The diagram shows the particles in the atom of an element.
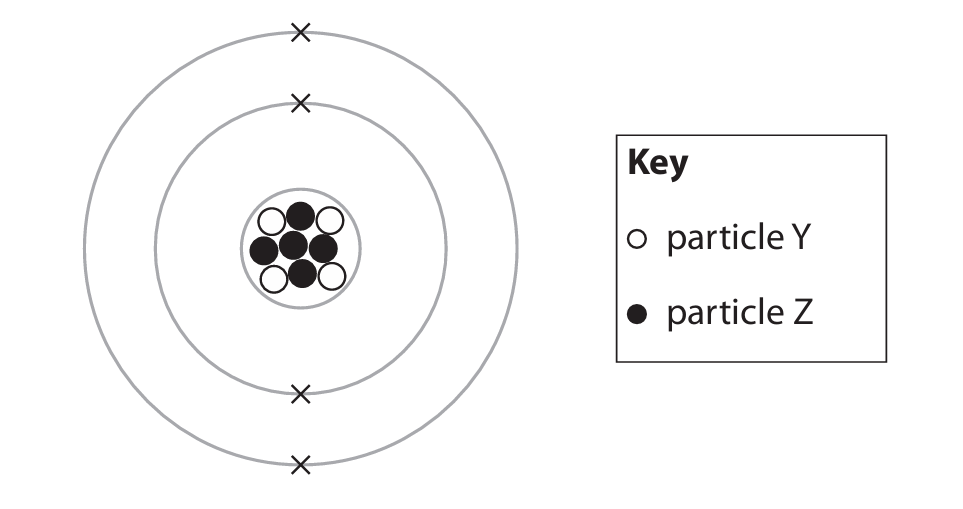
(a) Particle Y is a proton. What is particle Z?
A) an electron
B) a molecule
C) a neutron
D) a nucleus
(b) Which of these has the smallest mass?
A) an electron
B) a neutron
C) a nucleus
D) a proton
(c) What is the mass number of this atom?
A) 4
B) 5
C) 9
D) 13
(d) What is the atomic number of this atom?
A) 4
B) 5
C) 9
D) 13
(e) (i) Identify the element that contains this atom.
(e) (ii) State what is formed when this atom loses its outer shell electrons.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(d): The Periodic Table — part (e)(i)
• 1(f): Ionic bonding — part (e)(ii)
▶️ Answer/Explanation
(a) C (a neutron)
The only correct answer is C because the nucleus contains protons and neutrons. Protons are identified as the white dots.
A is not correct because electrons occur in the shells.
B is not correct because a molecule is not a particle found in the nucleus.
D is not correct because the nucleus contains protons and neutrons.
(b) A (an electron)
The only correct answer is A because electrons have a relative mass of \( \frac{1}{1836} \) compared to a proton or a neutron.
B is not correct because a neutron has a relative mass of 1.
C is not correct because the nucleus contains 4 protons and 5 neutrons.
D is not correct because a proton has a relative mass of 1.
(c) C (9)
The only correct answer is C because the mass number is the sum of the protons and neutrons (\(4 + 5 = 9\)).
A is not correct because the atomic number is 4.
B is not correct because 5 is the number of neutrons.
D is not correct because 13 is the total number of protons, neutrons and electrons.
(d) A (4)
The only correct answer is A because the atomic number is equal to the number of protons which is 4.
B is not correct because 5 is the number of neutrons.
C is not correct because 9 is the total number of particles in the nucleus.
D is not correct because 13 is the total number of protons, neutrons and electrons.
(e)(i) beryllium / Be
(e)(ii) (positive) ion
ALLOW ecf from the element given in (e)(i).
ACCEPT any positive beryllium ion (or other ecf ion).
REJECT any negative ion.
This question is about states of matter.
(a) The diagram shows how the particles of a substance are arranged in two different states.
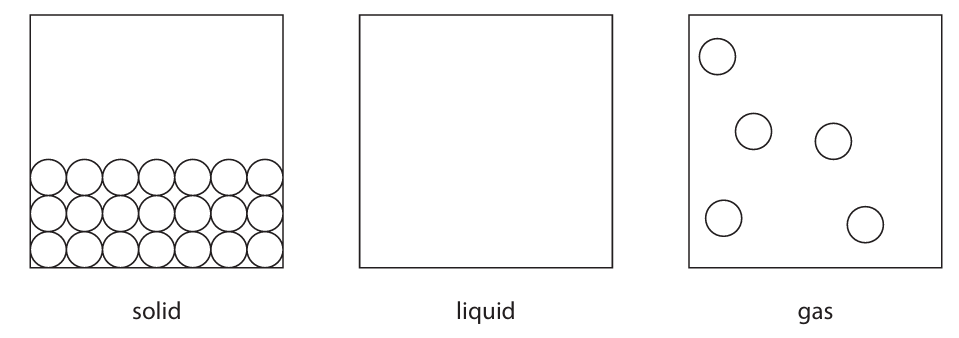
(i) Complete the diagram to show how particles are arranged in the liquid state.
(ii) Identify the state of matter in which the particles have the most energy.
(b) The state symbols \( (s) \), \( (l) \), \( (g) \) and \( (aq) \) are often used in chemistry. The table shows some physical changes. Complete the table by giving the state symbol before and after each change.
| Physical change | Before change | After change |
|---|---|---|
| water evaporates | ||
| crystals of iodine sublime | ||
| ice melts |
(c) Explain why hot water evaporates more quickly than cold water.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
▶️ Answer/Explanation
(a)(i) The diagram for the liquid state should show particles that are close together, filling from the bottom of the box, with some particles touching. The arrangement should be irregular, not in a regular lattice.
