iGCSE Physics (0625) 2.1.3 Gases and the absolute scale of temperature -Exam Style Questions Paper 2 - New Syllabus
Question
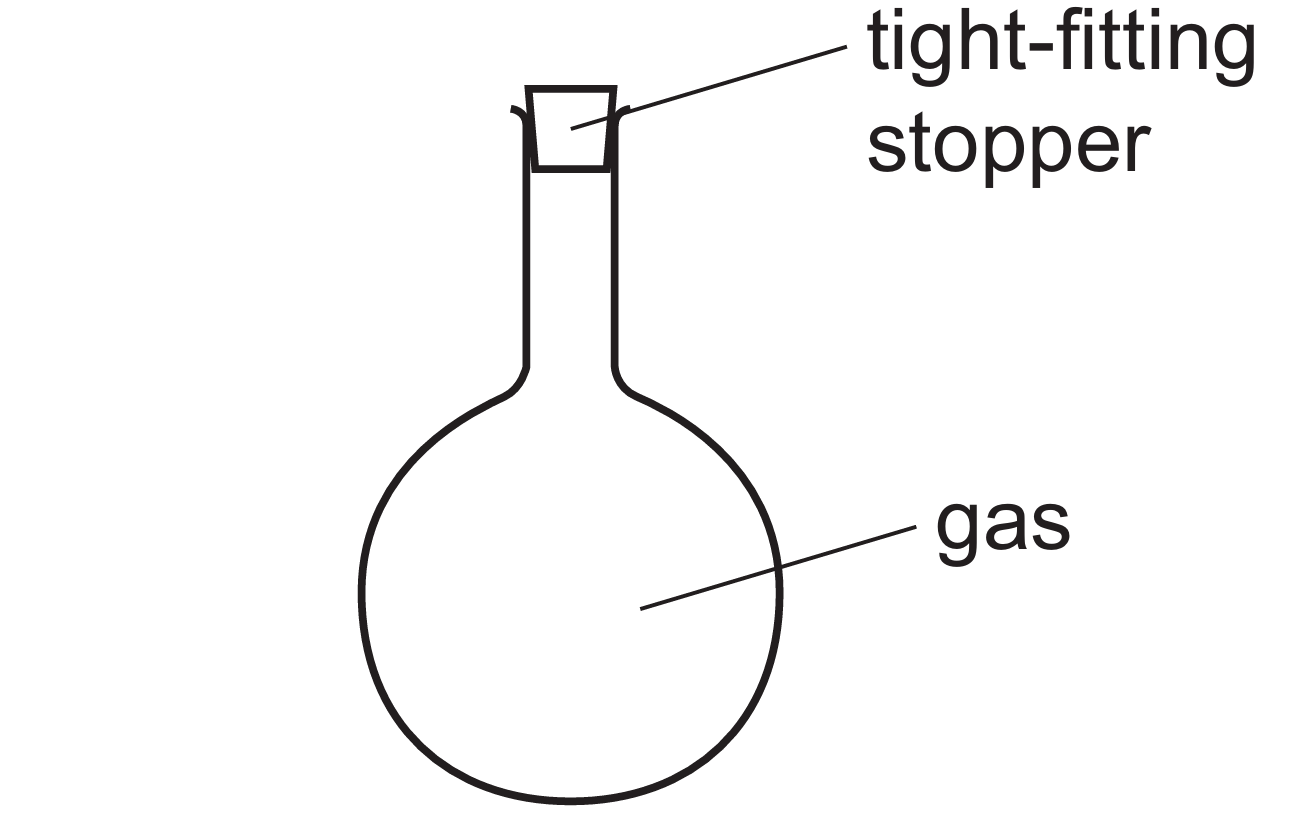

▶️ Answer/Explanation
Detailed solution:
Temperature is a measure of the average kinetic energy of the particles; thus, as the temperature decreases, the average speed of the gas particles also decreases.
The pressure of a gas is caused by particles colliding with the walls of the container, exerting a force per unit area.
When cooled, the slower-moving particles hit the walls less frequently and with less force, leading to a reduction in pressure.
Since the flask is sealed with a tight-fitting stopper, the volume remains constant while the temperature drops.
According to the kinetic particle model, both the particle velocity and the internal pressure must decrease under these conditions.
Therefore, Row A correctly identifies that both the average speed and the pressure decrease as the temperature falls.
Question
▶️ Answer/Explanation
Detailed solution:
Water exists as a liquid between its melting point ($0^{\circ}\text{C}$) and its boiling point ($100^{\circ}\text{C}$) at standard atmospheric pressure.
To convert these values to the Kelvin scale, we use the formula $T\text{ (in K)} = \theta\text{ (in }^{\circ}\text{C)} + 273$.
The melting point in Kelvin is $0 + 273 = 273\text{ K}$, and the boiling point is $100 + 273 = 373\text{ K}$.
Therefore, water is in the liquid state within the temperature range of $273\text{ K}$ to $373\text{ K}$.
Option D correctly identifies this specific interval on the absolute temperature scale.
▶️ Answer/Explanation
Detailed solution:
At constant temperature, a fixed mass of gas follows Boyle’s Law: $P_{1}V_{1} = P_{2}V_{2}$.
Initial values are $P_{1} = 100~kPa$ and $V_{1} = 25~cm^{3}$. The volume decreases by $15~cm^{3}$, so $V_{2} = 25 – 15 = 10~cm^{3}$.
Rearranging to find the new pressure: $P_{2} = \frac{P_{1}V_{1}}{V_{2}} = \frac{100 \times 25}{10} = 250~kPa$.
The question asks for the change in pressure: $\Delta P = P_{2} – P_{1} = 250~kPa – 100~kPa = 150~kPa$.
Thus, the change in pressure is $150~kPa$, which corresponds to option B.
