iGCSE Physics (0625) 2.1.1 States of matter Paper 3 -Exam Style Questions- New Syllabus
Question
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $1.4$ — Density (Parts $\mathrm{(a)(i)}$, $\mathrm{(a)(ii)}$)
• Topic $2.1.1$ — States of matter (Part $\mathrm{(b)(i)}$)
• Topic $2.1.2$ — Particle model (Part $\mathrm{(b)(ii)}$)
• Topic $2.2.3$ — Melting, boiling and evaporation (Part $\mathrm{(c)}$)
▶️ Answer/Explanation
(a)(i)
For the correct answer:
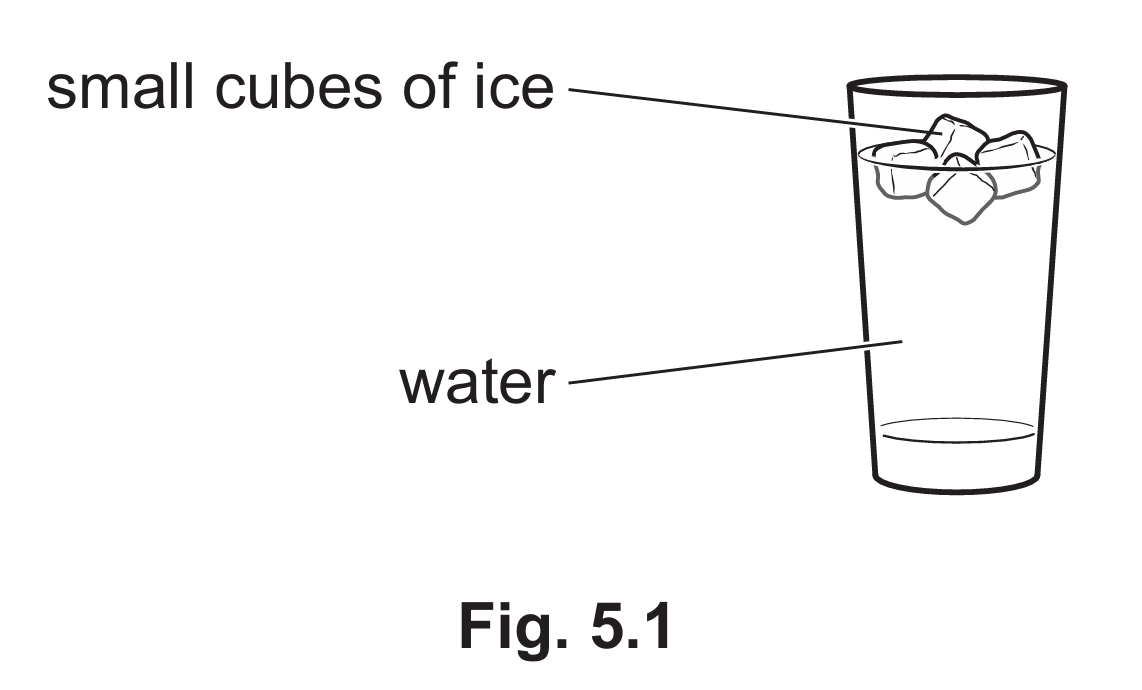
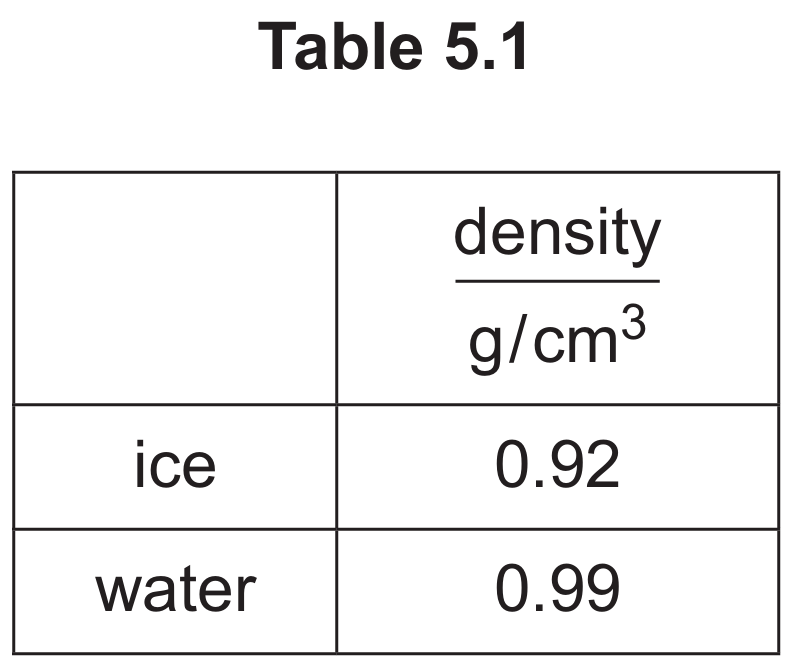
Ice is less dense than water.
