iGCSE Physics (0625) 2.3.1 Conduction Paper 4 -Exam Style Questions- New Syllabus
Question
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $2.3.1$ — Conduction (Part $\mathrm{(a)}$)
• Topic $2.22$ — Specific heat capacity (Part $\mathrm{(b)}$)
▶️ Answer/Explanation
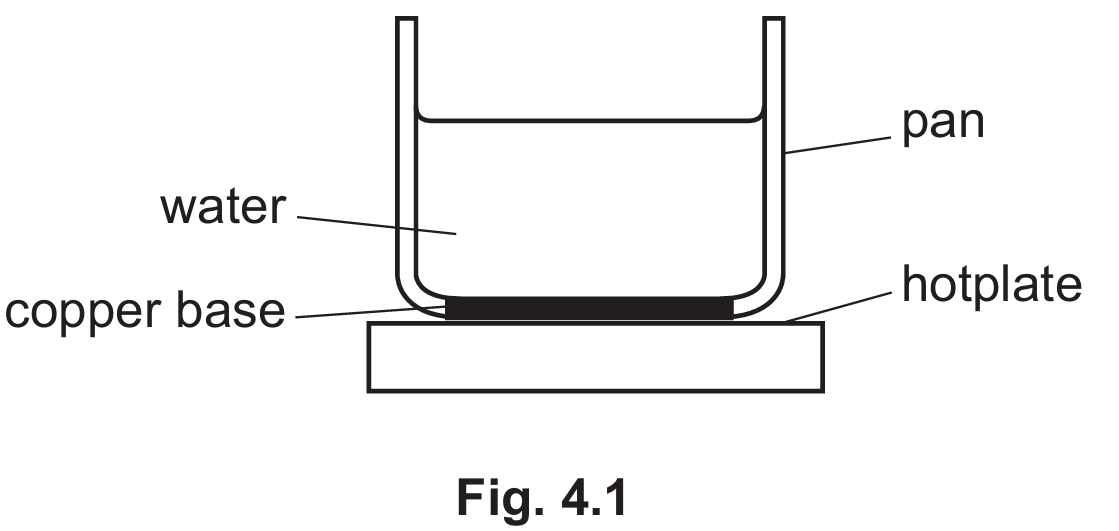
(a)
For the correct answer:
Free electrons carry thermal energy; lattice vibrations transfer energy to neighbouring ions.
Copper is a metal, meaning it contains free (delocalised) electrons. When heated by the hotplate, these free electrons gain kinetic energy and move rapidly through the metal, colliding with distant ions and transferring thermal energy. Additionally, the heated metal ions vibrate more vigorously. These lattice vibrations cause adjacent, cooler ions to vibrate, further transferring thermal energy through the solid copper base.
(b) (i)
For the correct answer:
Energy transferred per unit mass per unit temperature change.
Specific heat capacity is a physical property of matter that dictates how much thermal energy is required to change its temperature. By definition, it is the amount of energy transferred (or required) per unit mass of a substance to cause a unit change in its temperature. In calculations, this relationship is expressed as $c = \frac{\Delta E}{m\Delta\theta}$.
(b) (ii)
For the correct calculated value:
$0.51\text{ J}/(\text{g}^{\circ}\text{C})$
Assuming no heat loss to the surroundings, the thermal energy lost by the hot metal equals the thermal energy gained by the water. The energy gained by the water is: $\Delta E_{\text{water}} = 50 \times 4.2 \times (31 – 22) = 1890\text{ J}$. The energy lost by the metal is: $\Delta E_{\text{metal}} = 54 \times c \times (100 – 31) = c \times 3726$. Equating both expressions yields $c \times 3726 = 1890$, which gives $c = \frac{1890}{3726} \approx 0.507\text{ J}/(\text{g}^{\circ}\text{C})$. Rounding to two significant figures gives $0.51\text{ J}/(\text{g}^{\circ}\text{C})$.
