(I_2\) → \( \frac{50.8}{254} \times 48\,000 = 9\,600 \, \text{cm}^3\) HI (theoretical).
80% yield → \(9\,600 \times 0.80 = 7\,680 \, \text{cm}^3 = 7.68 \times 10^3 \, \text{cm}^3\).