The density of ice is $0.92\text{ g/cm}^3$, which is less than the density of water ($0.99\text{ g/cm}^3$). Objects with a lower density than the fluid they are placed in will experience an upthrust greater than their weight when fully submerged. Because ice is less dense than water, it floats on the surface.
(a)(ii)
For the correct answer:
$3.1\text{ g}$
To find the mass, we use the density formula $\rho = \frac{m}{V}$, which rearranges to $m = \rho \times V$. Substituting the given values for the ice cube, we get $m = 0.92\text{ g/cm}^3 \times 3.4\text{ cm}^3$. Calculating this yields a mass of $3.128\text{ g}$, which rounds to $3.1\text{ g}$ to two significant figures.
(b)(i)
For the correct answer:
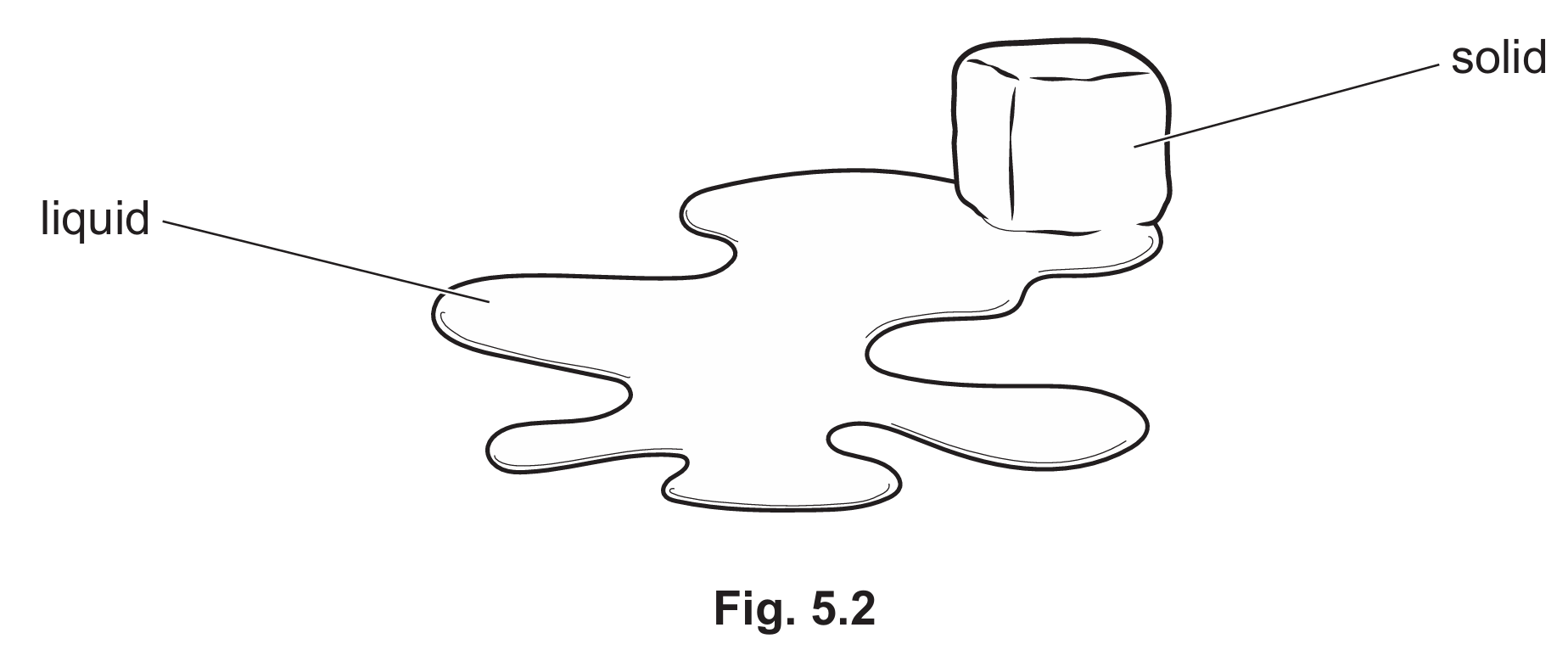
Melting
Melting is the physical process where a substance changes from a solid state to a liquid state. This occurs when the internal energy of the solid increases, usually by the application of heat, until it reaches its melting point.
(b)(ii)
For the correct answer:
Arrangement: regular in solid, random in liquid.
Motion: vibrate in solid, move around / flow over each other in liquid.
In a solid state, particles are arranged in a regular, fixed lattice and can only vibrate around fixed positions. As the ice melts into a liquid, the particles gain energy and the arrangement becomes random and irregular. The particles are no longer fixed and can now freely move around and slide past one another.
(c)
For the correct answer:
Evaporation
Evaporation is the process where molecules at the surface of a liquid gain enough kinetic energy to escape into the air as a gas. Unlike boiling, this process can happen at any temperature below the boiling point, eventually leaving the floor dry as all the water turns into water vapour.
