CHEMISTRY PAPER WITH SOLUTION
SECTION- A
- (A) Atoms of different elements differ in mass.
- (B) Matter consists of divisible atoms.
- (C) Compounds are formed when atoms of different element combine in a fixed ratio.
- (D) All the atoms of a given element have different properties including mass.
- (E) Chemical reactions involve reorganisation of atoms.
- (B), (D), (E) only
- (A), (B), (D) only
- (C), (D), (E) only
- (B), (D) only
▶️ Answer/Explanation
Fe2O3(s) + 3CO(g) ↔ 2Fe(l) + 3CO2(g)
Using Le-chatelier’s principle, predict which one of the following will not disturb the equilibrium.
- Addition of Fe2O3
- Addition of CO2
- Removal of CO
- Removal of CO2
▶️ Answer/Explanation
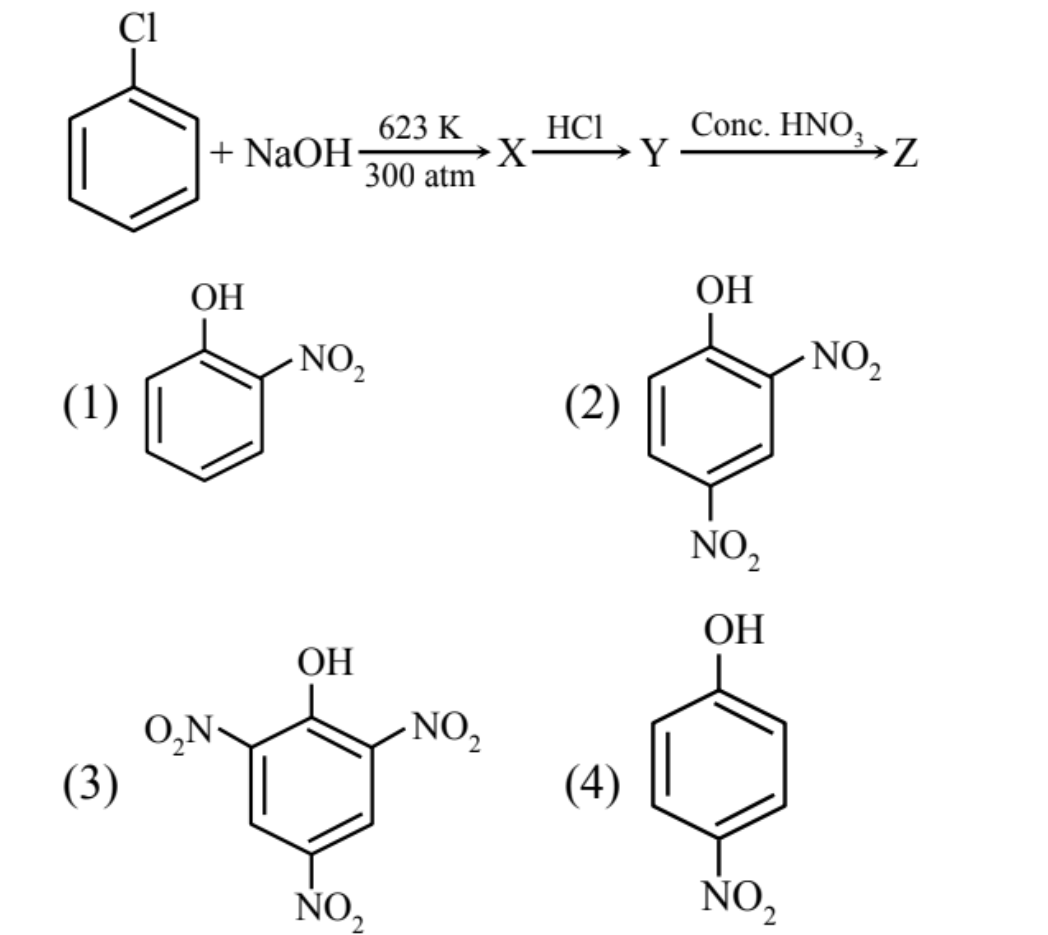
▶️ Answer/Explanation
Y: phenol
Z: 2,4,6-trinitrophenol (picric acid).
The correct compound Z is (3).
Assertion (A): Enthalpy of neutralisation of strong monobasic acid with strong monoacidic base is always –57 kJ mol–1
Reason (R): Enthalpy of neutralisation is the amount of heat liberated when one mole of H+ ions furnished by acid combine with one mole of OH– ions furnished by base to form one mole of water.
In the light of the above statements, choose the correct answer from the options below:
- (A) is true but (R) is false
- Both (A) and (R) are true and (R) is the correct explanation of (A)
- (A) is false but (R) is true
- Both (A) and (R) are true but (R) is not the correct explanation of (A)
▶️ Answer/Explanation
- (A) All are isoelectronic.
- (B) All have the same nuclear charge.
- (C) O2– has the largest ionic radii.
- (D) Mg2+ has the smallest ionic radii.
