iGCSE Physics (0625) 2.2.3 Melting, boiling and evaporation-Exam Style Questions- New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
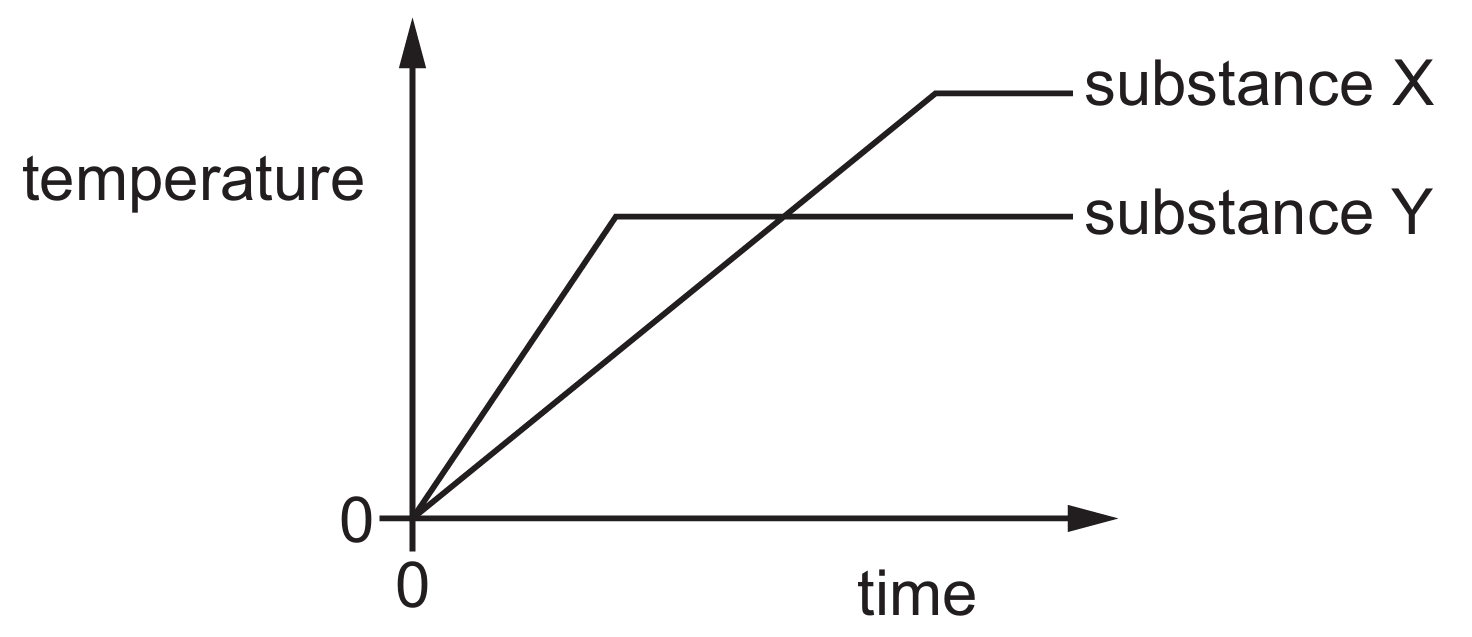
The substances start as solids; the first horizontal section on a heating curve represents the melting point where the temperature remains constant during the phase change.
By observing the vertical axis (temperature), the plateau for substance Y occurs at a lower position than the plateau for substance X.
This indicates that the temperature at which substance Y melts, T m(Y) , is less than the melting temperature of substance X, T m(X) .
Since the graph only shows the transition from solid to liquid, we cannot determine boiling points from this data.
Therefore, the graph confirms that substance Y has a lower melting point than substance X.
Option D correctly identifies this relationship based on the provided temperature-time coordinates.