(a)(ii) Gas
A is not the answer as particles in a solid have the least energy.
B is not the answer as particles in a liquid have more energy than a solid but less than a gas.
(b)
• Water evaporates: Before = \( (l) \), After = \( (g) \)
• Crystals of iodine sublime: Before = \( (s) \), After = \( (g) \)
• Ice melts: Before = \( (s) \), After = \( (l) \)
(c)
• Particles/molecules in hot water have more (kinetic) energy (1).
• Therefore, they can more easily overcome/break the (intermolecular) forces between water molecules and escape from the liquid surface (1).
Allow: “move faster”.
The Group 7 elements are called halogens. Halogens form compounds called halides.
Three of the halogens are represented by the formulae \( X_2 \), \( Y_2 \) and \( Z_2 \).
Solutions of these halogens are added separately to solutions of sodium halides, NaX, NaY and NaZ.
The table shows whether or not a reaction occurs.
| \( X_2 \) | \( Y_2 \) | \( Z_2 \) | |
|---|---|---|---|
| NaX | no | yes | yes |
| NaY | no | no | yes |
| NaZ | no | no | no |
(a) Use the information in the table to deduce the order of reactivity of the halogens \( X_2 \), \( Y_2 \) and \( Z_2 \).
(b) An aqueous solution of halogen \( Y_2 \) is orange. This solution is decolourised when it reacts with an alkene. Deduce the identity of halogen \( Y_2 \).
(c) (i) The table shows some physical properties of the halogens. Complete the table by predicting a boiling point for chlorine, the state of fluorine at room temperature and the colour of astatine.
| Halogen | Boiling point in °C | State at room temperature | Colour |
|---|---|---|---|
| fluorine | –188 | yellow | |
| chlorine | gas | green | |
| bromine | 59 | liquid | red-brown |
| iodine | sublimes | solid | grey |
| astatine | 337 | solid |
(ii) Why do the halogens have similar chemical properties?
A they are non-metals
B they are molecules
C they have the same number of outer shell electrons
D they are in the same period of the Periodic Table
(d) A teacher uses this apparatus to demonstrate the reaction between chlorine gas and iron wool. The teacher does the reaction in a fume cupboard.
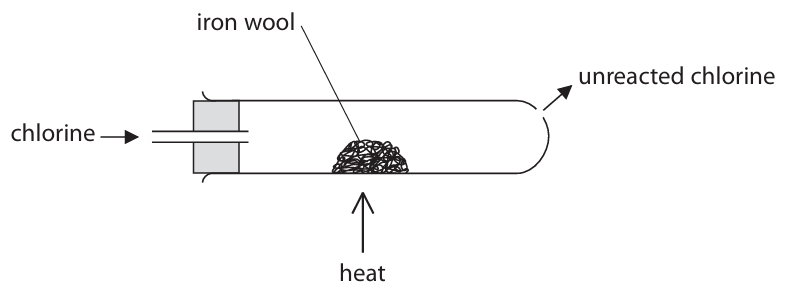
(i) Suggest why the teacher does the reaction in a fume cupboard.
(ii) The product of the reaction between iron and chlorine is iron(III) chloride. The ions in iron(III) chloride are \( \text{Fe}^{3+} \) and \( \text{Cl}^- \). Use this information to give the chemical equation for this reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(d) Reactivity series: part (a) displacement reactions
• 1(d) The Periodic Table: part (c)(ii)
• 4(d) Alkenes: part (b) reaction with bromine
• 2(h) Chemical tests: part (b) test for alkenes
• 2(e) Extraction and uses of metals: part (d) reaction of chlorine with iron
• 1(e) Chemical formulae, equations and calculations: part (d)(ii) writing balanced equations
▶️ Answer/Explanation
(a)
most reactive: \( Z_2 \)
least reactive: \( X_2 \)
Order: \( Z_2 > Y_2 > X_2 \)
Explanation: A more reactive halogen can displace a less reactive halogen from its halide solution. \( Z_2 \) reacts with NaX and NaY, so is more reactive than \( X_2 \) and \( Y_2 \). \( Y_2 \) reacts with NaX but not NaZ, so \( Y_2 \) is more reactive than \( X_2 \) but less reactive than \( Z_2 \).
(b) bromine / \( Br_2 \)
Explanation: Bromine solution is orange and is decolourised when it reacts with an alkene in an addition reaction (test for unsaturation).
(c)(i)
Fluorine state at room temperature: gas / vapour
Chlorine boiling point: between -150°C and 10°C (e.g., -34°C)
Astatine colour: dark grey / black
(c)(ii) C
Explanation: Halogens are in Group 7 and have similar chemical properties because they all have 7 electrons in their outer shell.
(d)(i) Chlorine is toxic / poisonous.
Explanation: The reaction should be done in a fume cupboard to prevent inhalation of chlorine gas.
(d)(ii)
\( 2\text{Fe} + 3\text{Cl}_2 \rightarrow 2\text{FeCl}_3 \)
Explanation: The balanced equation shows the formation of iron(III) chloride from its elements.
This question is about ammonium chloride.
(a) Give the formula of the ammonium ion.
(b) Describe a test to show that ammonium chloride contains ammonium ions.
(c) The equation shows the thermal decomposition of ammonium chloride.
