Question
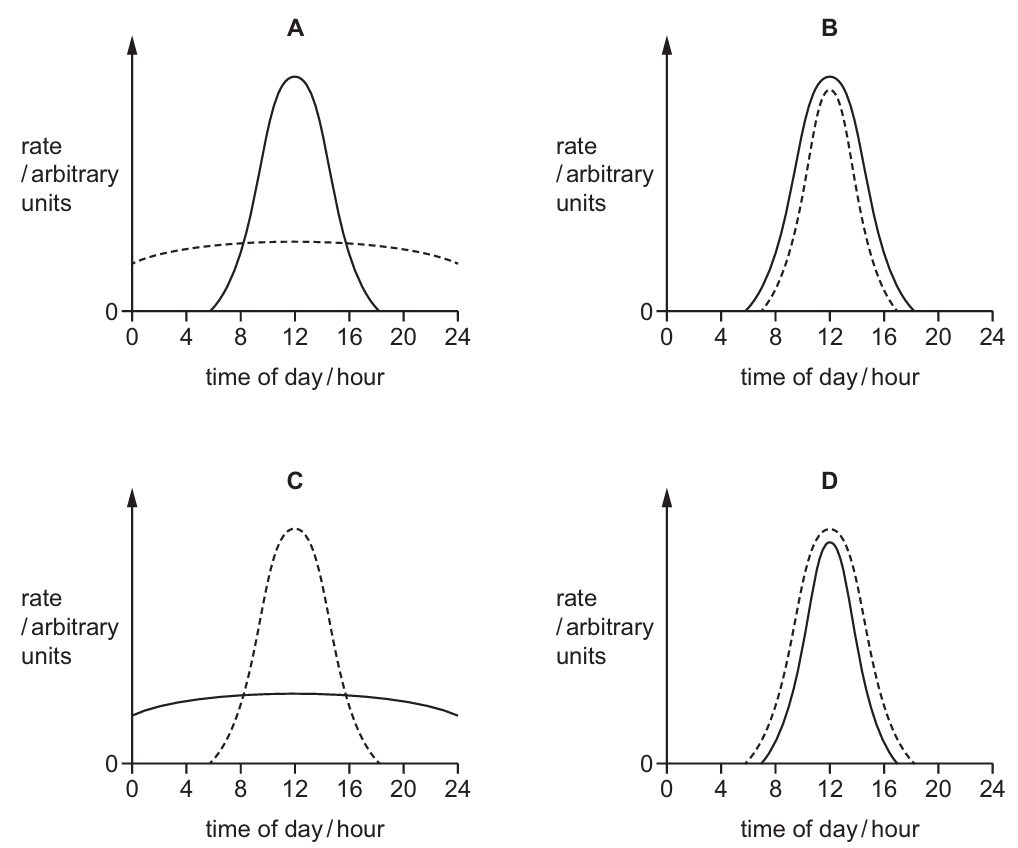
The graphs show how the rate of photosynthesis and the rate of respiration in a plant changed over a $24$-hour period.
Which graph shows the correct rate of photosynthesis and rate of respiration?
▶️ Answer/Explanation
Photosynthesis is a light-dependent process; therefore, its rate is zero during the night and peaks at midday ($12$ hours) when light intensity is highest. Respiration, however, is a continuous process that occurs in all living cells $24$ hours a day to provide energy for survival. While the rate of respiration may fluctuate slightly with temperature changes, it remains relatively constant compared to the dramatic spikes in photosynthesis. Graph A correctly depicts photosynthesis as a bell curve peaking at noon and respiration as a steady, ongoing line throughout the entire $24$-hour cycle. The other graphs incorrectly suggest that respiration stops in the dark or follows light intensity identically.
✅ Answer: (A)
✅ Answer: (A)
Question
The diagram shows an experiment to investigate gas exchange in a leaf.
