iGCSE Physics (0625) 2.3.3 Radiation Paper 3 -Exam Style Questions- New Syllabus
Question
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic 2.1.2 — Particle model (Part (a))
• Topic 2.1.3 — Gases and the absolute scale of temperature (Part (b))
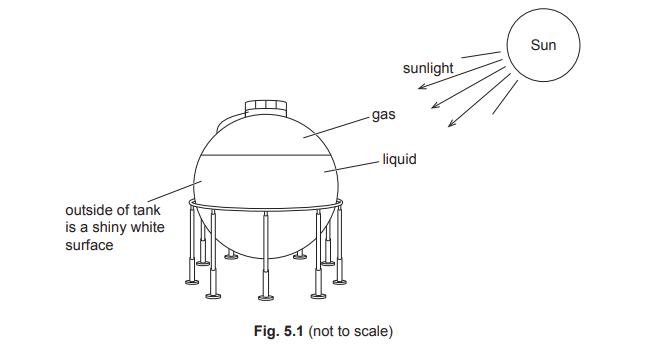
• Topic 2.3.3 — Radiation (Part (c))
▶️ Answer/Explanation
(a)
For the correct answer:
• particles are close together but arranged randomly • particles move randomly and can slide past each other • particles have some vibrational energy and collide with each other
In a liquid, particles are still held closely by intermolecular forces but have enough kinetic energy to move around each other. This gives liquids a fixed volume but no fixed shape, allowing them to flow and take the shape of their container.
(b)
For the correct answer:
pressure increases
• particles move faster / have increased kinetic energy
• more frequent and harder collisions with the container walls
At constant volume, raising the temperature of a gas increases the average kinetic energy of its particles. These faster-moving particles collide with the container walls more frequently and with greater force, resulting in a higher pressure exerted on the container.
(c)
For the correct answer:
infrared radiation (through space and atmosphere)
conduction (through the metal container)
Thermal energy from the Sun reaches the container as infrared radiation, which can travel through the vacuum of space. The metal container absorbs this radiation, heating up. The heat is then transferred through the metal walls to the petrol inside via conduction.