Question
▶️ Answer/Explanation
Detailed solution:
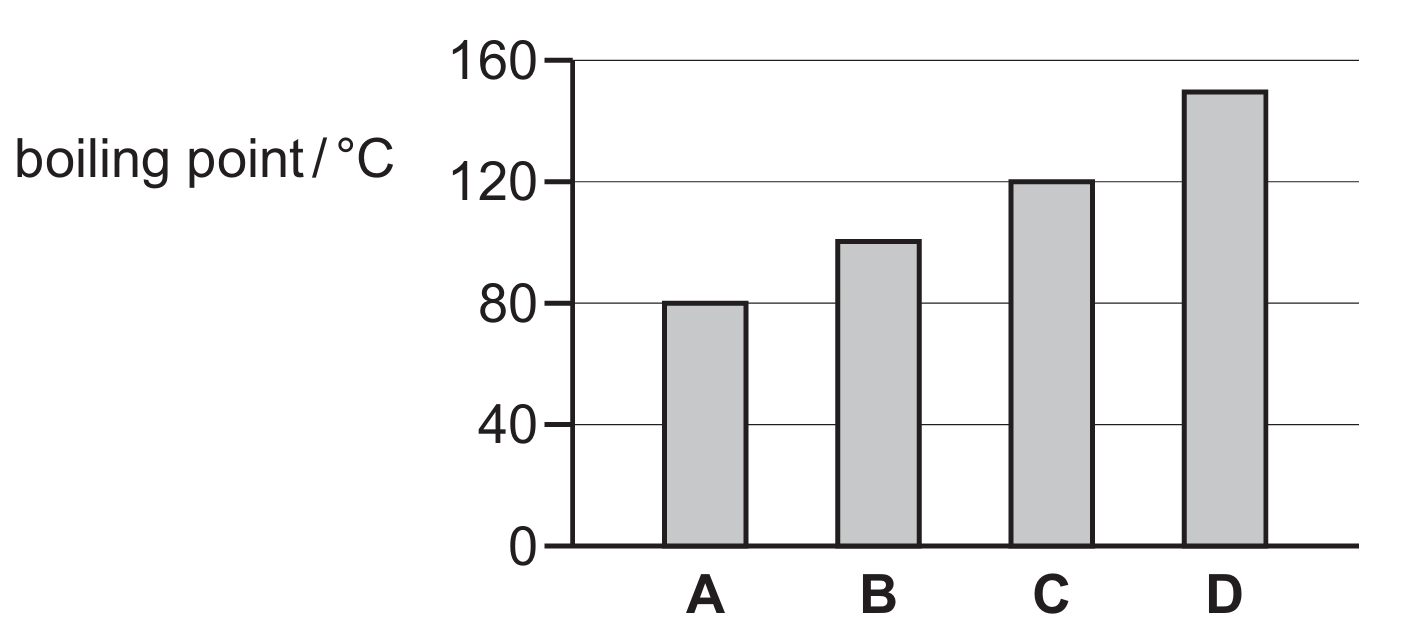
The boiling point of pure water at standard atmospheric pressure is exactly $100^{\circ}C$.
Looking at the provided bar chart, the y-axis represents the boiling point in $^{\circ}C$ with markings at intervals of $40$.
Liquid A shows a boiling point of $80^{\circ}C$, which is below the required value for water.
Liquid B corresponds to the $100^{\circ}C$ mark, as it sits exactly halfway between $80^{\circ}C$ and $120^{\circ}C$.
Liquids C and D have boiling points of $120^{\circ}C$ and approximately $150^{\circ}C$ respectively.
Since only Liquid B aligns with the known physical constant for water, it is the correct choice.
Question
▶️ Answer/Explanation
Detailed solution:
Solidification is the phase change from a liquid to a solid. In the liquid state, particles are close together but have enough energy to slide or move around each other.
As a substance solidifies, the particles lose energy and form a regular, fixed lattice structure where they can only vibrate about fixed positions.
Consequently, the particles stop moving around each other, which is accurately described in Option B.
Crucially, during any change of state, the temperature remains constant (at 0 % accuracy for pure substances) until the process is complete, as energy change involves breaking or forming bonds rather than changing kinetic energy.
Therefore, Options C and D are incorrect because the temperature does not change during the actual solidification process.
