iGCSE Physics (0625) 2.1.3 Gases and the absolute scale of temperature -Exam Style Questions Paper 1 - New Syllabus
Question

▶️ Answer/Explanation
Detailed solution:
Temperature is a measure of the average kinetic energy of particles, where $E_{k} = \frac{1}{2}mv^{2}$.
As the temperature decreases, the oxygen particles lose kinetic energy and move more slowly ($v$ decreases).
Pressure in a gas is caused by particles colliding with the cylinder walls, exerting a force per unit area ($p = \frac{F}{A}$).
Slower particles collide with the walls less frequently and with less force, leading to a reduction in total pressure.
According to Gay-Lussac’s Law, for a fixed volume, pressure is directly proportional to absolute temperature ($P \propto T$).
Therefore, a decrease in temperature results in a decrease in pressure because the particles move more slowly.
Question
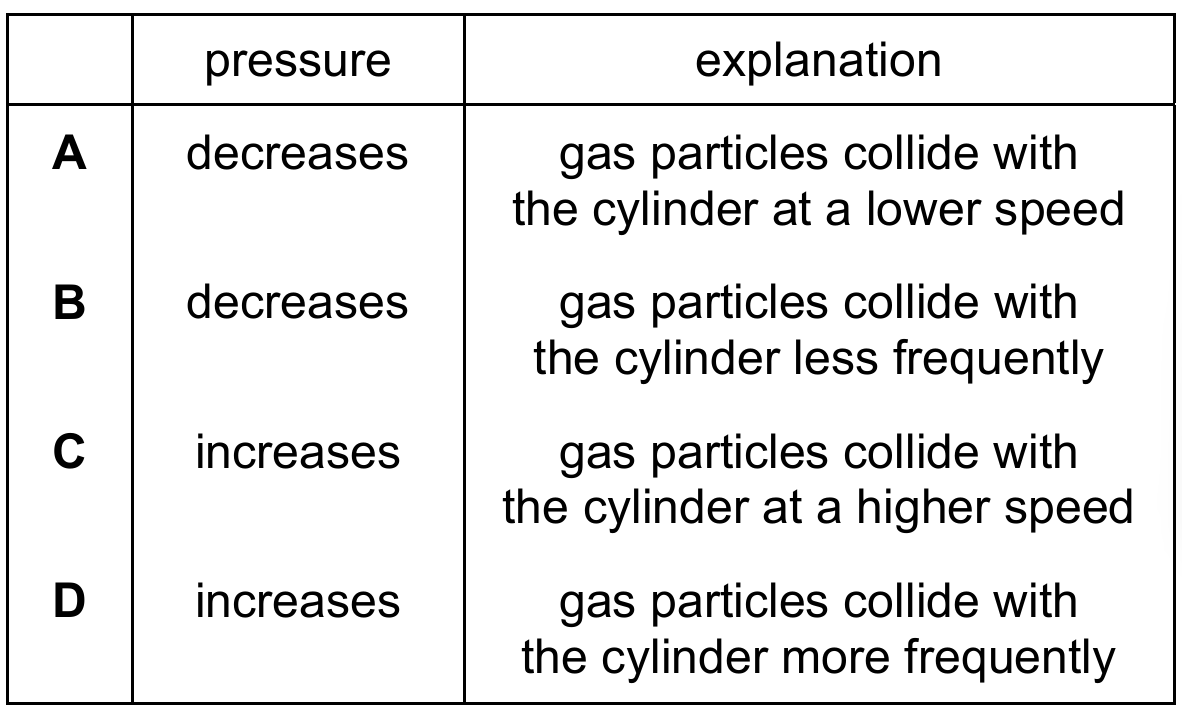
▶️ Answer/Explanation
Detailed solution:
At a constant temperature, the average kinetic energy and speed of gas particles remain unchanged. According to Boyle’s Law, $pV = \text{constant}$, so increasing the volume $V$ results in a decrease in pressure $p$. Microscopically, as the volume increases, the particles are spread over a larger space, leading to an increase in the average distance between them. This results in the gas particles colliding with the cylinder walls less frequently per unit area. Consequently, the total force exerted on the walls decreases, which explains why the pressure decreases. Therefore, Row B provides the correct state and explanation.
Question
What is the temperature of the object in kelvin?
▶️ Answer/Explanation
Detailed solution:
To convert a temperature from the Celsius scale to the Kelvin scale, we use the standard relation: T(in K)=θ(in ∘ C)+273.
Given that the temperature of the object is θ=37 ∘ C, we substitute this value into the conversion formula.
The calculation becomes T=37+273, which equals 310.
Therefore, the temperature in kelvin is 310 K.
This corresponds to option D, which correctly identifies the absolute temperature value.
Note that the kelvin scale starts at absolute zero, which is equivalent to −273 ∘ C.
