iGCSE Physics (0625) 2.3.2 Convection Paper 4 -Exam Style Questions- New Syllabus
Question
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $2.3.3$ — Radiation (Parts $\mathrm{(a)(i)}$, $\mathrm{(a)(ii)}$)
• Topic $2.3.2$ — Convection (Part $\mathrm{(a)(iii)}$)
• Topic $2.3.1$ — Conduction (Part $\mathrm{(b)}$)
▶️ Answer/Explanation
(a)(i)
For the correct answer:
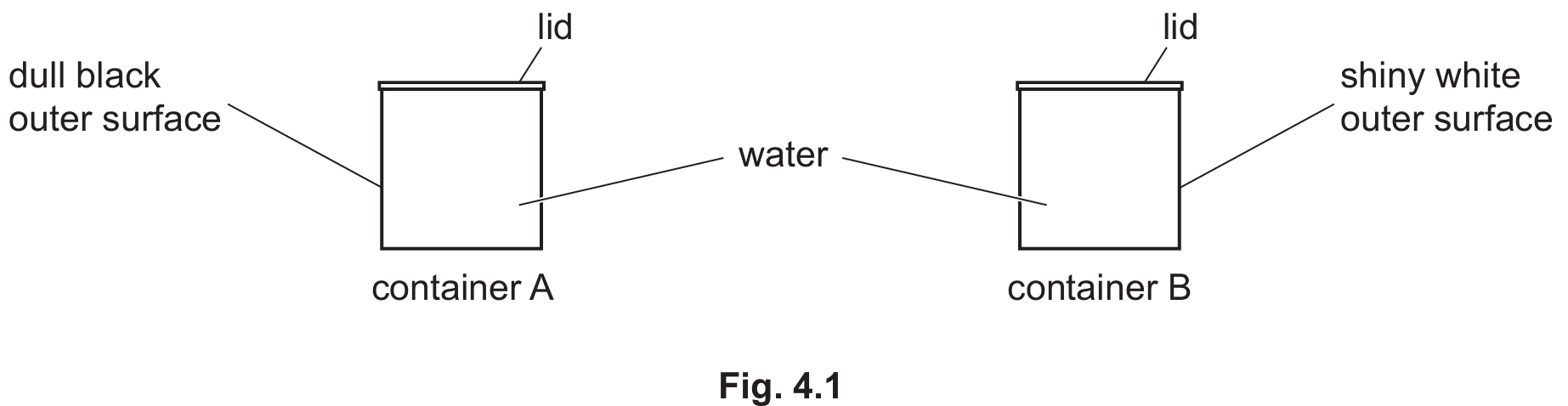
Container A (Dull black surface)
Dull black surfaces are much better emitters of infrared radiation compared to shiny white surfaces. Since container A is dull black, it loses thermal energy to the surroundings via radiation at a significantly higher rate, resulting in a larger temperature change.
(a)(ii)
For the correct answer:
Rate of energy loss equals rate of energy gain.
An object reaches a constant temperature when it is in thermal equilibrium with its surroundings. The rate at which the container transfers thermal energy away is exactly equal to the rate at which it receives energy from the environment.
(a)(iii)
For the correct answer:
Convection (or Evaporation)
Removing the lids allows the air directly above the hot water to be heated, become less dense, and rise, creating a convection current. Additionally, evaporation can occur more freely as energetic molecules escape the surface.
(b)
For the correct answer:
Presence of free (delocalised) electrons in metal.
Metals contain a “sea” of free electrons that can move rapidly. These electrons gain kinetic energy at the hot end and collide with distant ions, transferring energy much faster than the lattice vibrations occurring in wood (an insulator).
