IGCSE Chemistry Mock – Set 2 Paper 4-Updated 2026
IGCSE Chemistry Mock – Set 2 Paper 4 – Updated 2026
Preparing for your CIE IGCSE Chemistry exam can be daunting, but with the right approach, you can achieve your goals with CIE iGCSE Chemistry Mock test.
Ace your CIE iGCSE Chemistry exam! Find exam-style questions, detailed notes, and helpful resources to boost your understanding.
iGCSE Practice Questions, Past Papers, Flashcards and notes available for iGCSE Students at IITian Academy.
This question is about compounds of tin.
(a) Tin(IV) oxide has the formula SnO₂. The relative formula mass, \( M_r \) of SnO₂ is 151. Calculate the percentage by mass of tin in SnO₂.
(b) SnO₂ is an amphoteric oxide. SnO₂ reacts with aqueous sodium hydroxide, NaOH, to form a sodium salt and water only. The sodium salt contains a negative ion with the formula SnO₃²⁻.
(i) State the meaning of the term amphoteric.
(ii) Write the symbol equation for the reaction between SnO₂ and NaOH.
(c) Tin is a metal that forms both covalent and ionic compounds. Suggest why this is unusual for a metal.
(d) (i) Tin(IV) chloride, SnCl₄ is covalently bonded. A tin atom has four electrons in its outer shell. Complete the dot-and-cross diagram in Fig. 3.1 for a molecule of SnCl₄. Show the outer shell electrons only.
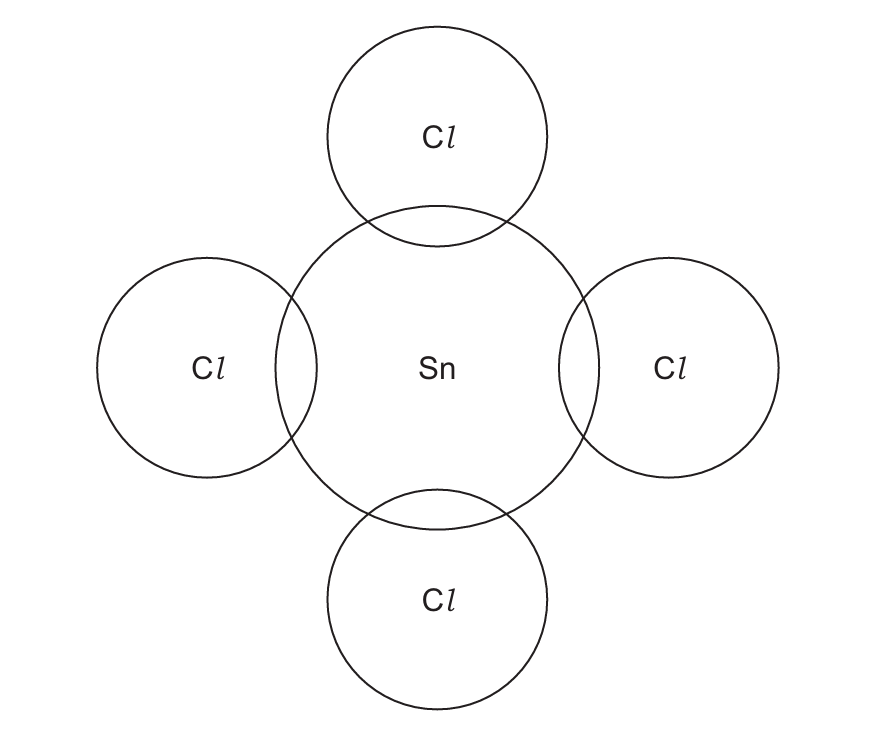
(ii) Tin(II) oxide, SnO, is ionically bonded. The melting points of SnCl₄ and SnO are shown in Table 3.1.
Table 3.1

Explain, in terms of structure and bonding, why SnCl₄ has a much lower melting point than SnO.
(e) Part of the reactivity series is shown.
