iGCSE Physics (0625) 2.2.3 Melting, boiling and evaporation -Exam Style Questions Paper 2 - New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
Changes of state that require an input of thermal energy are those where particles must overcome attractive forces to increase their separation.
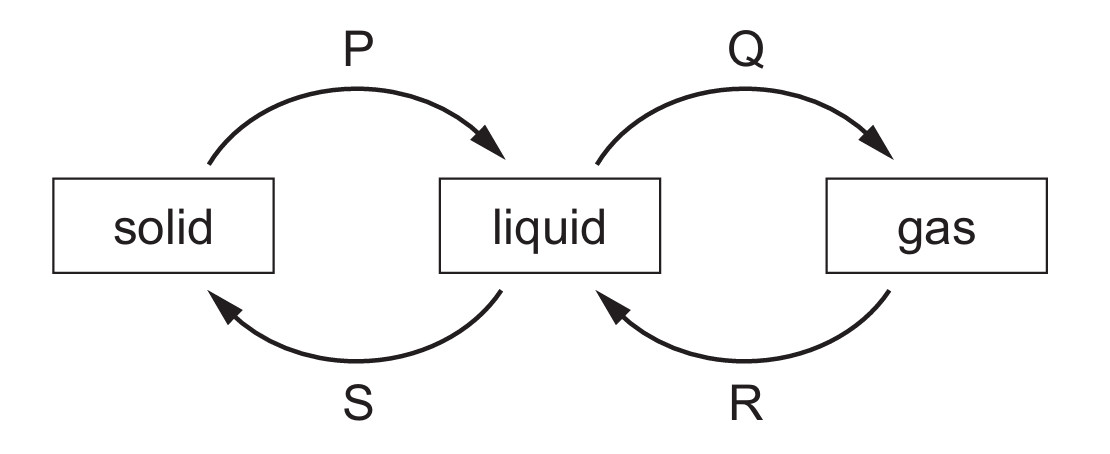
Process $P$ represents melting (solid to liquid), and process $Q$ represents boiling or evaporation (liquid to gas).
In both $P$ and $Q$, energy is absorbed to break or weaken the bonds between particles, increasing their internal potential energy.
Conversely, processes $R$ (gas to liquid) and $S$ (liquid to solid) involve the release of energy as particles move closer together.
Therefore, only $P$ and $Q$ require an external energy input to proceed.
This matches Option A, identifying the endothermic transitions in the heating curve cycle.
Question
The diagram shows the more energetic water molecules escaping from the surface of liquid water.
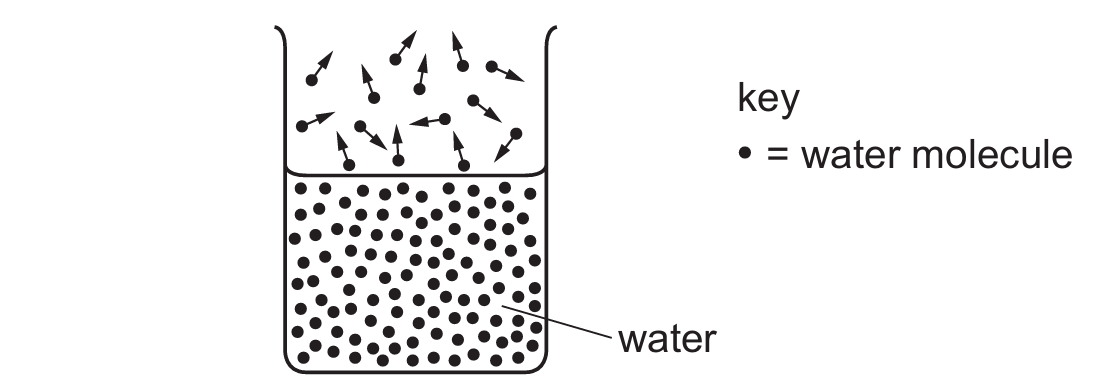
What is this process called?
▶️ Answer/Explanation
Detailed solution:
The diagram clearly illustrates molecules leaving the surface of a liquid state and entering the gaseous state.
In thermal physics, this specific surface-level phase change is defined as evaporation.
During this process, only the most energetic molecules possess sufficient kinetic energy to break free from the intermolecular forces binding the liquid.
Unlike boiling, which occurs throughout the entire volume of the liquid at a specific temperature, evaporation is strictly a surface phenomenon that can happen at any temperature.
Option A is incorrect because Brownian motion refers to the random, erratic movement of suspended microscopic particles.
Option B is the reverse phase change (gas to liquid), and Option D relates to the transfer of thermal energy through a material.
Therefore, the correct term for these escaping water molecules is evaporation.
▶️ Answer/Explanation
Detailed solution:
Evaporation is a surface phenomenon where particles at the liquid’s surface gain enough kinetic energy to overcome intermolecular forces.
Water consists of covalent $H_{2}O$ units known as molecules, rather than isolated atoms or subatomic protons.
During this process, the most energetic individual molecules break free from the liquid phase and enter the gaseous state.
This results in a decrease in the average kinetic energy of the remaining liquid, often referred to as evaporative cooling.
Option D is incorrect because “tiny drops” would still be in the liquid phase and are much larger than individual particles.
Therefore, the correct answer is B, as evaporation involves the escape of individual molecules from the surface.