\( \text{NH}_4\text{Cl}(s) \rightleftharpoons \text{NH}_3(g) + \text{HCl}(g) \)
State what the \( \rightleftharpoons \) symbol indicates about this reaction.
(d) The diagram shows the formation of ammonium chloride in a glass tube.
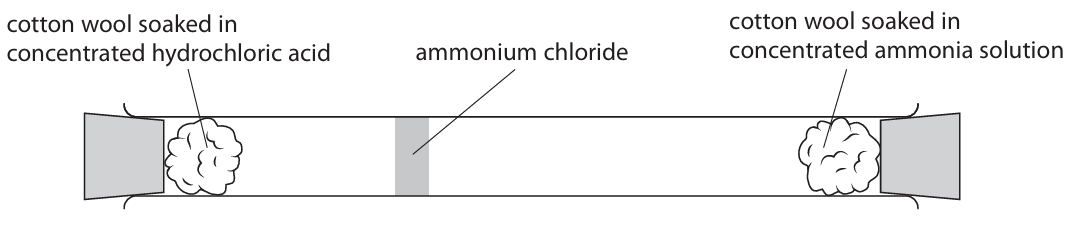
(i) Explain how the mean speed of ammonia molecules compares with the mean speed of hydrogen chloride molecules.
(ii) Gas particles travel very quickly. Give two reasons why it takes several minutes for the ammonium chloride to form.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(a): States of matter — parts (d)(i), (d)(ii)
• 3(c): Reversible reactions and equilibria — part (c)
• 1(f): Ionic bonding — part (a)
▶️ Answer/Explanation
(a) \( \text{NH}_4^+ \) or \( \text{NH}_4 \) (ALLOW both)
(b) M1: Add sodium hydroxide solution (and warm).
M2: Test the gas with damp red litmus paper or expose the gas to concentrated hydrochloric acid.
M3: (If using litmus) turns blue or (if using HCl) white smoke (ammonium chloride) is produced.
(If universal indicator is used, allow blue/purple for M3. M3 is dependent on correct method in M2. If sodium hydroxide is not added, max = 1 mark)
(c) The reaction is reversible. (ACCEPT: reaction goes both ways / both forwards and backwards reactions occur. IGNORE references to equilibrium.)
(d)(i)
M1: (Molecules/Particles of) ammonia move/diffuse faster.
M2: Because the ammonium chloride forms nearer to the HCl end or because the ammonia has travelled further in the same time.
(IGNORE references to masses/sizes of particles.)
(d)(ii)
Any two from:
M1: (Gas particles) move in random directions / do not travel in straight lines.
M2: (Gas particles) collide with air/other particles.
M3: (Gas particles) collide with the walls/sides of the tube.
(ALLOW: air/other particles slow them down. IGNORE any references to rate of reaction/collisions.)
A teacher uses the reaction between phosphorus and oxygen to calculate the percentage of oxygen in air.
She uses this apparatus and excess phosphorus.
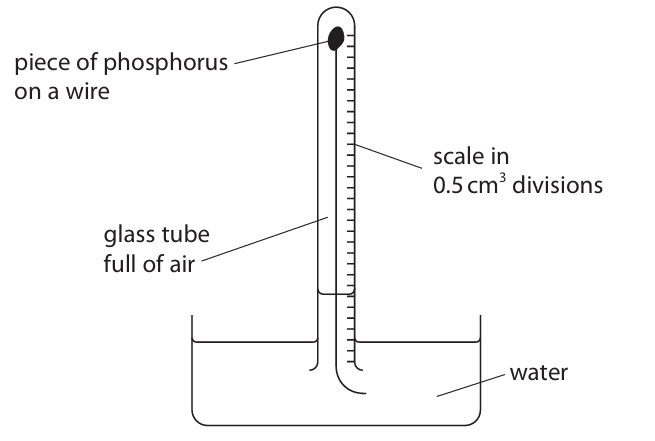
The volume of gas in the tube decreases as the phosphorus reacts with oxygen.
The teacher measures the volume of gas in the tube at one-minute intervals.
The table shows the teacher’s results.
| Time in minutes | Volume of gas in tube in cm3 |
|---|---|
| 0 | 48.5 |
| 1 | 41.0 |
| 2 | 38.0 |
| 4 | 37.5 |
| 5 | 37.0 |
| 6 | 37.0 |
| 7 | 37.0 |
(a) State how the results show that all the oxygen has reacted.
(b) Give one change to this experiment that would make the results more accurate.
(c) Use the results to calculate the percentage of oxygen in air. Give your answer to one decimal place.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2.10: Understand how to determine the percentage by volume of oxygen in air using experiments involving the reactions of metals (e.g. iron) and non-metals (e.g. phosphorus) with air.
• 2.14: Practical: determine the approximate percentage by volume of oxygen in air using a metal or a non-metal.
▶️ Answer/Explanation
(a) The results show that all the oxygen has reacted because the volume of gas in the tube becomes constant/stops decreasing (at 37.0 cm3 from 5 minutes onwards).
(b) Use a glass tube or scale with smaller divisions (e.g., 0.1 cm3 divisions) to measure the volume more precisely.
(c)
Step 1: Find the volume of oxygen used.
Initial volume = 48.5 cm3
Final constant volume = 37.0 cm3
Volume of oxygen = \( 48.5 – 37.0 = 11.5 \) cm3
Step 2: Calculate the percentage.
Percentage of oxygen = \( \frac{11.5}{48.5} \times 100 \)
Step 3: Compute and round.
