iGCSE Physics (0625) 5.2.2 The three types of nuclear emission-Exam Style Questions- New Syllabus
Question
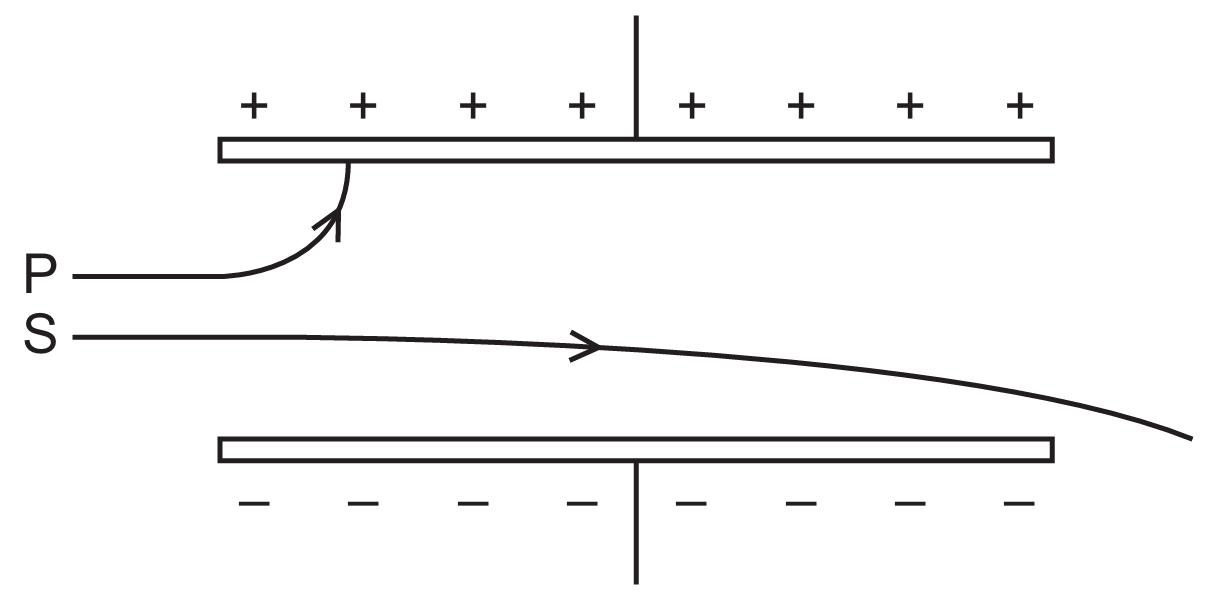

▶️ Answer/Explanation
Detailed solution:
The direction of deflection in an electric field depends on the charge of the radiation. Beam P is deflected toward the positive $(+)$ plate, indicating it carries a negative charge; $\beta$-particles (electrons) are negatively charged and thus attracted to the positive plate. Beam S is deflected toward the negative $(-)$ plate, indicating a positive charge; $\alpha$-particles consist of two protons and two neutrons, giving them a $+2e$ charge. Notably, $\beta$-particles show a larger deflection than $\alpha$-particles because they have a much smaller mass. $\gamma$-radiation carries no charge and would travel straight through without any deflection.
Question
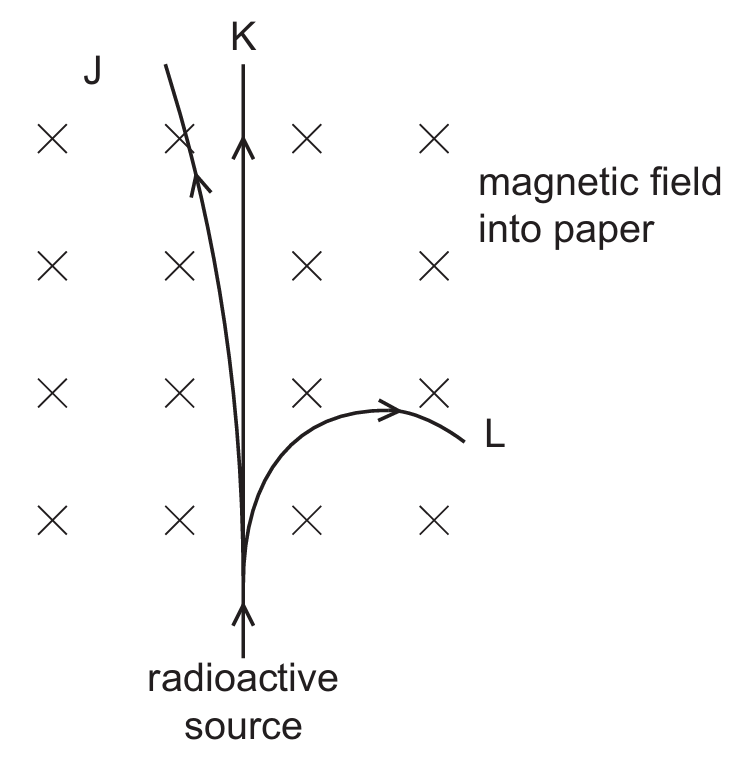
▶️ Answer/Explanation
Detailed solution:
$\gamma$-rays have no charge, so they are not deflected by magnetic fields and follow the straight path $K$.
