IGCSE Chemistry Mock – Set 3 Paper 3-Updated 2026
IGCSE Chemistry Mock – Set 3 Paper 3 – Updated 2026
Preparing for your CIE IGCSE Chemistry exam can be daunting, but with the right approach, you can achieve your goals with CIE iGCSE Chemistry Mock test.
Ace your CIE iGCSE Chemistry exam! Find exam-style questions, detailed notes, and helpful resources to boost your understanding.
iGCSE Practice Questions, Past Papers, Flashcards and notes available for iGCSE Students at IITian Academy.
(a) Table 5.1 shows some properties of five halogens.
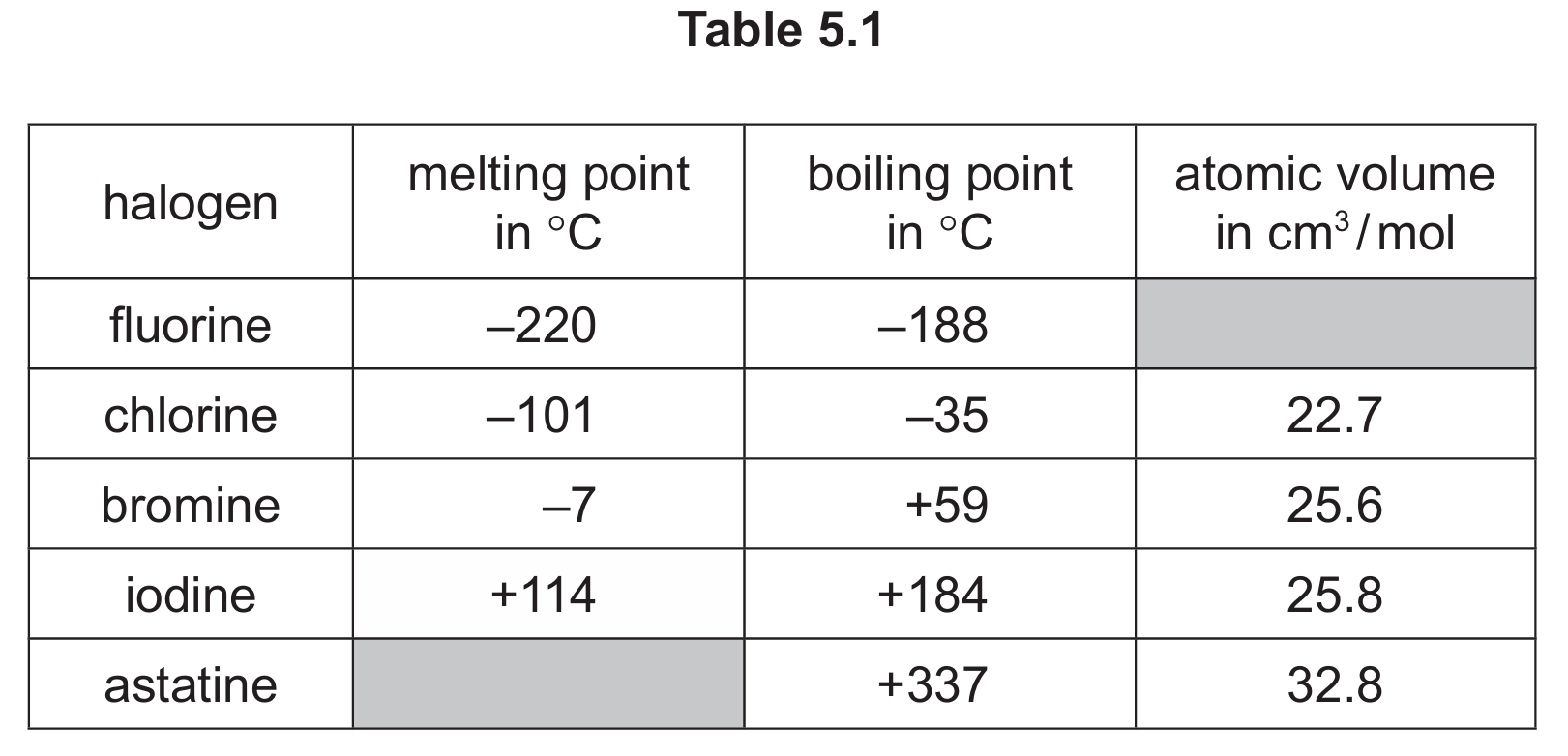
Use the information in Table 5.1 to predict:
(i) the melting point of astatine
(ii) the atomic volume of fluorine
(iii) the physical state of fluorine at −240°C. Give a reason for your answer.
