iGCSE Physics (0625) 1.4 Density -Exam Style Questions Paper 2 - New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
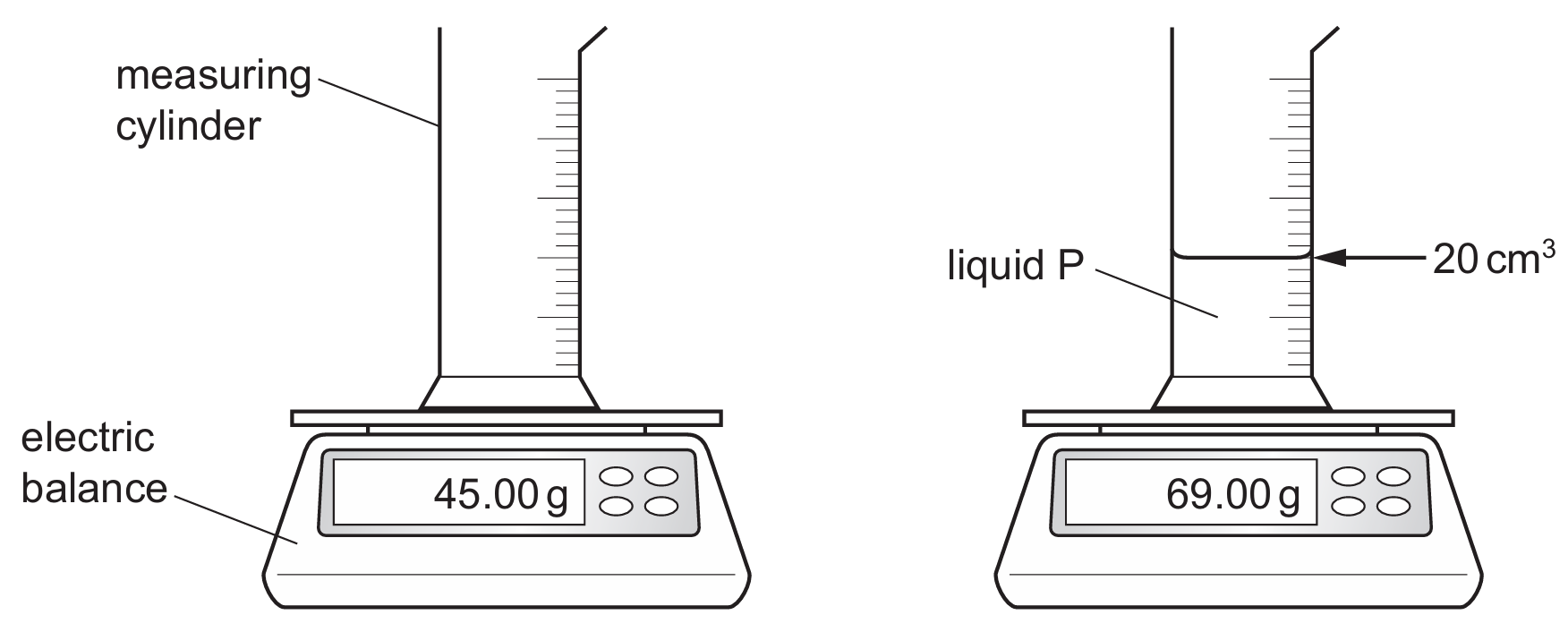
First, calculate the density of liquid P using ρ= V m . The mass of liquid P is 69.00 g−45.00 g=24.00 g and its volume is 20 cm 3 , giving ρ P = 20 24.00 =1.2 g/cm 3 .
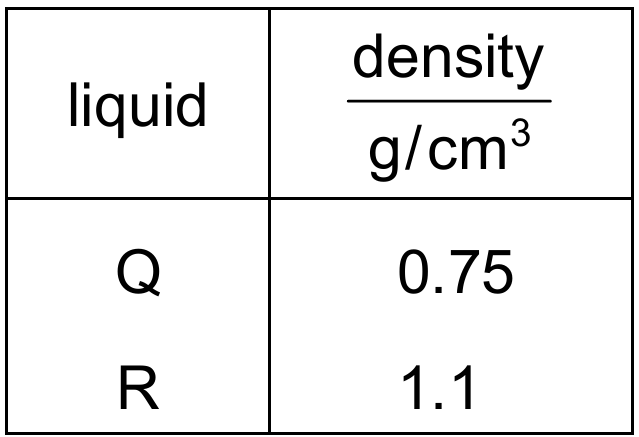
Comparing all densities: ρ Q =0.75 g/cm 3 , ρ R =1.1 g/cm 3 , and ρ P =1.2 g/cm 3 .
Liquids with lower density float above those with higher density.