Using Fleming’s Left-Hand Rule, where the magnetic field (Index finger) points into the paper and current (Middle finger) represents positive charge flow upward:
$\alpha$-particles are positively charged, so the force (Thumb) acts to the left, corresponding to path $J$.
$\beta$-particles are negatively charged, so the equivalent current is downward, resulting in a force to the right, corresponding to path $L$.
$\beta$-particles also have less mass than $\alpha$-particles, leading to the more significant deflection seen in path $L$.
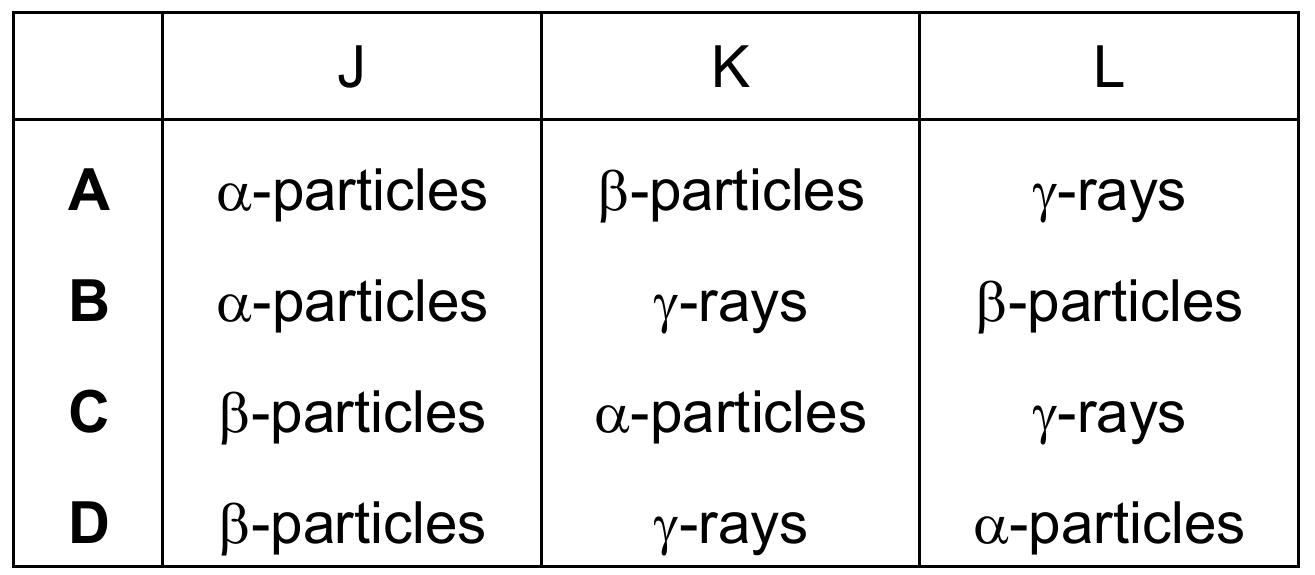
Therefore, path $J$ is $\alpha$, $K$ is $\gamma$, and $L$ is $\beta$, which matches Option B.
▶️ Answer/Explanation
Detailed solution:
To store a source emitting all three types of radiation safely, the shielding must stop the most penetrating type, which is $\gamma$-radiation.
While paper (Option C) stops $\alpha$-particles and a few millimetres of aluminium (Option A) stop $\beta$-particles, they are easily penetrated by $\gamma$-rays.
Lead is a very dense material with a high atomic number, making it highly effective at absorbing ionising radiation.
A thick layer of lead is required to significantly reduce the intensity of $\gamma$-radiation to safe levels.
Therefore, a lead container ensures that $\alpha$, $\beta$, and $\gamma$ emissions are all successfully contained.
This aligns with safety protocols for reducing exposure to ionising nuclear radiations.
