IGCSE Chemistry Mock – Set 2 Paper 3-Updated 2026
IGCSE Chemistry Mock – Set 2 Paper 3 – Updated 2026
Preparing for your CIE IGCSE Chemistry exam can be daunting, but with the right approach, you can achieve your goals with CIE iGCSE Chemistry Mock test.
Ace your CIE iGCSE Chemistry exam! Find exam-style questions, detailed notes, and helpful resources to boost your understanding.
iGCSE Practice Questions, Past Papers, Flashcards and notes available for iGCSE Students at IITian Academy.
Oxygen, water and ethene have simple molecular structures.
(a) (i) State the percentage of oxygen in clean, dry air.
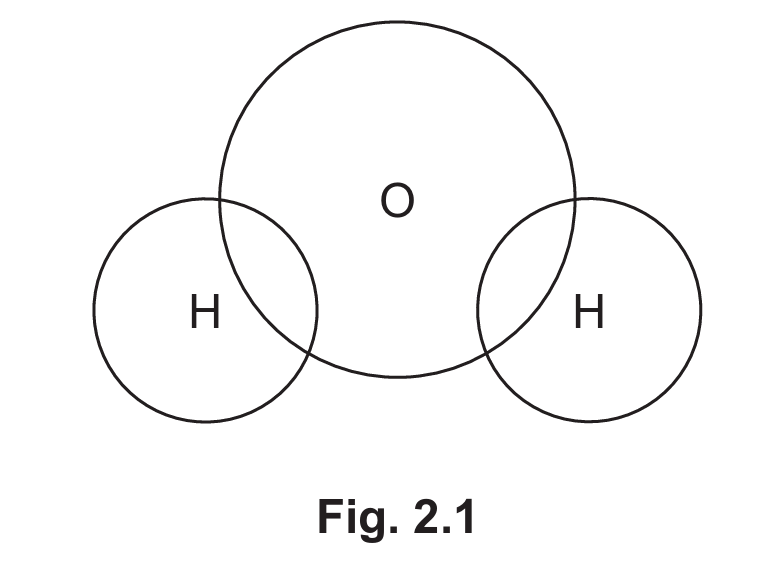
(ii) Complete Fig. 2.1 to show the dot-and-cross diagram for a molecule of water. Show outer shell electrons only.
(iii) Ethene is a small molecule used to make polymers. State the name of the polymer formed from ethene.
(iv) Complete this sentence about polymers. Polymers are large molecules built up from many smaller molecules called ________.
(b) Potassium chloride is an ionic compound.
(i) State two physical properties of ionic compounds.
(ii) Choose the correct statement that describes ionic bonding.
□ It is a weak electrostatic attraction between anions and cations.
□ It is a weak electrostatic attraction between cations.
□ It is a strong electrostatic attraction between anions.
□ It is a strong electrostatic attraction between cations and anions.
▶️ Answer/Explanation
(a) (i) 21%
The atmosphere contains approximately 21% oxygen by volume in clean, dry air.
(ii) The correct dot-and-cross diagram for water should show:
– Two hydrogen atoms each sharing one electron with oxygen (single covalent bonds)
– Oxygen with two lone pairs (four non-bonding electrons)
– No extra electrons on hydrogen atoms
(iii) poly(ethene)
Ethene monomers (C₂H₄) polymerize to form the addition polymer poly(ethene), commonly known as polyethylene.
(iv) monomers
Polymers are formed by linking together many smaller monomer units through chemical reactions.
(b) (i) Any two of:
– High melting points
– Conduct electricity when molten or in aqueous solution
– Soluble in water
– Form crystalline structures
Ionic compounds have strong electrostatic forces between ions, leading to high melting points. Their ions become mobile when molten or dissolved, allowing conductivity.
(ii) The correct statement is: “It is a strong electrostatic attraction between cations and anions.”
Ionic bonding involves the strong electrostatic attraction between positively charged cations and negatively charged anions, formed by electron transfer.
