IGCSE Chemistry Mock – Set 2 Paper 2-Updated 2026
IGCSE Chemistry Mock – Set 2 Paper 2 – Updated 2026
Preparing for your CIE IGCSE Chemistry exam can be daunting, but with the right approach, you can achieve your goals with CIE iGCSE Chemistry Mock test.
Ace your CIE iGCSE Chemistry exam! Find exam-style questions, detailed notes, and helpful resources to boost your understanding.
iGCSE Practice Questions, Past Papers, Flashcards and notes available for iGCSE Students at IITian Academy.
A cooling curve for a substance is shown.
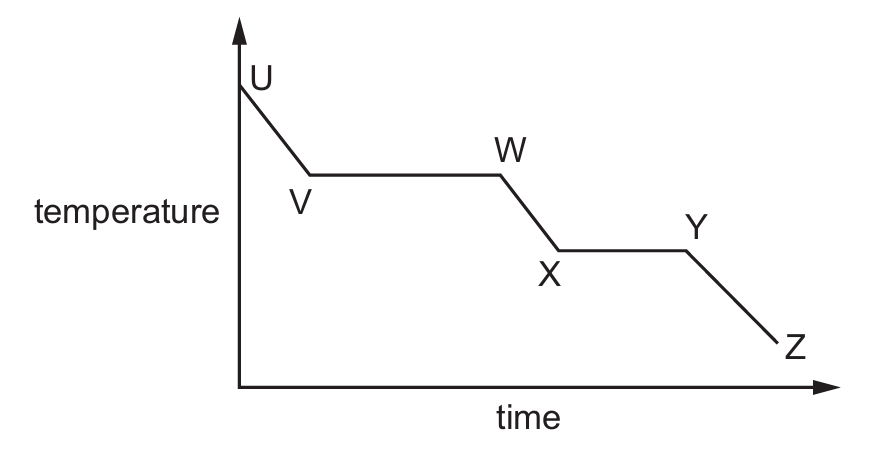
Which statement is correct?
A) Between U and V, the substance is condensing.
B) Between V and W, heat is being absorbed from the surroundings.
C) Between W and X, the particles are close together and randomly arranged.
D) Between Y and Z, the substance is changing from a liquid to a solid.
▶️ Answer/Explanation
Ans: C
On a cooling curve, the horizontal sections represent phase changes where temperature remains constant. Between W and X, the substance is in the liquid state where particles are close together but randomly arranged. Option A is incorrect because U-V shows cooling of gas, not condensation. B is incorrect because V-W shows gas-liquid transition where heat is being released, not absorbed. D is incorrect because Y-Z shows solid cooling, not liquid-solid transition.
Lithium is in Group I of the Periodic Table. Nitrogen is in Group V of the Periodic Table.
Lithium reacts with nitrogen to form the ionic compound lithium nitride, Li3N.
What happens to the electrons when lithium atoms and nitrogen atoms form ions?
| lithium | nitrogen | |
|---|---|---|
| A) | each lithium atom loses one electron to form an Li+ ion | each nitrogen atom gains three electrons to form an N3- ion |
| B) | each lithium atom loses one electron to form an Li+ ion | each nitrogen atom gains five electrons to form an N5- ion |
| C) | each lithium atom gains one electron to form an Li– ion | each nitrogen atom loses three electrons to form an N3+ ion |
| D) | each lithium atom gains one electron to form an Li– ion | each nitrogen atom loses five electrons to form an N5+ ion |
▶️ Answer/Explanation
Ans: A
Lithium is in Group I, so it has 1 valence electron. To achieve stability, it loses this electron to form Li+ ion.
Nitrogen is in Group V with 5 valence electrons. It needs 3 more electrons to complete its octet, so it gains 3 electrons to form N3- ion.
The formula Li3N shows that 3 Li+ ions balance 1 N3- ion, confirming option A is correct.
The value of the Avogadro constant is \(6.02 \times 10^{23}\).
What is the total number of atoms in 2.00 mol of ammonia gas?
A) \(1.20 \times 10^{24}\)
B) \(2.41 \times 10^{24}\)
C) \(4.82 \times 10^{24}\)
D) \(2.89 \times 10^{25}\)
▶️ Answer/Explanation
Ans: C
1. First, determine the number of molecules in 2.00 mol of ammonia:
\(2.00 \text{ mol} \times 6.02 \times 10^{23} \text{ molecules/mol} = 1.204 \times 10^{24} \text{ molecules}\)
2. Each ammonia molecule (NH3) contains 4 atoms (1 N + 3 H).
3. Total number of atoms:
\(1.204 \times 10^{24} \text{ molecules} \times 4 \text{ atoms/molecule} = 4.816 \times 10^{24} \text{ atoms}\)
4. This matches option C (\(4.82 \times 10^{24}\)) when rounded to 3 significant figures.
