iGCSE Physics (0625) 5.2.2 The three types of nuclear emission Paper 4 -Exam Style Questions- New Syllabus
Question
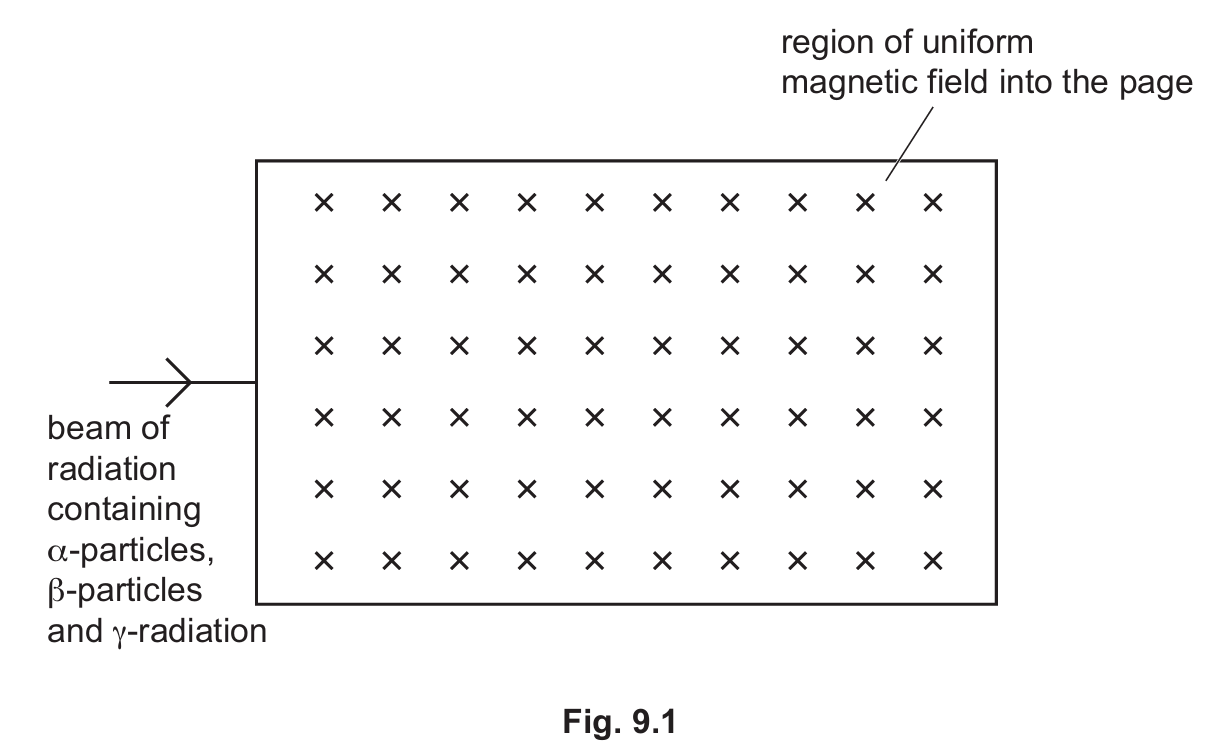
(i) $\alpha$-particles (label this path $\alpha$)
(ii) $\beta$-particles (label this path $\beta$)
(iii) $\gamma$-radiation (label this path $\gamma$)
(ii) Fig. 9.2 shows a simplified diagram of a machine that produces thin sheets of aluminium of constant thickness.
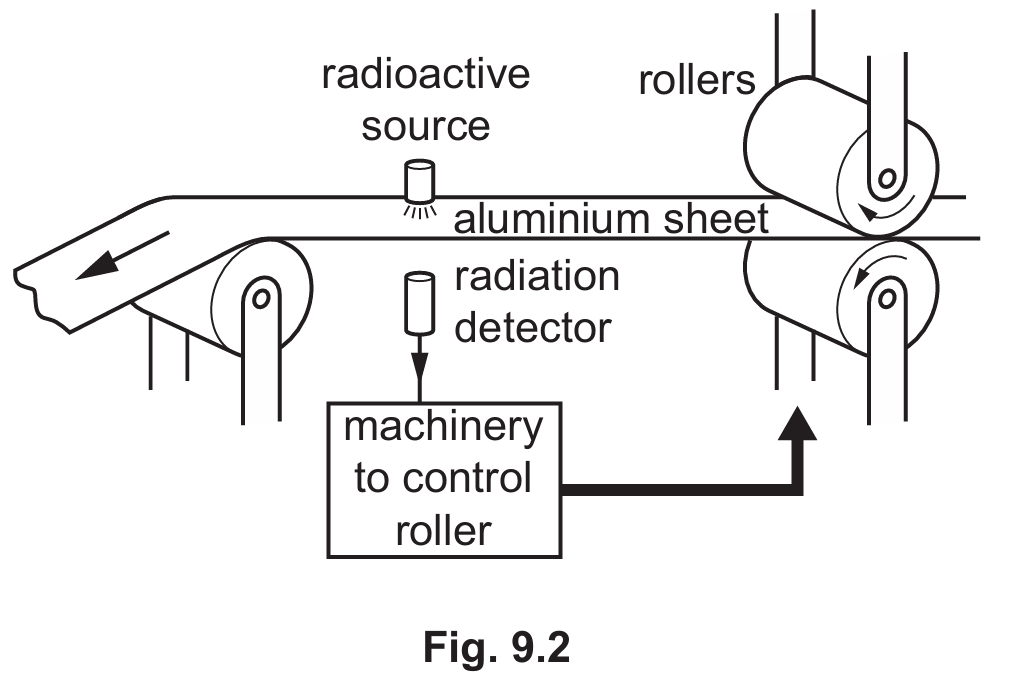
State the most suitable radioactive source in Table 9.1 for the machine in Fig. 9.2.
