iGCSE Physics (0625) 5.2.5 Safety precautions-Exam Style Questions- New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
Ionising radiation, such as $\alpha$-particles, $\beta$-particles, and $\gamma$-rays, carries enough energy to remove electrons from atoms, creating ions.
When this occurs in living tissue, it can damage $DNA$ molecules directly or through the creation of free radicals.
This damage can lead to cell death if the dose is high, or mutations if the cell repairs itself incorrectly.
Such mutations are a primary cause of the development of cancer over time.
In contrast, an infection is caused by biological pathogens like bacteria or viruses, not by exposure to radiation.
Therefore, infection is the only effect listed that is not a direct result of ionising radiation.
Question
▶️ Answer/Explanation
Detailed solution:
Radiation dose is defined as the total amount of energy absorbed by an organism per unit mass.
Since the source emits radiation at a relatively constant rate, the total dose accumulated is directly proportional to the duration of exposure, t.
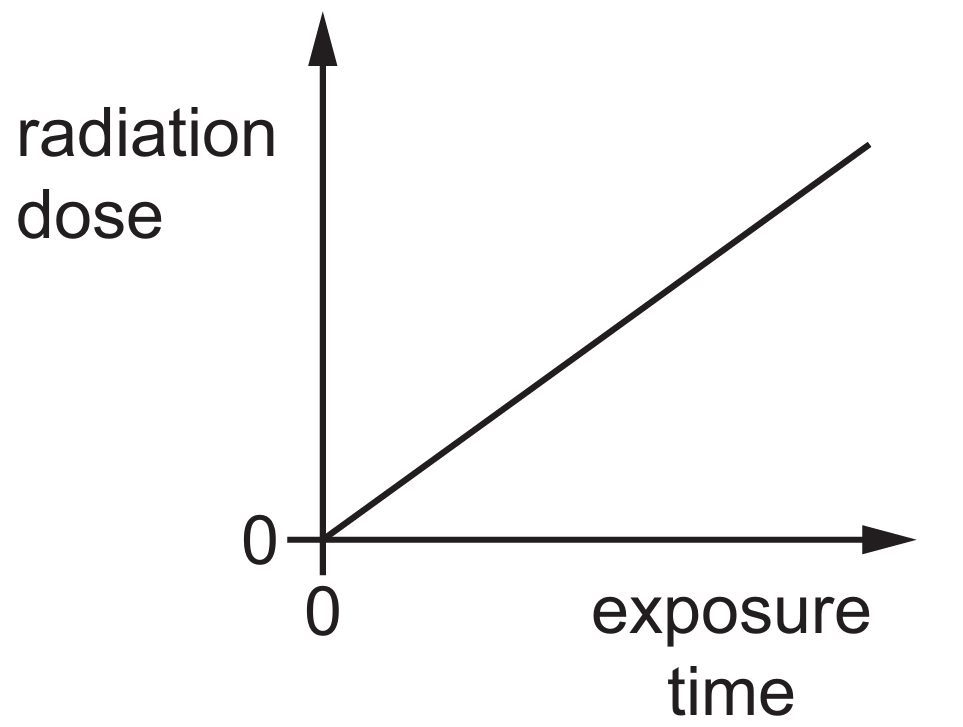
Mathematically, this relationship can be expressed as Dose∝time, which results in a linear trend.
