iGCSE Physics (0625) 5.2.5 Safety precautions Paper 4 -Exam Style Questions- New Syllabus
Question
(ii) Complete the nuclide equation for the decay of strontium-90 to yttrium:
$_{38}^{90}\text{Sr} \longrightarrow \dots \dots \text{Y} + \dots \dots \beta$
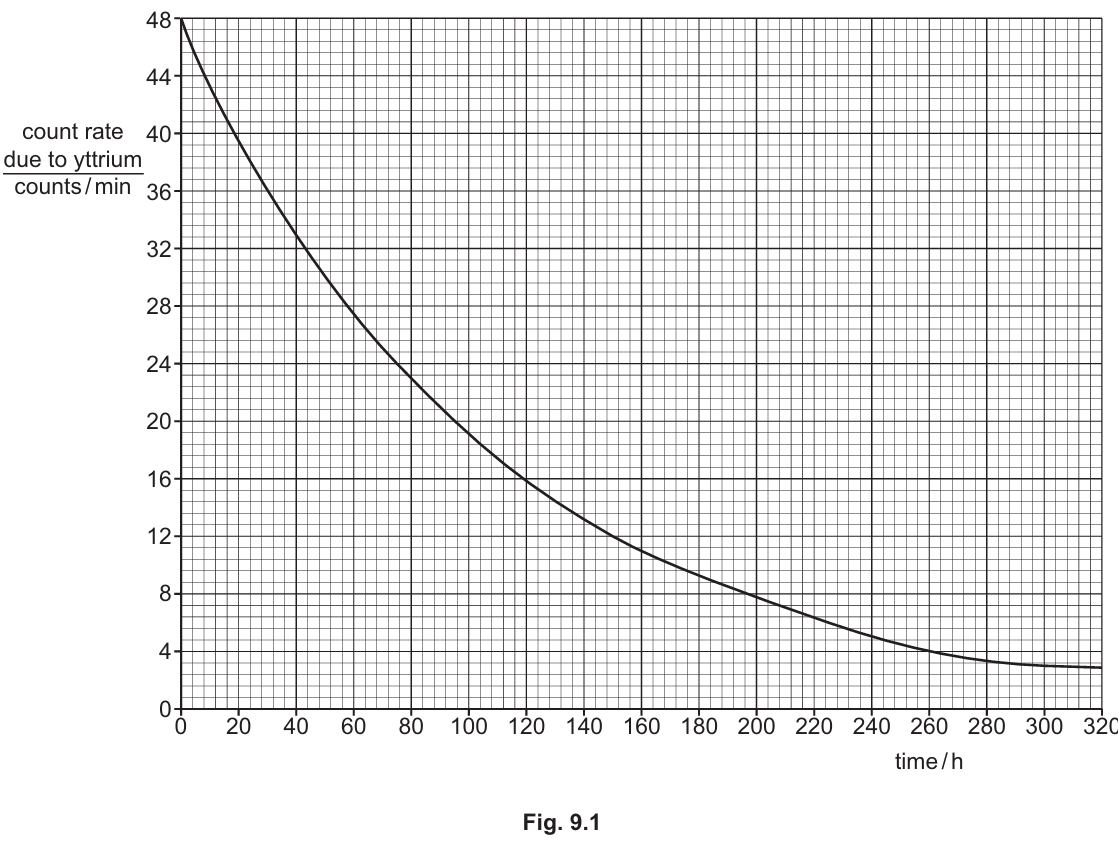
(i) Use Fig. 9.1 to determine the half-life of yttrium. Show your working.