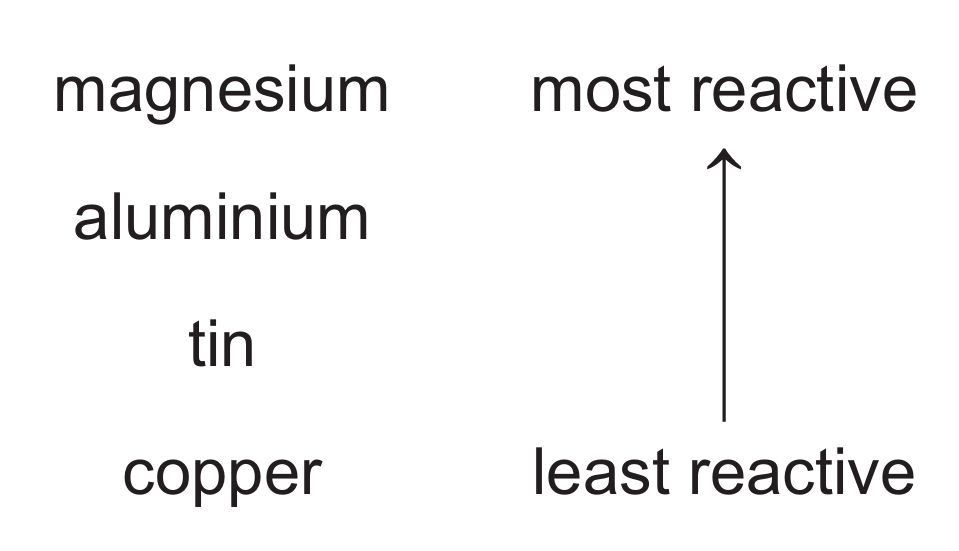
(i) When aluminium foil is added to aqueous tin(II) sulfate, a reaction does not occur even though aluminium is above tin in the reactivity series. Explain why a reaction does not occur.
(ii) An aqueous solution of tin(II) sulfate contains Sn²⁺ ions. Two experiments are carried out.
Experiment 1 Copper is added to aqueous tin(II) sulfate.
Experiment 2 Magnesium is added to aqueous tin(II) sulfate.
Write an ionic equation for any reaction that occurs in each experiment. If no reaction occurs, write ‘no reaction’.
(f) Hydrated tin(II) nitrate, Sn(NO₃)₂•20H₂O, decomposes when it is heated.
(i) State what is meant by the term hydrated.
(ii) Complete the equation for the decomposition of Sn(NO₃)₂•20H₂O.
2Sn(NO₃)₂•20H₂O → ……SnO + ……NO₂ + O₂ + ……H₂O
▶️ Answer/Explanation
(a) 78.8%
Calculation: Relative atomic mass of Sn = 119, O = 16. Percentage of Sn = (119/151) × 100 = 78.8%.
(b)(i) Reacts with acids and with bases to produce a salt and water.
(b)(ii) \( \text{SnO}_2 + 2\text{NaOH} \rightarrow \text{Na}_2\text{SnO}_3 + \text{H}_2\text{O} \)
The reaction shows tin(IV) oxide reacting with sodium hydroxide to form sodium stannate (Na₂SnO₃) and water.
(c) Metals typically form ionic compounds only, while covalent compounds usually contain non-metals only. Tin’s ability to form both is unusual.
(d)(i) The dot-and-cross diagram should show:
- Four single bonds between tin and chlorine atoms
- Three lone pairs on each chlorine atom
- No lone pairs on the tin atom
(d)(ii)
- SnCl₄ has simple molecular structure with weak intermolecular forces between molecules
- SnO has giant ionic structure with strong electrostatic forces between ions
- Much more energy is needed to overcome the strong ionic bonds in SnO than the weak intermolecular forces in SnCl₄
(e)(i) Aluminium forms an unreactive coating of aluminium oxide that prevents reaction.
(e)(ii)
- Experiment 1: no reaction (copper is below tin in reactivity series)
- Experiment 2: \( \text{Mg} + \text{Sn}^{2+} \rightarrow \text{Mg}^{2+} + \text{Sn} \)
(f)(i) A substance that is chemically combined with water or contains water of crystallization.
(f)(ii) \( 2\text{Sn(NO}_3\text{)}_2•20\text{H}_2\text{O} \rightarrow 2\text{SnO} + 4\text{NO}_2 + \text{O}_2 + 40\text{H}_2\text{O} \)
Balancing: For every 2 formula units of the hydrated salt, we get 2 SnO, 4 NO₂ (from 4 nitrate groups), 1 O₂ (remaining oxygen), and 40 H₂O (from 20 × 2 water molecules).