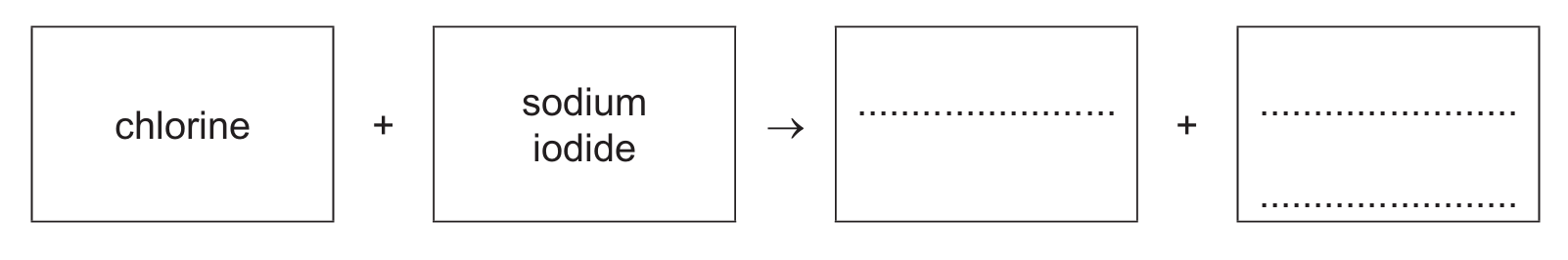
(b) Aqueous chlorine reacts with aqueous sodium iodide.
(i) Complete the word equation for this reaction.
(ii) Explain why aqueous bromine does not react with aqueous sodium chloride.
(c) Fluorine reacts with water to produce hydrogen fluoride and oxygen.
Complete the symbol equation for this reaction.
\[ 2F_2 + \ldots H_2O \rightarrow 4HF + \ldots \]
(d) Name an anhydrous compound used to test for water. State the colour of the compound after water is added.
▶️ Answer/Explanation
(a)(i) Between 116°C and 335°C (inclusive)
Looking at the trend in melting points down Group 17 (halogens), we see they increase: F (-220°C), Cl (-101°C), Br (-7°C), I (114°C). Astatine, being below iodine, would have a higher melting point than iodine but lower than its boiling point of 337°C.
(a)(ii) Less than 22.7 cm³/mol (but not below 1.0)
The atomic volume increases down the group: Cl (22.7), Br (25.6), I (25.8), At (32.8). Fluorine, being above chlorine, would have a smaller atomic volume.
(a)(iii) Solid (1), because −240°C is below fluorine’s melting point of −220°C (1)
At temperatures below its melting point, a substance exists as a solid. Since −240°C is 20 degrees below fluorine’s melting point, it would be solid.
(b)(i) iodine (1) + sodium chloride (1)
This is a displacement reaction where the more reactive chlorine displaces iodine from sodium iodide: Cl₂ + 2NaI → I₂ + 2NaCl
(b)(ii) Bromine is less reactive than chlorine / chlorine is more reactive than bromine (1)
In the reactivity series of halogens, chlorine is above bromine, meaning it can displace bromine but bromine cannot displace chlorine. Therefore, bromine cannot displace chlorine from sodium chloride.
(c) \[ 2F_2 + 2H_2O \rightarrow 4HF + O_2 \] (2 marks: 1 for each correct coefficient)
The reaction shows fluorine reacting with water to produce hydrogen fluoride and oxygen. Balancing the equation requires 2 water molecules to produce 1 oxygen molecule while maintaining the hydrogen and fluorine counts.
(d) Copper(II) sulfate (1) → blue (1) OR Cobalt(II) chloride (1) → red/pink (1)
Anhydrous copper(II) sulfate is white and turns blue when hydrated. Anhydrous cobalt(II) chloride is blue and turns pink when hydrated. Both are common tests for water.