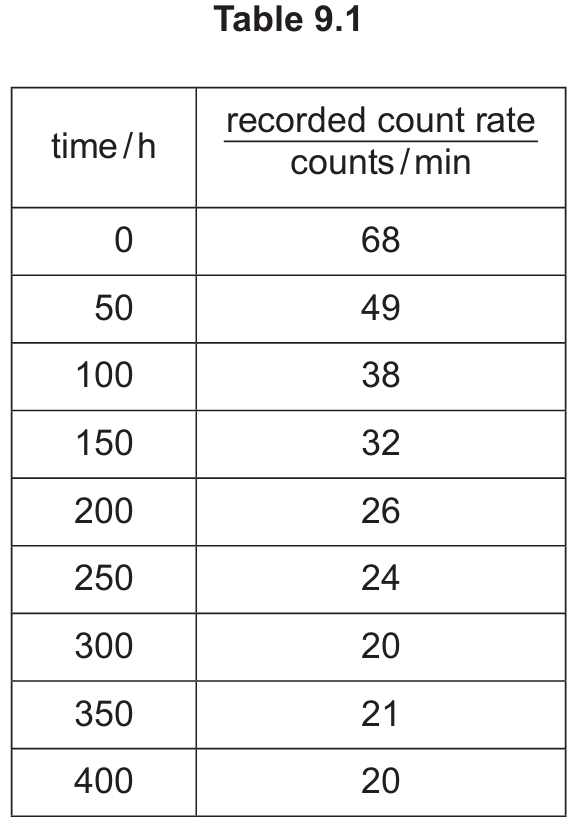
(ii) Explain the difference between the count rate in Table 9.1 and the count rate due to yttrium plotted on the graph in Fig. 9.1.
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic $5.1.2$ — The nucleus (Explain what is meant by an isotope)
• Topic $5.2.3$ — Radioactive decay (Use decay equations to show the emission of $\beta$-particles)
• Topic $5.2.5$ — Safety precautions (State the effects of ionising nuclear radiations on living things)
• Topic $5.2.4$ — Half-life (Calculate half-life from data or decay curves)
• Topic $5.2.1$ — Detection of radioactivity (Use measurements of background radiation to determine a corrected count rate)
▶️ Answer/Explanation
Suggestion: The stable isotope has fewer neutrons.
Detailed solution: Isotopes of the same element must have the same number of protons ($38$ for Strontium) but different numbers of neutrons. Radioactive isotopes like $_{38}^{90}\text{Sr}$ often have an excess of neutrons, making the nucleus unstable. A stable version would typically have a lower neutron-to-proton ratio, meaning it contains fewer than the $52$ neutrons found in $_{38}^{90}\text{Sr}$.
Correct Answer: $_{38}^{90}\text{Sr} \longrightarrow _{39}^{90}\text{Y} + _{-1}^{0}\beta$
Detailed solution: In beta decay, a neutron in the nucleus changes into a proton and an electron (the $\beta$ particle). The nucleon number ($A$) remains $90$ because the total count of protons and neutrons is unchanged. However, the proton number ($Z$) increases by $1$, moving from $38$ to $39$. The beta particle is represented as $_{-1}^{0}\beta$ to balance the charge and mass in the equation.
Detailed solution: Beta particles are a form of ionising radiation, which means they have enough energy to remove electrons from atoms in living cells. This ionisation can lead to molecular damage, resulting in cell death or DNA mutations that may cause cancer. Limiting exposure time reduces the total “dose” of radiation received, thereby decreasing the statistical risk of these harmful biological effects.
Correct Answer: $72\text{ h}$ to $76\text{ h}$
Detailed solution: Half-life is the time taken for the initial count rate to halve. According to Fig. 9.1, the initial count rate at $t = 0$ is $48\text{ counts/min}$. Half of this value is $24\text{ counts/min}$. By drawing a horizontal line from $24$ on the y-axis to the curve and then a vertical line down to the x-axis, the time is found to be approximately $74\text{ h}$ (accepting values within the $72\text{–}76$ range).
Detailed solution: The count rate in Table 9.1 is the “raw” recorded count, which includes both the radiation from the yttrium sample and the background radiation from the environment. In contrast, the graph in Fig. 9.1 plots the “corrected” count rate, where the constant background value (approximately $20\text{ counts/min}$, as seen where the table levels off) has been subtracted to show only the activity due to the yttrium.
