iGCSE Physics (0625) 2.2.2 Specific heat capacity Paper 4 -Exam Style Questions- New Syllabus
Question
(a) A 12 V, 50 W immersion heater is used to heat 0.15 kg of water in a beaker. The water is initially at a room temperature of 20 °C. The specific heat capacity of water is 4200 J / (kg °C).
Calculate the energy supplied to raise the temperature of the water from 20 °C to 58 °C.
(b) The immersion heater is removed from the beaker.
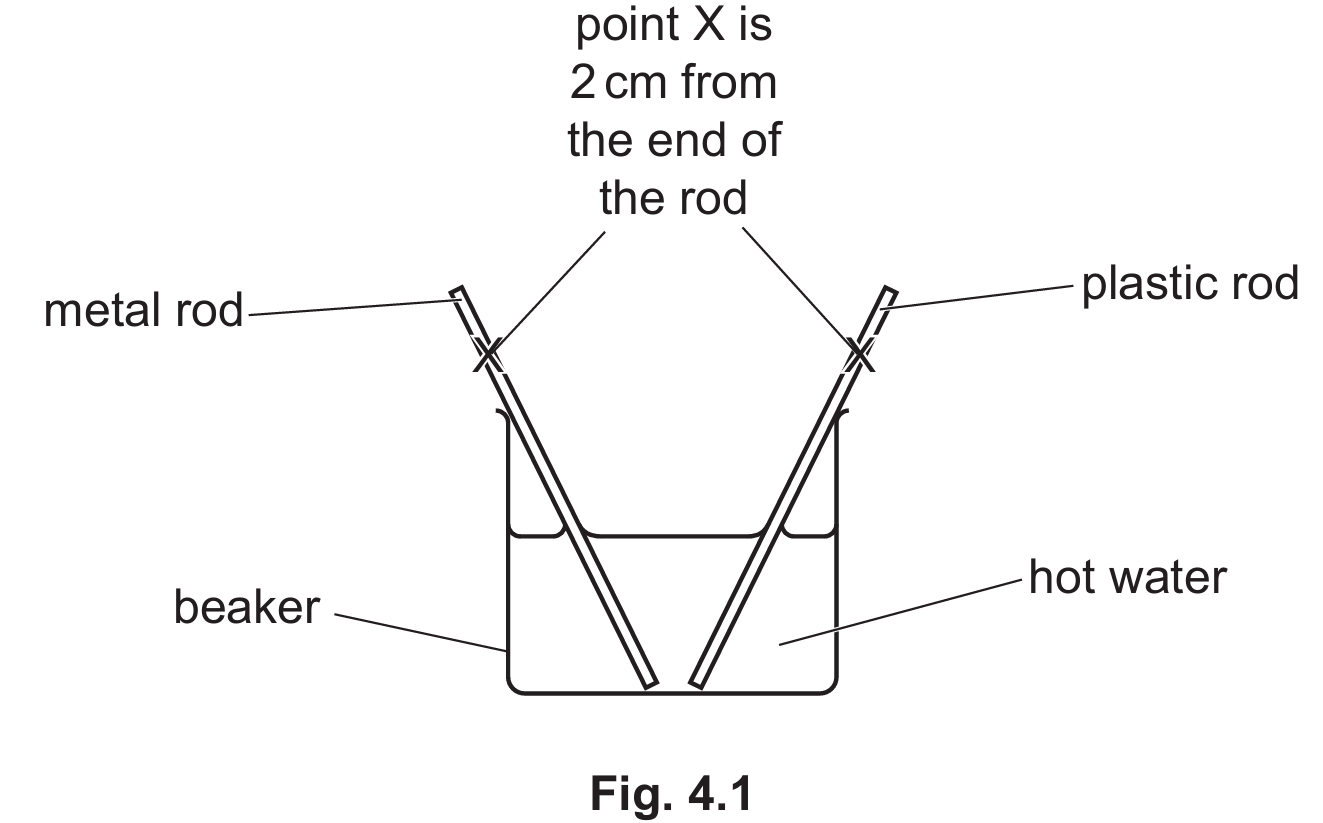
One metal rod and one plastic rod are placed in the beaker of hot water as shown in Fig. 4.1. The rods are at room temperature (20 °C) before they are placed into the beaker.
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $2.2.2$ — Specific heat capacity (Part $\mathrm{(a)}$)
• Topic $2.3.1$ — Conduction (Part $\mathrm{(b)}$)
▶️ Answer/Explanation
(a)
For the correct calculated value:
$24000\text{ J}$ or $2.4 \times 10^4\text{ J}$
To find the energy supplied to the water, we use the specific heat capacity formula: $\Delta E = mc\Delta\theta$. First, calculate the change in temperature ($\Delta\theta$) by subtracting the initial temperature from the final temperature: $58\text{ }^\circ\text{C} – 20\text{ }^\circ\text{C} = 38\text{ }^\circ\text{C}$. Then, substitute the given values into the equation: mass $m = 0.15\text{ kg}$, specific heat capacity $c = 4200\text{ J}/(\text{kg }^\circ\text{C})$, and $\Delta\theta = 38\text{ }^\circ\text{C}$. This gives $\Delta E = 0.15 \times 4200 \times 38 = 23940\text{ J}$. Rounding to an appropriate number of significant figures yields $24000\text{ J}$.
(b)
For the correct answer:
The temperature of point X on the metal rod increases faster than on the plastic rod.
Both rods will experience an increase in temperature at point X over time as thermal energy is transferred from the hot water by conduction. However, the metal rod is a good thermal conductor, while the plastic rod is a thermal insulator. In the metal rod, thermal energy is transferred rapidly due to the fast movement of free (delocalised) electrons. The plastic rod lacks these free electrons and relies entirely on slower lattice vibrations to transfer thermal energy, resulting in a slower temperature rise at point X.