At an exposure time of t=0, the radiation dose received must also be 0, meaning the graph must pass through the origin.
Graph A correctly illustrates this direct proportionality, where the dose increases steadily starting from zero.
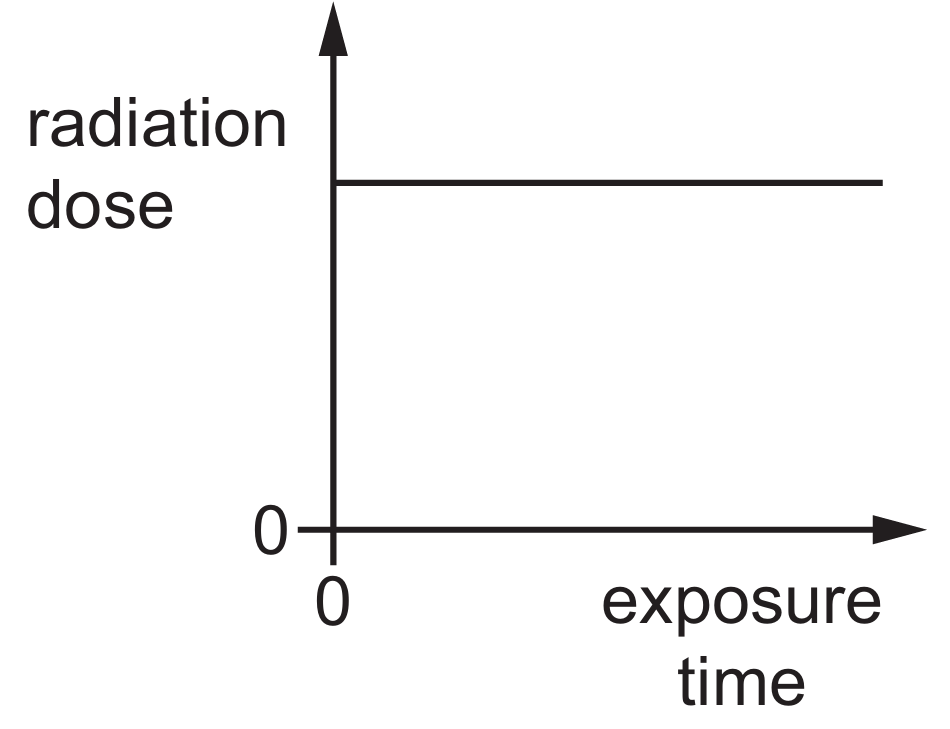
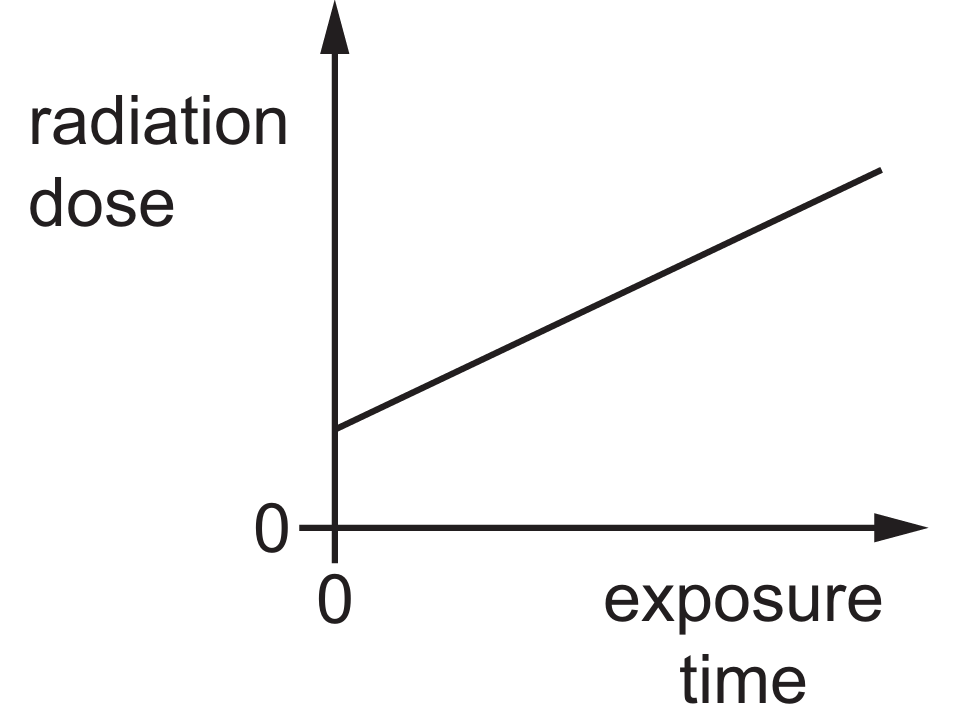
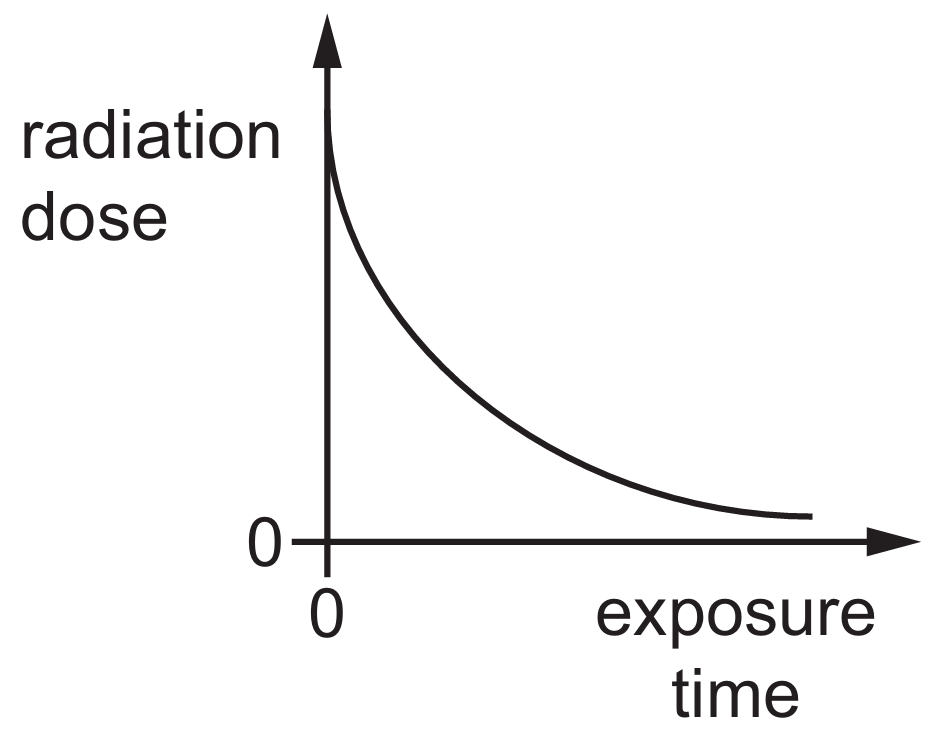
Options B, C, and D are incorrect as they suggest constant, pre-existing, or decreasing doses over time.
▶️ Answer/Explanation
Detailed solution:
To store a source emitting all three types of radiation safely, the shielding must stop the most penetrating type, which is $\gamma$-radiation.
While paper (Option C) stops $\alpha$-particles and a few millimetres of aluminium (Option A) stop $\beta$-particles, they are easily penetrated by $\gamma$-rays.
Lead is a very dense material with a high atomic number, making it highly effective at absorbing ionising radiation.
A thick layer of lead is required to significantly reduce the intensity of $\gamma$-radiation to safe levels.
Therefore, a lead container ensures that $\alpha$, $\beta$, and $\gamma$ emissions are all successfully contained.
This aligns with safety protocols for reducing exposure to ionising nuclear radiations.
