iGCSE Physics (0625) 5.1.1 The atom-Exam Style Questions- New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
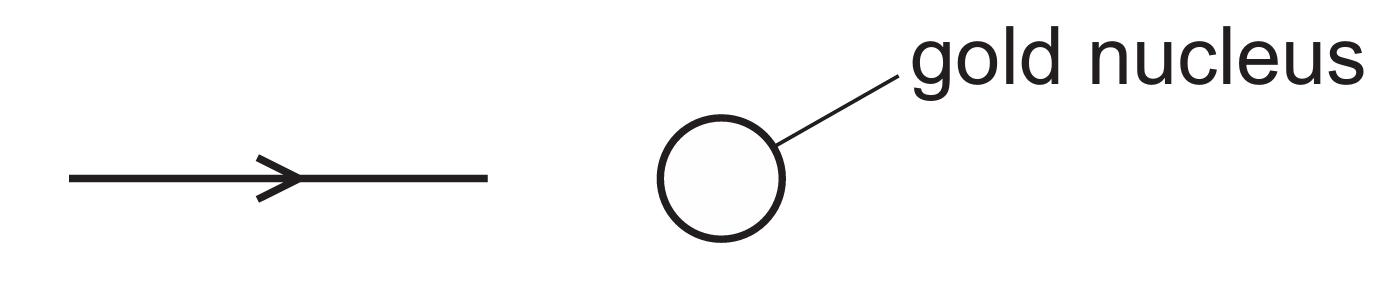
The $\alpha$-particle and the gold nucleus both possess a positive charge ($+2e$ and $+79e$ respectively).
As the $\alpha$-particle travels directly toward the center of the nucleus, it experiences a strong electrostatic force of repulsion.
Since the path is headed straight for the center, the repulsive force acts exactly opposite to the particle’s direction of motion.
This causes the particle to slow down, momentarily stop, and then be repelled directly backward ($180^{\circ}$ deflection).
Option B correctly describes this “back-scattering” effect observed in the Rutherford scattering experiment.
Other paths (A or C) would only occur if the particle was off-center or if the nucleus was not positively charged.
Question
▶️ Answer/Explanation
Detailed solution:
The nuclear model of the atom was established by the Rutherford Gold Foil experiment. When positively charged $\alpha$-particles were fired at a thin metal foil, most passed through, but a small fraction was deflected at large angles. This scattering provided evidence for a very small, dense, and positively charged nucleus containing most of the atom’s mass. Other options, like $\gamma$-ray emission or basic charge attraction, describe radioactive decay or electrostatic forces but do not prove the specific “nuclear” structure. Therefore, the scattering of $\alpha$-particles is the definitive observation for this model.
▶️ Answer/Explanation
Detailed solution:
The large-angle deflection of alpha particles ($\alpha$) occurs due to the electrostatic repulsion between the positively charged alpha particle and the nucleus. For such a significant change in momentum to occur, the positive charge and nearly all the atomic mass must be concentrated in a very small, dense region called the nucleus. Option A explains why most particles pass straight through, but not the deflection itself. Option C describes the “Plum Pudding” model, which was disproved by this experiment as it could not account for large deflections. Option D is incorrect because the center (nucleus) is positively charged, while negative electrons orbit the periphery. Thus, the concentration of mass and positive charge in a small volume (the nucleus) is the only valid explanation for the observed scattering.
