iGCSE Physics (0625) 2.3.1 Conduction-Exam Style Questions- New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
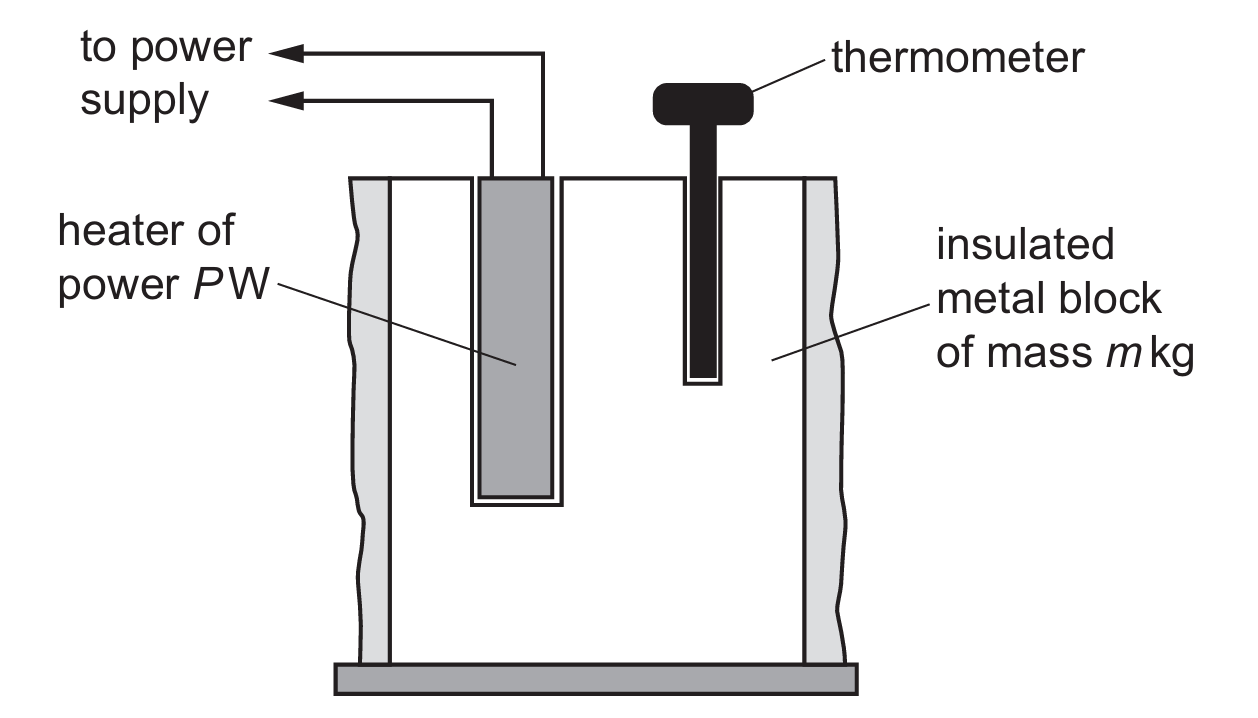
The energy transferred by the heater is given by $\Delta E = P \times t$, where $P$ is power and $t$ is time.
Specific heat capacity $c$ is defined by the thermal energy equation $\Delta E = mc\Delta \theta$.
By substituting the expression for energy, we get $Pt = mc\Delta \theta$.
Rearranging this formula to make $c$ the subject gives $c = \frac{Pt}{m\Delta \theta}$.
This matches the expression provided in Option B.
Question
An experiment is done to compare the rate of conduction of thermal energy in metal and plastic spoons.
An ice cube is placed on one end of each spoon and the other end is heated gently.

Which row has the correct prediction and correct explanation?

▶️ Answer/Explanation
Detailed solution:
Metals are excellent conductors of thermal energy, whereas plastics are poor conductors (often acting as thermal insulators).
When heat is applied to one end of the metal spoon, thermal energy is rapidly transferred along its length to the ice cube.
This rapid transfer of energy causes the ice cube on the metal spoon to absorb heat and melt more quickly than the one on the plastic spoon.
The plastic spoon restricts the flow of thermal energy, meaning much less heat reaches its ice cube in the same amount of time.
Therefore, the correct prediction is that the ice cube melts more quickly on the metal spoon.
The correct explanation for this is simply that metal is a good thermal conductor, perfectly matching row A.
B. Electrons in iron vibrate with more energy than those in rubber.
C. Positive particles in iron can move from one part of the iron to another. Positive particles in rubber cannot move in this way.
D. Positive particles in iron vibrate with more energy than those in rubber.
▶️ Answer/Explanation
Detailed solution:
Thermal conduction in metals like iron occurs through two main mechanisms: lattice vibrations and the movement of delocalised (free) electrons.
In metallic conductors, these free electrons gain kinetic energy in hotter regions and rapidly transfer it to cooler parts by moving through the structure.
Rubber is an insulator and lacks these free electrons; therefore, it relies solely on much slower atom-to-atom vibrations for energy transfer.
Positive ions in a solid lattice remain in fixed positions and do not move from one part of the material to another, making options C and D incorrect.
Option A correctly identifies that the mobility of electrons is the primary reason for iron’s superior thermal conductivity compared to rubber.