\( \frac{11.5}{48.5} \approx 0.2371 \)
\( 0.2371 \times 100 = 23.71\% \)
Rounded to one decimal place: 23.7%
| Question number | Answer | Notes | Marks |
|---|---|---|---|
| (a) | Results are the same at the end | ALLOW there is a constant volume in the tube ALLOW the volume of gas stops decreasing ALLOW no change after 4 / 5 minutes | 1 |
| (b) | (use a glass tube / scale with) smaller divisions. | ALLOW use a glass tube / scale with 0.1cm³ divisions ALLOW use a smaller scale | 1 |
| (c) | M1 Volume of oxygen = 11.5(cm³) M2 (11.5 ÷ 48.5) x 100 M3 23.7% | Correct answer to 1 dp with or without working scores 3 ALLOW ecf from M1 M3 must be to 1dp | 3 |
The reaction between magnesium and copper(II) sulfate solution is exothermic. This apparatus is used to measure the temperature increase when excess magnesium is added to 100 cm3 of copper(II) sulfate solution.
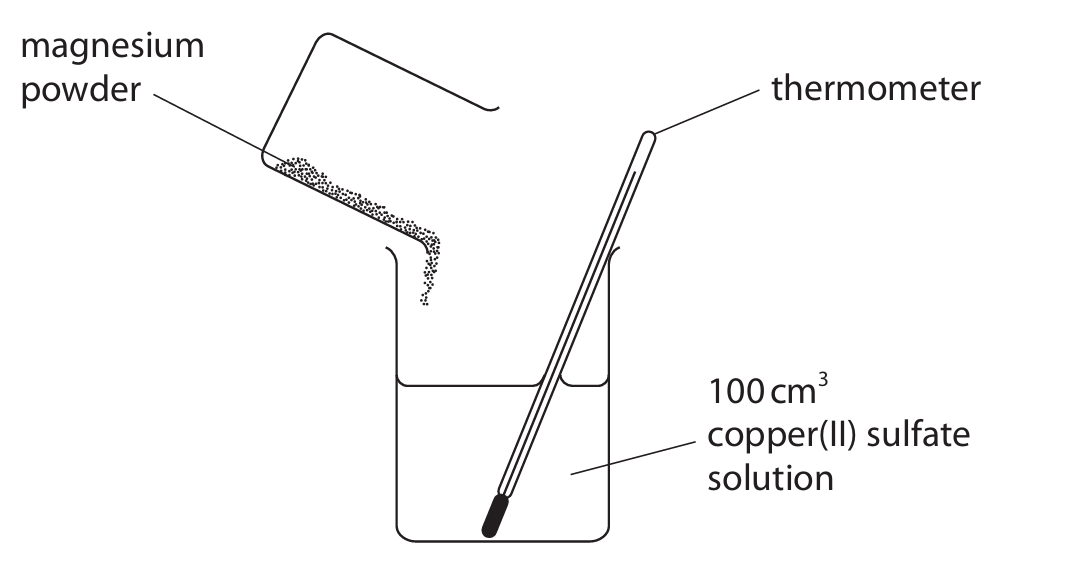
(a)(i) State why a reaction occurs when magnesium is added to copper(II) sulfate solution.
(ii) Complete the word equation for this reaction.
magnesium + copper(II) sulfate → ______ + ______
(b) The temperature at the start of the reaction is 20.2°C. The maximum temperature recorded is 56.3°C.
(i) Calculate the heat energy change, in joules, for the reaction.
(ii) Explain why it is better to use a polystyrene cup rather than a glass beaker in this experiment.
(c) The reaction between zinc and copper(II) sulfate solution is also exothermic.
(i) A mass of 0.500 g of zinc is reacted with an excess of copper(II) sulfate solution. The heat energy change is 1.67 kJ. Calculate the molar enthalpy change, \(\Delta H\), in kJ/mol. Include a sign in your answer. Give your answer to three significant figures.
(ii) The ionic equation for the reaction between zinc and copper(II) sulfate is:
\[\text{Zn(s)} + \text{Cu}^{2+}\text{(aq)} \rightarrow \text{Zn}^{2+}\text{(aq)} + \text{Cu(s)}\]
Explain why this is a redox reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(e): Chemical formulae, equations and calculations — Word equations, mole calculations
• 3(a): Energetics — Calorimetry, \(Q = mc\Delta T\), molar enthalpy change
▶️ Answers/Explanations
Question 6(a)(i)
Magnesium is more reactive than copper / Magnesium is higher than copper in the reactivity series.
This allows magnesium to displace copper from its compound.
Question 6(a)(ii)
magnesium + copper(II) sulfate → magnesium sulfate + copper
Both products required in either order. “Copper” not “copper(II)”.
Question 6(b)(i)
1. Calculate \(\Delta T\): \(\Delta T = 56.3^\circ \text{C} – 20.2^\circ \text{C} = 36.1^\circ \text{C}\)
2. Mass of solution: \(100\ \text{cm}^3 \times 1.00\ \text{g/cm}^3 = 100\ \text{g}\)
3. Use \(Q = mc\Delta T\): \(Q = 100\ \text{g} \times 4.2\ \text{J g}^{-1}\text{°C}^{-1} \times 36.1^\circ \text{C} = 15162\ \text{J}\)
Answer: \(\mathbf{15162\ J}\) (or \(\mathbf{15.2\ kJ}\))
Question 6(b)(ii)
Polystyrene is a thermal insulator / poor conductor of heat.
• This reduces heat loss to the surroundings.
