iGCSE Physics (0625) 2.1.2 Particle model Paper 4 -Exam Style Questions- New Syllabus
Question
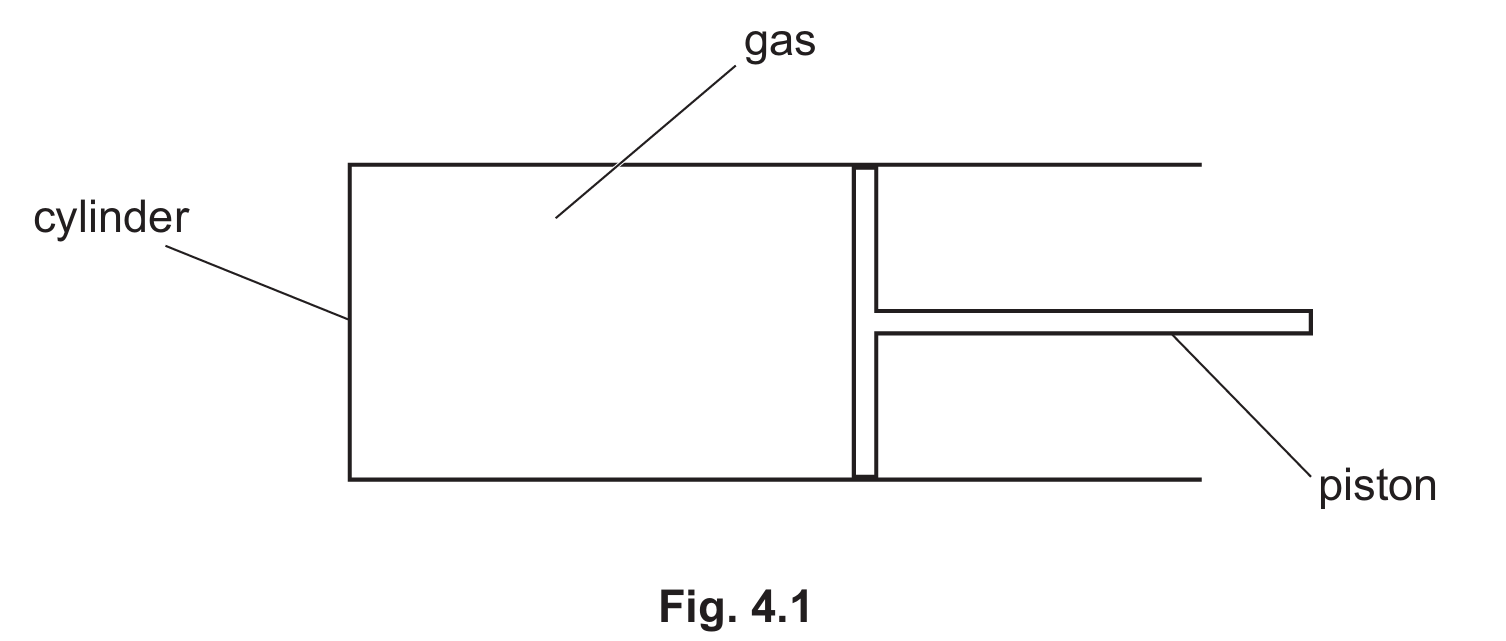
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $2.1 .3$ — Gases and the absolute scale of temperature (Part $\mathrm{(a)(i)}$)
• Topic $1.8$ — Pressure (Part $\mathrm{(a)(ii)}$)
• Topic $1.7.2$ — Work (Part $\mathrm{(a)(iii)}$)
• Topic $2.1 .2$ — Particle model (Part $\mathrm{(b)}$)
▶️ Answer/Explanation
Correct Answer: $170\text{ cm}^3$
Detailed solution: Since the temperature is constant, we use Boyle’s Law: $p_1V_1 = p_2V_2$. Substituting the values: $(1.0 \times 10^5\text{ Pa}) \times (240\text{ cm}^3) = (1.4 \times 10^5\text{ Pa}) \times V_2$. Rearranging for $V_2$ gives $V_2 = \frac{1.0 \times 10^5 \times 240}{1.4 \times 10^5} \approx 171.4\text{ cm}^3$. Rounding to two significant figures as per the input data gives $170\text{ cm}^3$.
Correct Answer: $270\text{ N}$
Detailed solution: Force is calculated using the formula $F = p \times A$. We use the new pressure $p = 1.4 \times 10^5\text{ Pa}$ and the given area $A = 1.9 \times 10^{-3}\text{ m}^2$. Thus, $F = (1.4 \times 10^5\text{ Pa}) \times (1.9 \times 10^{-3}\text{ m}^2) = 266\text{ N}$. Rounding to two significant figures consistent with the precision of the provided values results in $270\text{ N}$.
Correct Answer: $7.9\text{ J}$ (Equation: $W = Fd$)
Detailed solution: Mechanical work done is defined as the product of the average force applied and the distance moved in the direction of the force: $W = Fd$. Using the average force $F = 220\text{ N}$ and distance $d = 0.036\text{ m}$, the calculation is $W = 220\text{ N} \times 0.036\text{ m} = 7.92\text{ J}$. This is expressed to two significant figures as $7.9\text{ J}$.
Detailed solution: In a gas, the particles are very far apart with large empty spaces between them, allowing them to be pushed closer together. In contrast, particles in a liquid are already touching or very close to each other with negligible space between them. Because liquid particles are packed tightly, there is no room for further compression under normal conditions.