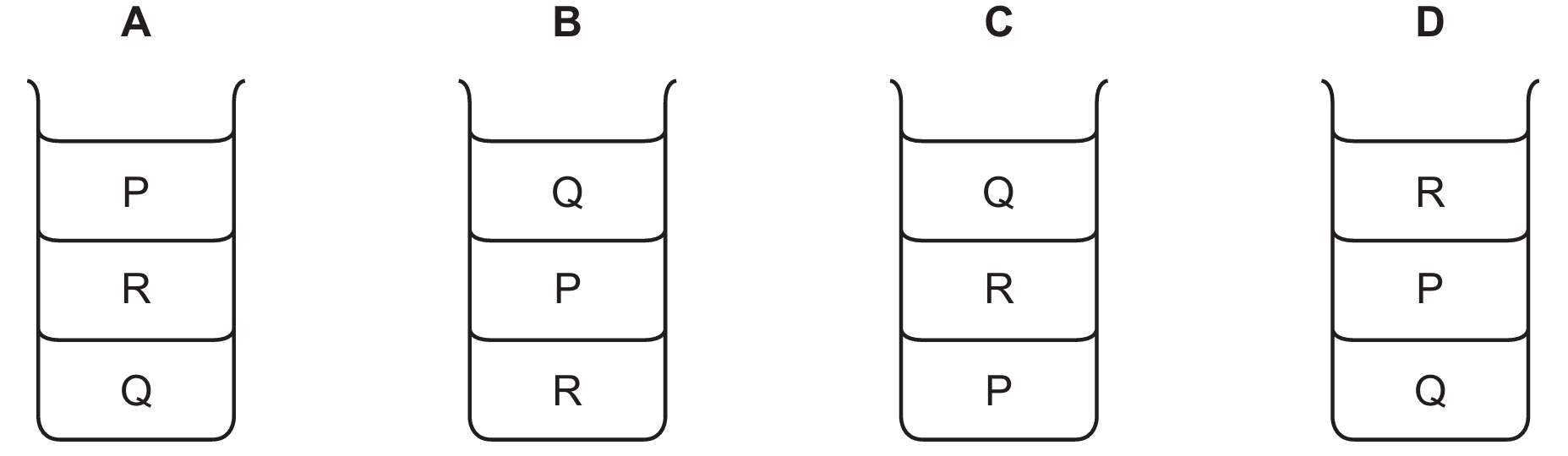
The correct order from top to bottom (lowest to highest density) is Q, then R, then P.
This sequence matches diagram C.
Question
B. The mass of $1\text{ cm}^{3}$ of liquid Q is greater than the mass of $1\text{ cm}^{3}$ of liquid P.
C. The volume of liquid Q is greater than the volume of liquid P.
D. The volume of $1\text{ g}$ of liquid Q is greater than the volume of $1\text{ g}$ of liquid P.
▶️ Answer/Explanation
Detailed solution:
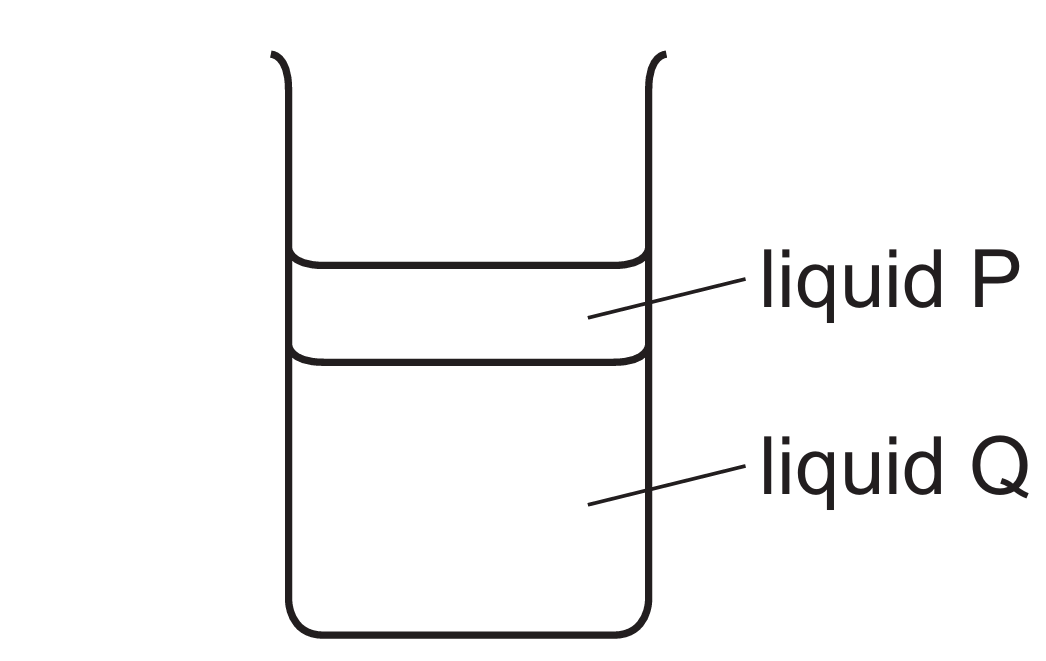
Floating is determined by density, defined as $\rho = \frac{m}{V}$. For two immiscible liquids, the less dense liquid floats on the more dense one.
Liquid P floats on liquid Q because its density is lower: $\rho_{P} < \rho_{Q}$.
Density represents the mass per unit volume; thus, $1\text{ cm}^{3}$ of liquid Q has more mass than $1\text{ cm}^{3}$ of liquid P.
Option A and C are incorrect because total mass and volume do not determine floating behavior.
Option D is incorrect because a greater volume for the same mass implies a lower density, which describes liquid P, not Q.
Therefore, statement B correctly identifies that liquid Q is denser than liquid P.
The volumes of the samples are \(190 cm^3\) , \(200 cm^3\) and \(235 cm^3\) . The liquids are poured into a large measuring cylinder and the liquids form three separate layers because they do not mix. What are the readings on the measuring cylinder at the two boundaries between the samples?
▶️ Answer/Explanation
Detailed solution:
Density is defined as ρ= V m . Since all liquids have equal masses, the liquid with the smallest volume (\(190 cm^3\) ) is the densest and will sink to the bottom.
The liquid with the largest volume (\(235 cm^3\) ) is the least dense and will float at the top, leaving the 200 cm 3 sample in the middle.
The first boundary is at the top of the densest liquid: \(190 cm^3\) .
The second boundary is at the top of the middle layer: \(190 cm^3\) +\(200 cm^3\) =\(390 cm^3\) .
Thus, the two boundary readings are \(190 cm^3\) and \(390 cm^3\) , matching option A.