- (B), (C) and (D) only
- (A), (B), (C), (D)
- (C), (D) only
- (A), (C), (D) only
▶️ Answer/Explanation
(A) H3C–CH2–O–CH2–CH2–CH3
(B) H3C–CH2–CH2–CH2–CH3
(C) CH3–CH2–C(=O)–CH2–CH3
(D) H3C–CH–CH2–CH2–CH3 (with –OH on 2nd C)
The increasing order of boiling point is:
- (A) < (B) < (C) < (D)
- (B) < (A) < (C) < (D)
- (D) < (C) < (A) < (B)
- (B) < (A) < (D) < (C)
▶️ Answer/Explanation
Boiling point: hydrogen bonding > high polarity > low polarity > non-polar.
Statement I: In group 13, the stability of +1 oxidation state increases down the group.
Statement II: The atomic size of gallium is greater than that of aluminium.
Choose the most appropriate answer:
- Statement I is incorrect but Statement II is correct
- Both Statement I and Statement II are correct
- Both Statement I and Statement II are incorrect
- Statement I is correct but Statement II is incorrect
▶️ Answer/Explanation
- 3 and 1
- 5 and 2
- 4 and 1
- 5 and 1
H σ
H—C ≡πσ—C—H
σ
So it has 5 σ bonds and 1 π bond.
Ans. (4)
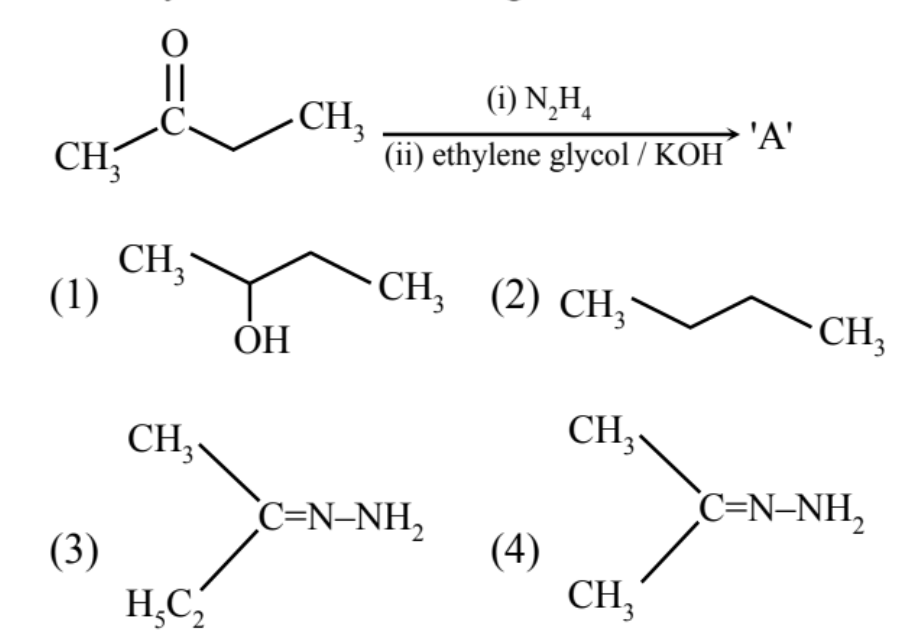
- CH3CH(OH)CH3
- CH3CH2CH3
- CH3CH=N–NH2
- C2H5CH=N–NH2
▶️ Answer/Explanation
- reduction of Mn from +4 to +3
- oxidation of Mn from +3 to +4
- reduction of Mn from +7 to +2
- oxidation of Mn from +2 to +7
▶️ Answer/Explanation
[Atomic number, Cr = 24, Mn = 25, Fe = 26, Co = 27]
- [Co(H2O)6]2+
- [Fe(H2O)6]2+
- [Mn(H2O)6]2+
- [Cr(H2O)6]2+
▶️ Answer/Explanation
| Complex | Number of unpaired e– | μ = √n(n + 2) B.M. |
|---|---|---|
| [Co(H2O)6]2+ | 3 | 3.87 |
| [Fe(H2O)6]2+ | 4 | 4.89 |
| [Mn(H2O)6]2+ | 5 | 5.92 |
| [Cr(H2O)6]2+ | 4 | 4.89 |
Assertion (A): Cis form of alkene is found to be more polar than the trans form.
Reason (R): Dipole moment of trans isomer of 2-butene is zero.
In the light of the above statements, choose the correct answer:
- Both (A) and (R) are true but (R) is NOT the correct explanation of (A)
- (A) is true but (R) is false
- Both (A) and (R) are true and (R) is the correct explanation of (A)
- (A) is false but (R) is true
▶️ Answer/Explanation
Cis (μ > 0) > Trans (μ = 0)
Given below are two statements:
Statement I: Nitration of benzene involves the following step –
H
|
H–O–NO2 ⇌ H2O + NO2+
Statement II: Use of Lewis base promotes the electrophilic substitution of benzene.