• Therefore, the measured temperature change is more accurate / closer to the true value.
Question (c)(i)
1. Moles of zinc: \(n = \frac{0.500\ \text{g}}{65.4\ \text{g/mol}} = 0.007645\ \text{mol}\) (using \(M_r = 65.4\))
2. Molar enthalpy change: \(\Delta H = \frac{1.67\ \text{kJ}}{0.007645\ \text{mol}} = 218.4\ \text{kJ/mol}\)
3. Sign: Reaction is exothermic, so \(\Delta H\) is negative.
Answer (to 3 s.f.): \(\mathbf{\Delta H = -218\ \text{kJ/mol}}\)
Note: Using \(M_r = 65\) gives \(-217\ \text{kJ/mol}\) as per mark scheme.
Question (c)(ii)
This is a redox reaction because:
• Oxidation occurs: Zinc atoms lose electrons to form Zn²⁺ ions (\(\text{Zn} \rightarrow \text{Zn}^{2+} + 2\text{e}^-\)).
• Reduction occurs: Copper(II) ions gain electrons to form copper atoms (\(\text{Cu}^{2+} + 2\text{e}^- \rightarrow \text{Cu}\)).
• Both oxidation and reduction happen simultaneously.
A student investigates the reaction between sodium hydroxide solution and hydrochloric acid.
He uses this method.
Step 1: add 50 cm3 of dilute hydrochloric acid to a conical flask
Step 2: add a 5 cm3 portion of sodium hydroxide solution to the conical flask
Step 3: test the pH of the mixture using both universal indicator paper and a pH meter
The student repeats step 2 and step 3 until a total of 50 cm3 of sodium hydroxide solution has been added.
(a)(i) State the piece of apparatus that should be used to measure 50 cm3 of hydrochloric acid.
(ii) Name the type of reaction that occurs between hydrochloric acid and sodium hydroxide.
(b) Graph 1 shows how the pH of the mixture changes as the sodium hydroxide solution is added.
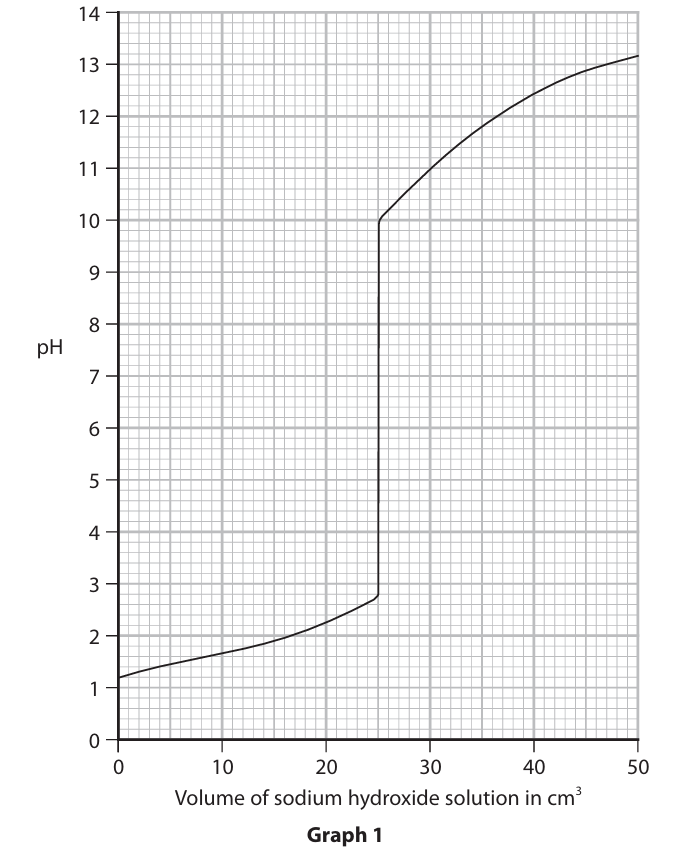
(i) Determine the pH after 40 cm3 of sodium hydroxide solution has been added.
(ii) Suggest the colour of the universal indicator paper when these volumes of sodium hydroxide solution have been added.
(iii) Give the formula of the ion that causes sodium hydroxide to be alkaline.
(c) Another student investigates how the temperature changes when the sodium hydroxide solution is added to the hydrochloric acid. The hydrochloric acid and the sodium hydroxide solution are at the same temperature at the start of the investigation. The student records the temperature of the mixture after adding each 5 cm3 portion of sodium hydroxide solution.
Graph 2 shows her results.
