iGCSE Physics (0625) 2.1.2 Particle model -Exam Style Questions Paper 2 - New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
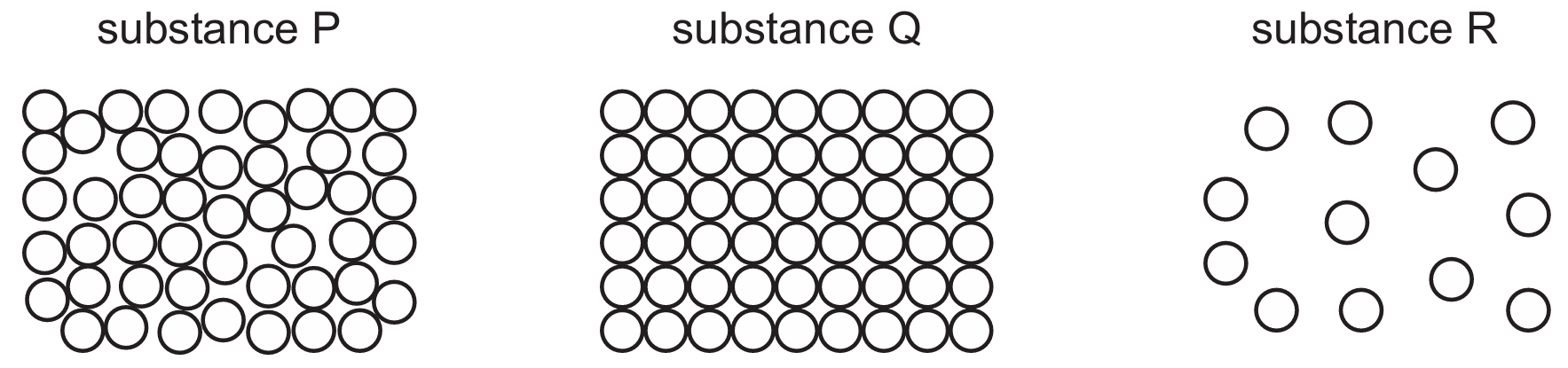
Based on the particle diagrams, substance $Q$ is a solid (regularly arranged), $P$ is a liquid (randomly arranged but touching), and $R$ is a gas (widely spaced).
Gases expand significantly more than liquids, and liquids expand more than solids for the same temperature increase.
Therefore, the order of percentage increase in volume must be: $\text{Gas } (R) > \text{Liquid } (P) > \text{Solid } (Q)$.
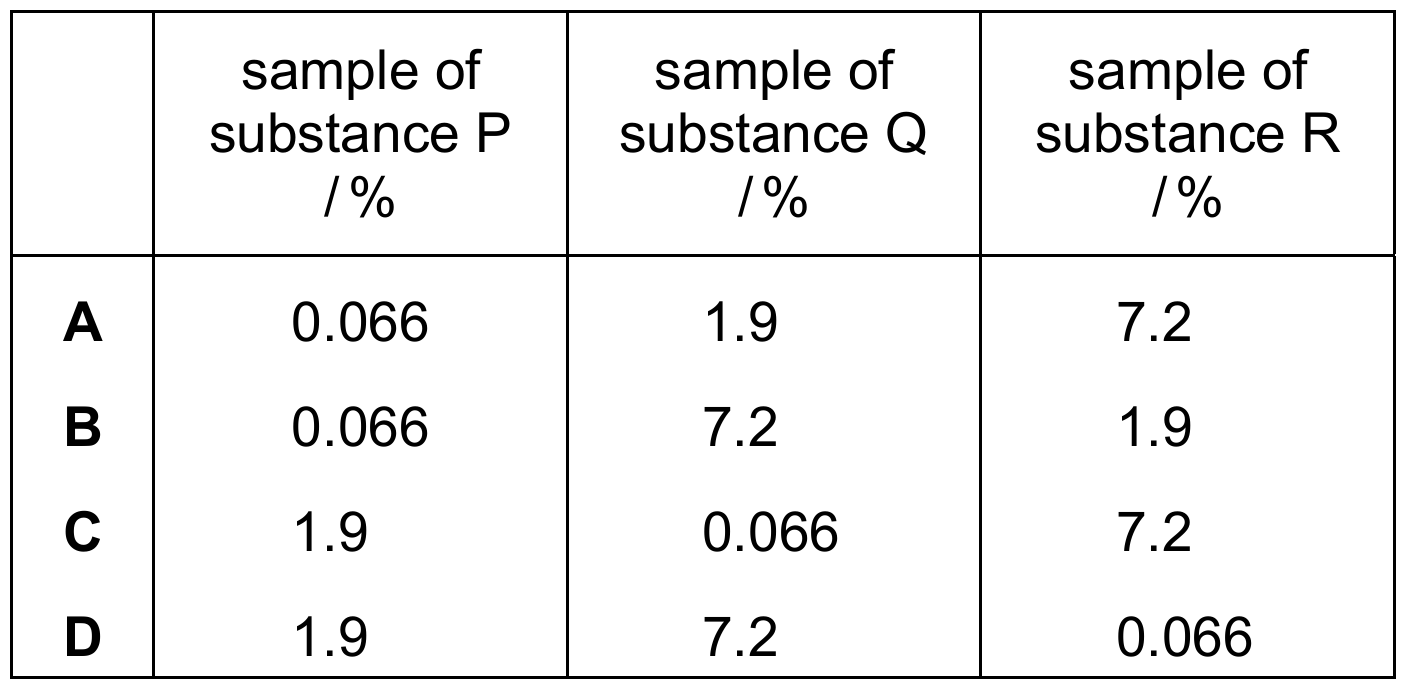
Comparing the values: $7.2\% > 1.9\% > 0.066\%$.