Explain why this radioactive source is the most suitable and why the other sources are unsuitable.
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $5.2.2$ — The three types of nuclear emission (Describe the deflection of $\alpha$-particles, $\beta$-particles and $\gamma$-radiation in magnetic fields)
• Topic $5.2.4$ — Half-life (Define half-life; Explain how the type of radiation and half-life determine the isotope used for applications)
▶️ Answer/Explanation
Correct Answer: $\alpha$ curves upward; $\beta$ curves downward with a sharper turn; $\gamma$ stays straight.
Detailed solution: Using Fleming’s Left-Hand Rule, positively charged $\alpha$-particles experience an upward force. Negatively charged $\beta$-particles experience a downward force; because they have much less mass, their radius of curvature is smaller (a sharper turn) than $\alpha$. $\gamma$-radiation consists of uncharged electromagnetic waves, so it is not deflected by the magnetic field and continues in a straight line.
Correct Answer: The time taken for half the nuclei of a sample to decay.
Detailed solution: Half-life is a constant characteristic of a radioactive isotope. It can also be defined as the time taken for the activity (or count rate) of a source to fall to half of its initial value. This process is spontaneous and random, meaning we can only predict the behavior of a large number of atoms over this specific time interval.
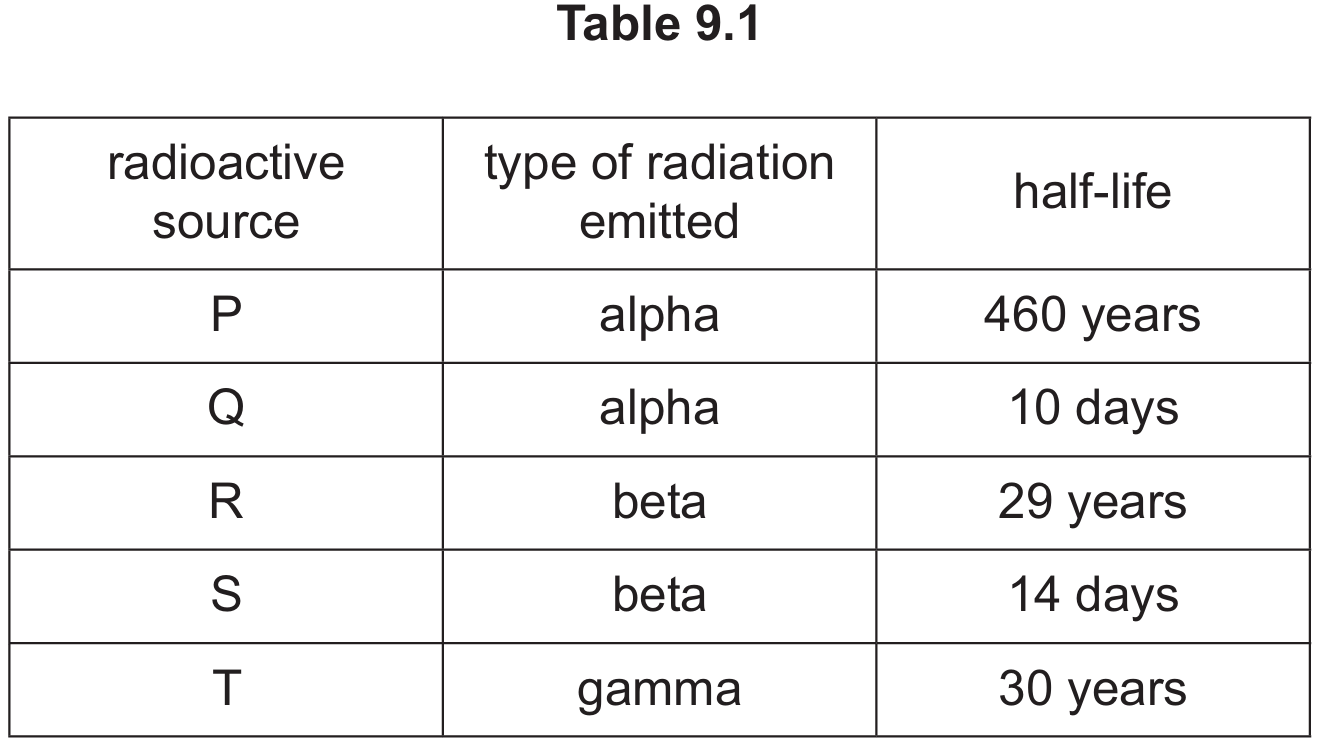
Correct Answer: Source R
Detailed solution: $\beta$-particles (Source R) are ideal because they are partially absorbed by aluminium; if the sheet thickens, the count rate drops, allowing for control. $\alpha$-particles would be completely blocked by any thickness, while $\gamma$-rays would pass through virtually unaffected. Additionally, Source R has a long half-life ($29$ years), ensuring the source does not need frequent replacement and provides a stable count rate over time.