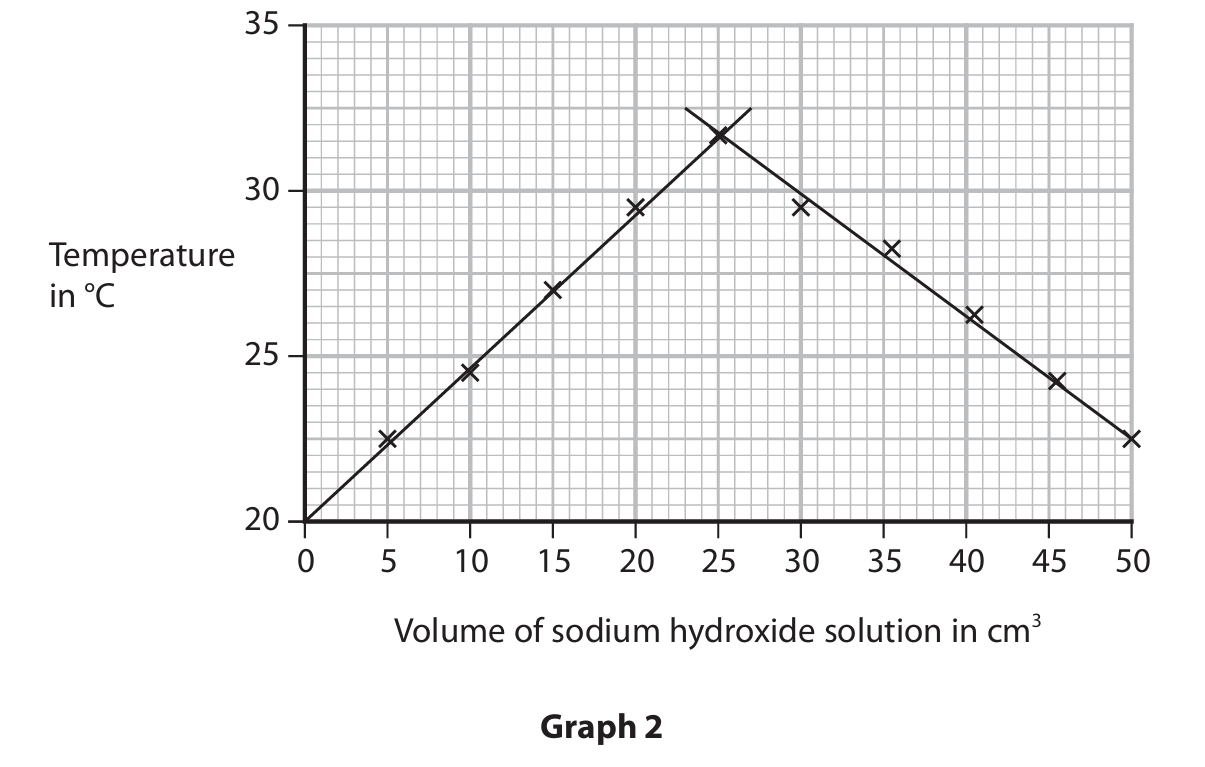
Explain the shape of graph 2.
Most-appropriate topic codes (Edexcel IGCSE Chemistry – 4CH1):
• 2(g): Acids, bases and salt preparations — part (a)(ii) (neutralisation), 2.32, 2.38
• 3(a): Energetics — part (c), 3.1, 3.8 (neutralisation as exothermic)
• Appendix 5: Command word taxonomy — State, Name, Determine, Suggest, Give, Explain
• 2: Chemistry content – Experimental skills — parts (a)(i), (b)(ii), practical techniques and data interpretation
▶️ Answer/Explanation
(a)(i) Measuring cylinder / burette / (volumetric) pipette
(a)(ii) Neutralisation
(b)(i) 12.4
(b)(ii)
• 15 cm3 — red/orange
• 30 cm3 — blue/purple
(b)(iii) OH– / hydroxide (ion)
(c)
M1: The reaction is exothermic (therefore the temperature rises).
M2: (After 25 cm3 of sodium hydroxide) the reaction is complete / neutralisation happens.
M3: So adding more sodium hydroxide / liquid / solution cools the mixture down / no more heat energy is given out.
(a) The diagram shows the arrangement of electrons in an atom of calcium and in an atom of chlorine.
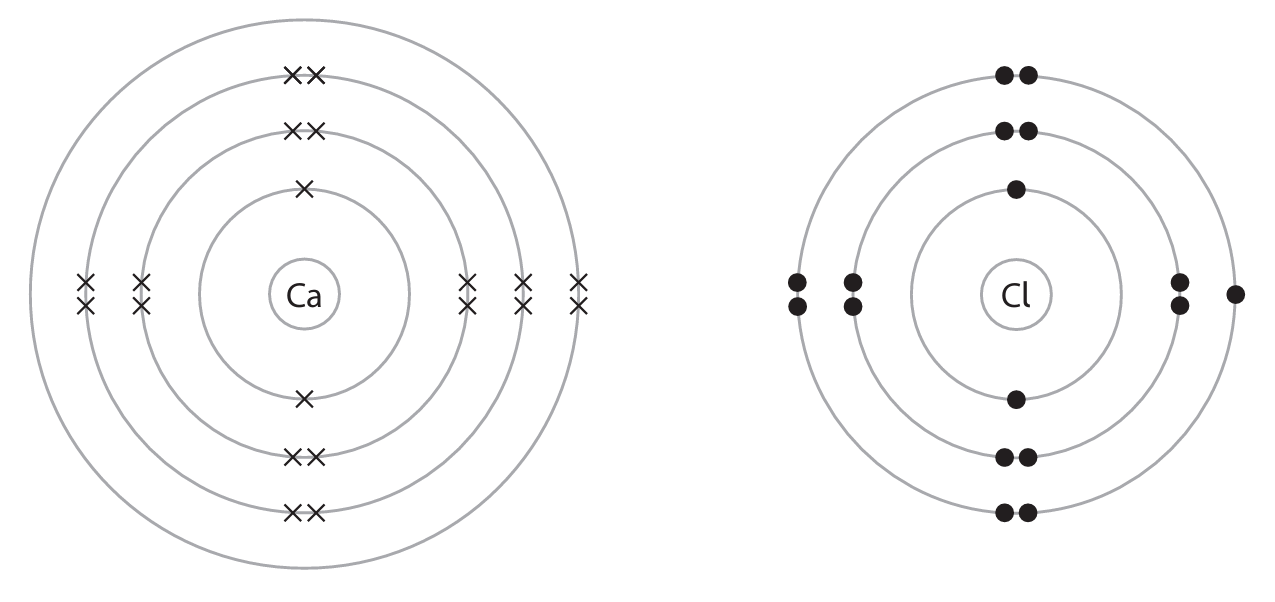
Describe, in terms of electrons, what happens when calcium reacts with chlorine to form the ionic compound calcium chloride, \( \text{CaCl}_2 \)
(b) Describe tests to show that an aqueous solution of calcium chloride contains calcium ions and chloride ions.