This matches Row C, where $R = 7.2\%$, $P = 1.9\%$, and $Q = 0.066\%$.
Question
▶️ Answer/Explanation
Detailed solution:
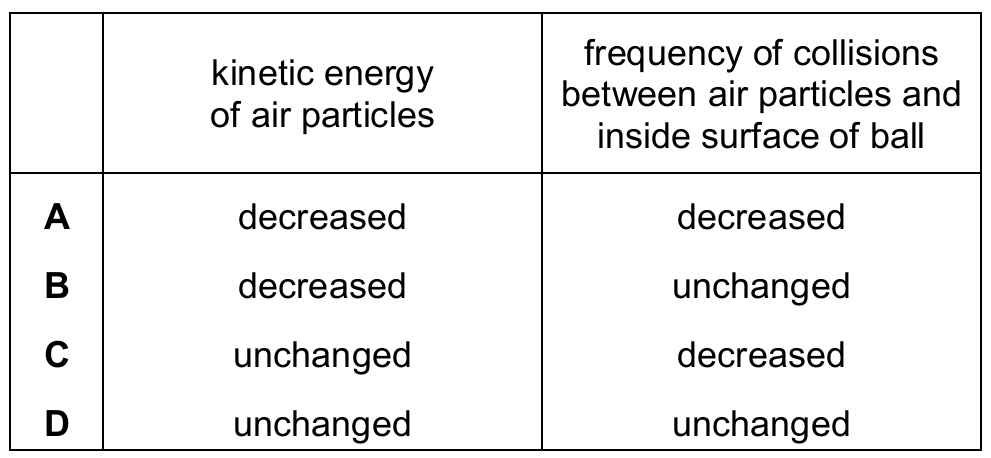
Temperature is a measure of the average kinetic energy (E k ) of the particles; as temperature decreases, the particles move slower and their kinetic energy decreases.
Since the particles move more slowly at a lower temperature, they take longer to travel across the ball and hit the walls.
This leads to a decrease in the frequency of collisions between the air particles and the inside surface of the ball.
Pressure in a gas is caused by these collisions; fewer and less forceful impacts result in lower pressure.
Because both the kinetic energy and the collision frequency decrease, Row A provides the correct explanation.
This follows the kinetic particle model where P∝T for a constant volume.
B. Much heavier fast-moving particles that cannot be seen collide with the microscopic particles.
C. Much lighter fast-moving particles that cannot be seen collide with the microscopic particles.
D. The microscopic particles move to avoid the fast-moving liquid or gas molecules.
▶️ Answer/Explanation
Detailed solution:
Brownian motion provides evidence for the kinetic particle model of matter.
It is the observed random, “zigzag” movement of visible microscopic particles, such as smoke or pollen grains.
This movement is caused by continuous, unequal collisions with much smaller, lighter molecules of the surrounding fluid.
These fluid molecules (atoms or molecules) are too small to be seen even under a microscope but move at high speeds.
Because these molecules are much lighter and faster than the microscopic particles, their impact causes the visible particles to change direction randomly.
Therefore, option C correctly identifies that the collisions involve much lighter, invisible, fast-moving particles.
