iGCSE Physics (0625) 2.2.1 Thermal expansion of solids, liquids and gases-Exam Style Questions- New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
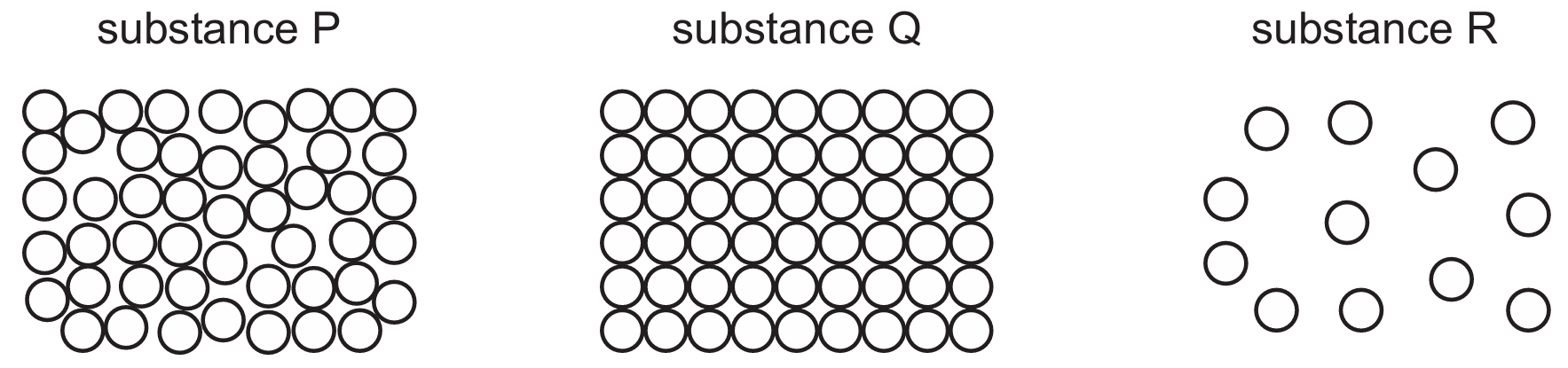
Based on the particle diagrams, substance $Q$ is a solid (regularly arranged), $P$ is a liquid (randomly arranged but touching), and $R$ is a gas (widely spaced).
Gases expand significantly more than liquids, and liquids expand more than solids for the same temperature increase.
Therefore, the order of percentage increase in volume must be: $\text{Gas } (R) > \text{Liquid } (P) > \text{Solid } (Q)$.
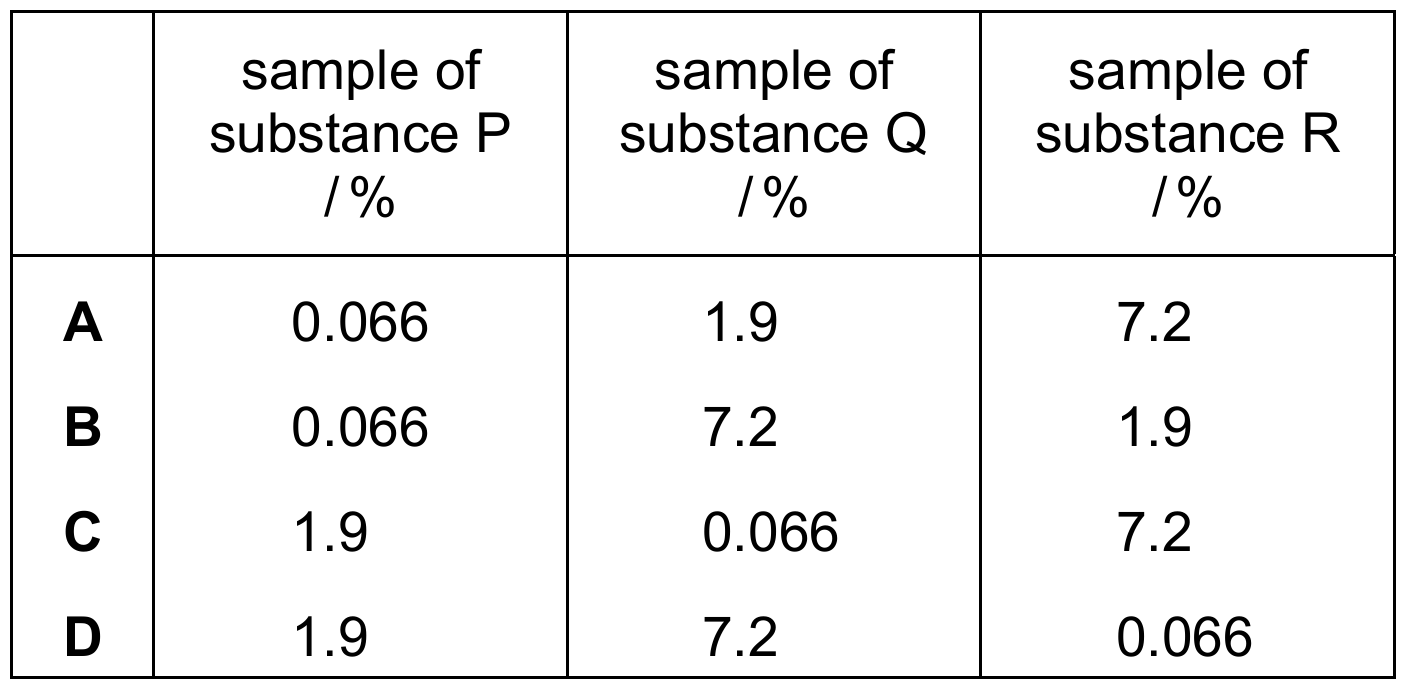
Comparing the values: $7.2\% > 1.9\% > 0.066\%$.
This matches Row C, where $R = 7.2\%$, $P = 1.9\%$, and $Q = 0.066\%$.
Question
▶️ Answer/Explanation
Detailed solution:
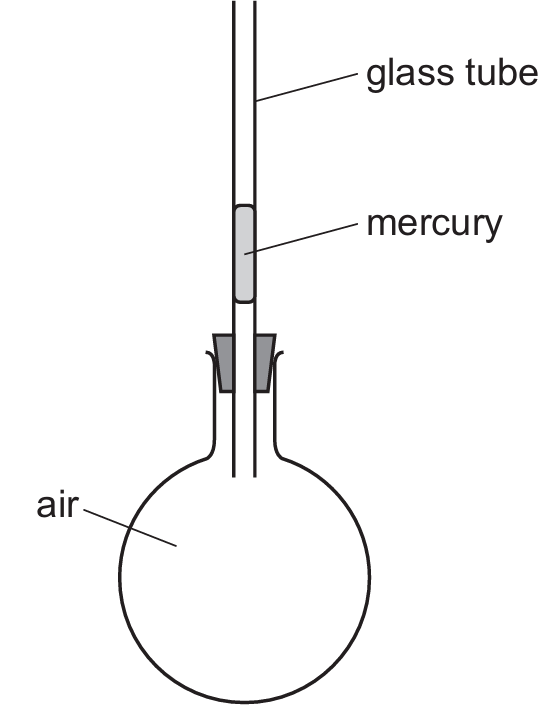
When the flask is warmed, the thermal energy is transferred to the air trapped inside. This increase in temperature causes the air molecules to move faster and push further apart, leading to significant thermal expansion. Gases expand much more than solids or liquids for the same increase in temperature. As the volume of the air increases, it exerts pressure on the mercury thread, forcing it to rise up the glass tube. While the glass and mercury also expand, their expansion is negligible compared to the air. Thus, the expansion of air is the primary cause of the mercury’s movement.
Question
▶️ Answer/Explanation
Detailed solution:
Most materials, including metal cables, undergo thermal expansion when heated and contraction when cooled.
In cold weather, the kinetic energy of the particles decreases, causing the cable to shorten in length.
If the 62 m cable were stretched tightly across the 60 m gap, contraction could cause it to snap or pull the pylons inward.
The extra 2 m of length provides a “slack” that ensures the cable remains intact even at very low temperatures.
Options B, C, and D are incorrect as they describe electrical or magnetic properties unrelated to the physical length of the wire.