(c) Solid calcium chloride does not conduct electricity. Aqueous solutions of calcium chloride do conduct electricity.
A student uses this method to investigate how the conductivity of a solution changes when calcium chloride is dissolved in pure water.
- add \(100 \, \text{cm}^3\) of pure water to a beaker
- add one spatula of solid calcium chloride to the beaker
- stir the solution
- measure the conductivity of the solution
- repeat until nine spatulas of solid calcium chloride have been added
The table shows the student’s results.
| Number of spatulas of calcium chloride | Conductivity of solution in arbitrary units |
|---|---|
| 0 | 0 |
| 1 | 6 |
| 2 | 12 |
| 3 | 12 |
| 4 | 24 |
| 5 | 30 |
| 6 | 36 |
| 7 | 36 |
| 8 | 36 |
| 9 | 36 |
(i) Plot the results on the grid and draw two straight lines of best fit. Ignore the anomalous result.
(ii) State the trend shown on the graph for the first six spatulas of calcium chloride.
(iii) Suggest an error the student could have made to cause the anomalous result.
(d) Describe another way to make solid calcium chloride conduct electricity.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(h): Chemical tests — part (b)
• 1(i): Electrolysis — parts (c), (d)
• Appendix 4: Mathematical skills — part (c)(i) graph plotting
• Appendix 5: Command word taxonomy (Describe, State, Suggest) — parts (a), (b), (c)(ii), (c)(iii), (d)
▶️ Answer/Explanation
(a)
| Answer | Notes | Marks |
|---|---|---|
| M1 calcium loses electrons M2 chlorine gains electrons M3 two atoms of chlorine each gain one electron OR M3 calcium loses 2 electrons and chlorine gains 1 electron | IGNORE references to redox Allow 1 mark from M1 and M2 for electron transfer from chlorine to calcium If chlorine molecules are gaining electrons do not award M3 Any reference to sharing electrons or covalent or metallic bonding scores 0 | 3 |
(b)
| Answer | Notes | Marks |
|---|---|---|
| (test for \( \text{Ca}^{2+} \) ions) M1 flame test (allow description of a flame test) M2 orange-red flame colour (test for \( \text{Cl}^- \) ions) M3 add silver nitrate M4 white precipitate | ALLOW brick-red ALLOW M1 add sodium hydroxide | 4 |
(c)(i) 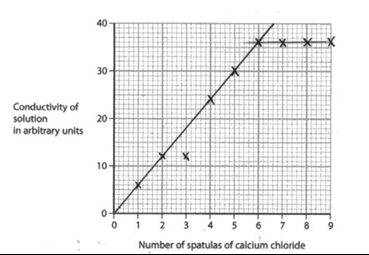
| Answer | Notes | Marks |
|---|---|---|
| M1 and M2 all points correct ± half a square M3 2 straight lines of best fit ignoring the anomalous point | One plotting error scores M1 | 3 |
(c)(ii) the conductivity is (directly) proportional (to the number of spatulas of calcium chloride added)
OR
the conductivity increases (as the number of spatulas of calcium chloride increases) (1 mark)
(c)(iii) Any one from:
• The student took the reading before adding the calcium chloride
• The student forgot to stir the mixture OR did not stir the mixture properly
IGNORE any references to human error (1 mark)
(d)
M1 Heat (the calcium chloride)
M2 until molten / melts
IGNORE references to electrons / ions (2 marks)
This question is about alkenes and polymers.
(a) (i) Ethene \( \text{(C}_2\text{H}_4\text{)} \) can be represented by different types of formula. Complete the table by giving the missing information.
| Molecular formula | \( \text{C}_2\text{H}_4 \) |
|---|---|
| Empirical formula | |
| General formula |
(ii) Ethene is a member of the homologous series of alkenes. All members of the same homologous series have the same general formula. Give two other characteristics of a homologous series.
(b) Ethene is used to make poly(ethene).
(i) State the type of polymerisation used to form poly(ethene).
(ii) Complete the equation for the polymerisation of ethene.
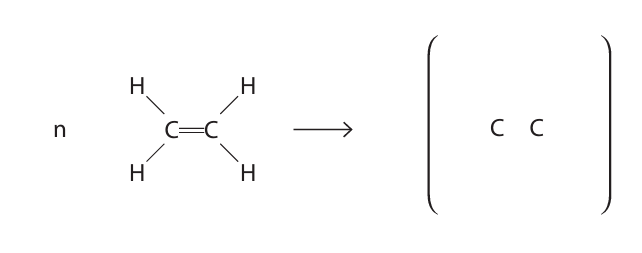
(iii) Poly(ethene) is used to make plastic bags.
Corn starch from plants can also be used to make polymers for plastic bags.
