iGCSE Physics (0625) 5.1.1 The atom Paper 4 -Exam Style Questions- New Syllabus
Question
(i) Observation: most of the particles pass straight through the foil.
(ii) Observation: a few particles bounce back or are deflected through large angles.
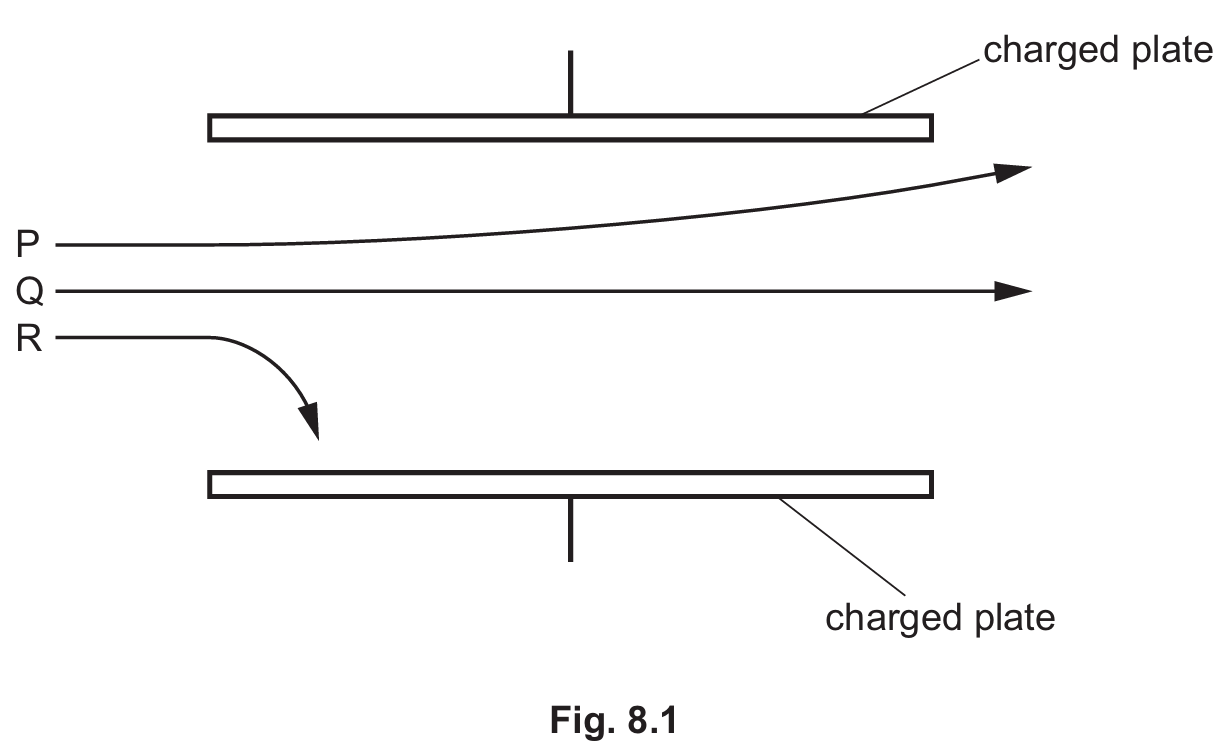
(ii) State and explain the direction of the electric field between the plates.
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $5.1.1$ — The atom (Part $\mathrm{(a)}$)
• Topic $5.2.2$ — The three types of nuclear emission (Part $\mathrm{(b)}$)
• Topic $5.2.4$ — Half-life (Part $\mathrm{(c)}$)
▶️ Answer/Explanation
Correct Answer: (i) Most of the atom is empty space. (ii) The nucleus is positively charged / concentrated mass.
Detailed solution: (i) Since the majority of α particles pass through without any deflection, it implies they do not encounter any obstacles, concluding that the atom is mostly empty space. (ii) Large-angle deflections occur when the positive α particles come close to a concentrated positive center. This confirms the existence of a small, dense, positively charged nucleus containing most of the atom’s mass.
Correct Answer: (i) Path P. (ii) Direction: Upwards.
Detailed solution: (i) Path P represents alpha particles because they are much more massive than beta particles (Path R), resulting in a smaller deflection for the same field strength. (ii) The electric field is directed upwards from the positive plate to the negative plate. Alpha particles are positively charged and thus experience a force in the direction of the field, causing them to curve toward the top (negative) plate.
Correct Answer: $1.9 \times 10^{-2}\text{ g}$
Detailed solution: First, determine the number of half-lives: $n = \frac{480}{160} = 3$ half-lives. To find the initial mass, we multiply the final mass by $2^n$, which is $2^3 = 8$. Calculating the initial mass: $M_0 = 2.4 \times 10^{-3}\text{ g} \times 8 = 19.2 \times 10^{-3}\text{ g}$. This is expressed in standard form as $1.92 \times 10^{-2}\text{ g}$, rounded to $1.9 \times 10^{-2}\text{ g}$ for two significant figures.