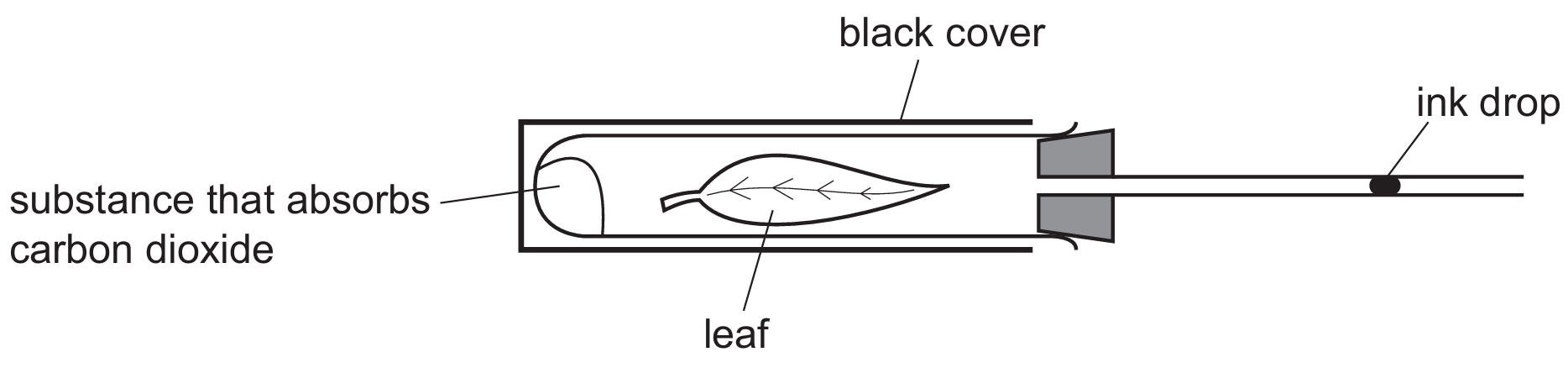
In which direction does the ink drop move and for what reason?

▶️ Answer/Explanation
In this setup, the black cover prevents light from reaching the leaf, meaning photosynthesis cannot occur. Consequently, the leaf only undergoes aerobic respiration, taking in oxygen ($O_2$) and releasing carbon dioxide ($CO_2$). The substance in the tube absorbs any $CO_2$ produced, meaning there is a net decrease in the volume of gas inside the sealed tube as $O_2$ is consumed. This drop in internal pressure pulls the ink drop towards the leaf to equalize the pressure. Therefore, the movement is caused by the depletion of gas during respiration.
✅ Answer: (B)
✅ Answer: (B)
Question
What is the balanced chemical equation for photosynthesis?
(A) $3CO_{2} + 3H_{2}O \rightarrow C_{6}H_{12}O_{6} + 3O_{2}$
(B) $6CO_{2} + 6O_{2} \rightarrow C_{6}H_{12}O_{6} + 6H_{2}O$
(C) $6CO_{2} + 6H_{2}O \rightarrow C_{6}H_{12}O_{6} + 6O_{2}$
(D) $6CO_{2} + 6H_{2}O \rightarrow 6C_{6}H_{12}O_{6} + 6O_{2}$
(B) $6CO_{2} + 6O_{2} \rightarrow C_{6}H_{12}O_{6} + 6H_{2}O$
(C) $6CO_{2} + 6H_{2}O \rightarrow C_{6}H_{12}O_{6} + 6O_{2}$
(D) $6CO_{2} + 6H_{2}O \rightarrow 6C_{6}H_{12}O_{6} + 6O_{2}$
▶️ Answer/Explanation
Photosynthesis is the process by which plants convert carbon dioxide and water into glucose and oxygen using light energy. To balance the equation, we must ensure the number of atoms for each element is equal on both sides. Glucose ($C_{6}H_{12}O_{6}$) contains 6 carbon atoms, requiring $6CO_{2}$ as a reactant. It also contains 12 hydrogen atoms, necessitating $6H_{2}O$. Summing the oxygen atoms on the reactant side gives 18, which is balanced by the 6 oxygens in glucose and the 12 oxygens in $6O_{2}$. Thus, option (C) correctly represents the balanced stoichiometry.
✅ Answer: (C)
✅ Answer: (C)
