IGCSE Chemistry Mock – Set 1 Paper 2-Updated 2026
IGCSE Chemistry Mock – Set 1 Paper 2 – Updated 2026
Preparing for your CIE IGCSE Chemistry exam can be daunting, but with the right approach, you can achieve your goals with CIE iGCSE Chemistry Mock test.
Ace your CIE iGCSE Chemistry exam! Find exam-style questions, detailed notes, and helpful resources to boost your understanding.
iGCSE Practice Questions, Past Papers, Flashcards and notes available for iGCSE Students at IITian Academy.
The diagrams show containers of gas at the same temperature. All containers have the same size.
Which container contains gas at the highest pressure?
A) 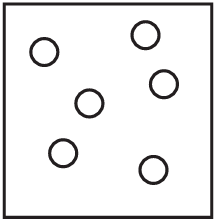
B) 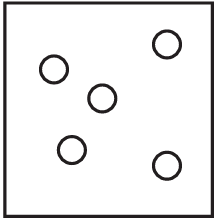
C) 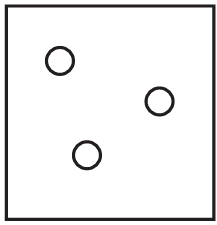
D) 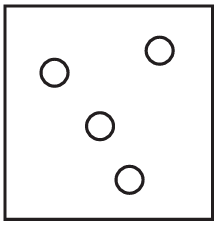
▶️ Answer/Explanation
Ans: A
Pressure is determined by the number of gas particles colliding with the container walls when temperature and volume are constant. Container A has the most particles (5), meaning more collisions per unit time, resulting in higher pressure. The other containers have fewer particles (B has 4, C has 3, D has 2), so they would have lower pressures.
Which statements about hydrogen-oxygen fuel cells are correct?
- They convert chemical energy into electrical energy.
- Hydrogen is reduced in the fuel cells.
- They do not produce any atmospheric pollutants.
A) 1, 2 and 3
B) 1 and 2 only
C) 1 and 3 only
D) 2 and 3 only
▶️ Answer/Explanation
Ans: C
Let’s evaluate each statement:
1. Correct: Fuel cells convert the chemical energy of hydrogen and oxygen directly into electrical energy.
2. Incorrect: In fuel cells, hydrogen is oxidized (loses electrons) at the anode, not reduced. Oxygen is reduced at the cathode.
3. Correct: The only product of a hydrogen-oxygen fuel cell is water, making it pollution-free.
Therefore, only statements 1 and 3 are correct.
Which statements explain why increasing the temperature in a reaction involving gases increases the rate of reaction?
- It increases the collision frequency between the gas particles.
- It lowers the activation energy.
- It increases the kinetic energy of the gas particles.
- It increases the number of gas particles per unit volume.
A) 1 and 3
B) 1 and 4
C) 2 and 3
D) 2 and 4
▶️ Answer/Explanation
Ans: A
Increasing temperature affects reaction rate through two main mechanisms:
1. Increases collision frequency: At higher temperatures, particles move faster, leading to more frequent collisions per second.
3. Increases kinetic energy: More particles have sufficient energy to overcome the activation energy barrier, increasing the proportion of successful collisions.
Statement 2 is incorrect because temperature doesn’t change the activation energy – it just gives more particles enough energy to overcome it. Statement 4 is incorrect because the number of particles per unit volume (concentration) remains constant unless the container size changes.