In the light of the above statements, choose the most appropriate answer:
- Both Statement I and Statement II are incorrect
- Statement I is correct but Statement II is incorrect
- Both Statement I and Statement II are correct
- Statement I is incorrect but Statement II is correct
▶️ Answer/Explanation
In nitration of benzene, concentrated H2SO4 and HNO3 generate the electrophile NO2+ in steps:
H2SO4 + HNO3 ⇌ HSO4– + H–O–NO2+
HSO4– + H2O + NO2+
Lewis acids—not Lewis bases—promote the formation of electrophiles.
- Cl– < OH– < Br– < CN–
- F– < Br– < I– < NH3
- Br– < F– < H2O < NH3
- H2O < OH– < CN– < NH3
▶️ Answer/Explanation
- Cellulose
- Starch
- Polyvinyl chloride
- Egg albumin
▶️ Answer/Explanation
- Fe
- Mn
- Ti
- Co
▶️ Answer/Explanation
- C11H18O12
- C12H20O12
- C14H20O10
- C12H22O11
▶️ Answer/Explanation
Statement I: Bromination of phenol in solvent with low polarity such as CHCl3 or CS2 requires Lewis acid catalyst.
Statement II: The Lewis acid catalyst polarises the bromine to generate Br+.
In the light of the above statements, choose the correct answer:
- Statement I is true but Statement II is false
- Both Statement I and Statement II are true
- Both Statement I and Statement II are false
- Statement I is false but Statement II is true
▶️ Answer/Explanation
- 65 S cm2 mol–1
- 130 S cm2 mol–1
- 187 S cm2 mol–1
- 260 S cm2 mol–1
▶️ Answer/Explanation
Λ+2A = 73 S cm2mol–1
ΛSolution = 57 + 73 = 130 S cm2mol–1
- 4
- 6
- 3
- 9
▶️ Answer/Explanation
B + O2 → B2O3 (oxidation state +3, y = 3)
x + y = 6 + 3 = 9
SECTION-B
The velocity of electron having mass 9.1×10–31 kg in Bohr’s first orbit of hydrogen atom = ……… × 105 ms–1 (nearest integer)
▶️ Answer/Explanation
For hydrogen, n = 1, Z = 1
V = 2.18 × 106 × 1/1 = 2.18 × 106 ms–1 = 21.8 × 105 ≈ 22 × 105 ms–1 (nearest integer)
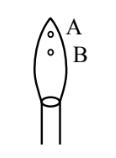 In a borax bead test under hot condition, a metal salt (one from the given) is heated at point B of the flame, resulted in green colour salt bead. The spin-only magnetic moment value of the salt is ……….. BM (Nearest integer)
In a borax bead test under hot condition, a metal salt (one from the given) is heated at point B of the flame, resulted in green colour salt bead. The spin-only magnetic moment value of the salt is ……….. BM (Nearest integer)[Given atomic number of Cu = 29, Ni = 28, Mn = 25, Fe = 26]
▶️ Answer/Explanation
▶️ Answer/Explanation
Δng = –0.5;
ΔH = –321.30 – (1/2 × 300) = –321.30 – 150R
So x = 150.
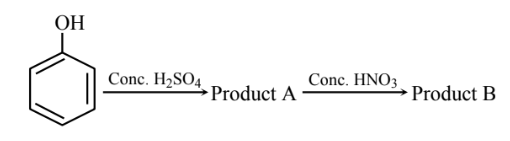
Total sum of oxygen atoms in Product A and Product B are ………
▶️ Answer/Explanation
(Given atomic numbers: Ti : 22, V : 23, Cr : 24, Co : 27)
▶️ Answer/Explanation
No. of unpaired e– in Co3+ (3d6) = 4
μ = √n(n+2) = √24 = 4.89 ≈ 5
| A[M] | B[M] | initial rate of formation of D |
|---|---|---|
| 0.1 | 0.1 | 6.0 × 10–3 |
| 0.3 | 0.2 | 7.2 × 10–2 |
| 0.3 | 0.4 | 2.88 × 10–1 |
| 0.4 | 0.1 | 2.40 × 10–2 |
▶️ Answer/Explanation
[Given: R = 0.083 L bar mol–1 K–1]
Assume complete dissociation of NaCl.
▶️ Answer/Explanation
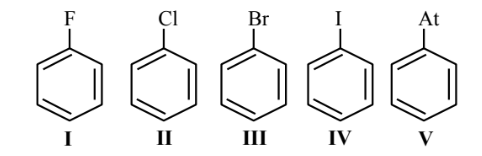
The number of halobenzenes from the following that can be prepared by Sandmeyer’s reaction is ….
▶️ Answer/Explanation
▶️ Answer/Explanation
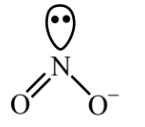
▶️ Answer/Explanation
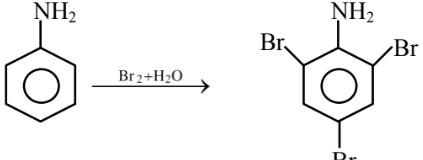 93 g aniline produces 330 g of tribromoaniline.
93 g aniline produces 330 g of tribromoaniline.9.3 g aniline → 33 g tribromoaniline.
% yield = 26.4/33 × 100 = 80%