The table gives some information about poly(ethene) and polymers made from corn starch.
| Poly(ethene) | Polymers from corn starch | |
|---|---|---|
| Cost per tonne | £1500 | £3700 |
| Relative strength | 100 | 50 |
| Time to decompose | estimated 450 years | 3–6 months |
Use the information in the table and your knowledge to discuss the advantages and disadvantages of using poly(ethene) to make plastic bags.
(c) The diagram shows the repeat unit of another polymer.
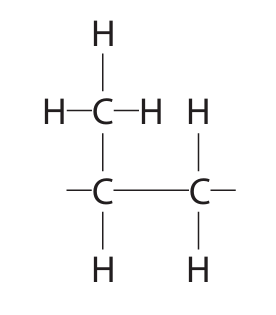
Draw the displayed formula of the monomer used to make this polymer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4.4 Alkenes: General formula, homologous series characteristics (a)(i), (a)(ii)
• 4.8 Synthetic Polymers: Addition polymerisation; drawing polymerisation equations; properties and disposal of polymers (b)(i), (b)(ii), (b)(iii), (c)
• 4.8C Condensation Polymerisation: Identifying monomers from polyesters (c)
▶️ Answer/Explanation
(a)(i)
| Molecular formula | \( \text{C}_2\text{H}_4 \) |
|---|---|
| Empirical formula | \( \text{CH}_2 \) |
| General formula | \( \text{C}_n\text{H}_{2n} \) |
(a)(ii)
Any two from:
1. Each member differs from the next by a \( \text{CH}_2 \) group.
2. They have the same functional group.
3. They have similar chemical properties / react in similar ways.
4. There is a trend in their physical properties (e.g., boiling point).
(b)(i) Addition (polymerisation)
(b)(ii) 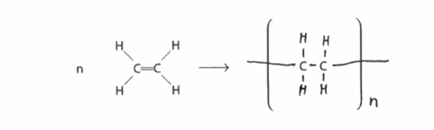
$$ n \text{H}_2\text{C}=\text{CH}_2 \rightarrow \text{—[CH}_2\text{—CH}_2\text{—]}_n— $$ or the displayed structure with single bonds between carbons and hydrogens, and trailing bonds through brackets and ‘n’ to the right.
(b)(iii)
Advantages of poly(ethene):
• Cheaper per tonne (£1500 vs £3700).
• Stronger (relative strength 100 vs 50).
• Does not use land that could be used for food crops.
• Comes from crude oil (a well-established industrial process).
Disadvantages of poly(ethene):
• Non-biodegradable / takes much longer to decompose (450 years vs 3–6 months).
• Causes problems with litter and landfill disposal.
• Burning it can release toxic fumes/greenhouse gases.
• Made from a non-renewable, finite resource (crude oil).
(c) 
The monomer is a dicarboxylic acid and a diol. The displayed formula of the monomer pair that forms this polyester is:
HOOC—COOH (Ethanedioic acid) and HO—CH\(_2\)—CH\(_2\)—OH (Ethanediol)
Or, if drawn as a single monomer for the repeat unit, it is not a single alkene but a combination of these two molecules. The displayed structure should show:
H—O—C(=O)—C(=O)—O—H and H—O—CH\(_2\)—CH\(_2\)—O—H
Note: The polymer is formed by condensation polymerisation between these two monomers.
This question is about carbon and its compounds.
(a)(i) Draw a dot-and-cross diagram to show the outer shell electrons in a molecule of carbon dioxide, \( \text{CO}_2 \).
(ii) The atoms in carbon dioxide are held together by covalent bonds. Describe the forces of attraction in a covalent bond.
(b) The diagram shows three different structures of carbon.
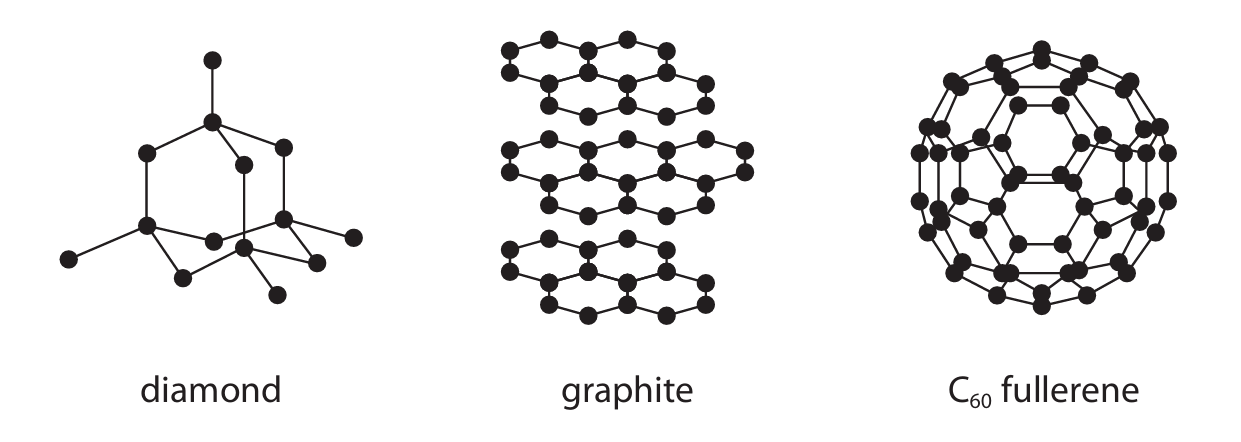
(i) Explain why graphite conducts electricity.
(ii) Explain why diamond has a much higher melting point than \( C_{60} \) fullerene. Refer to structure and bonding in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(g): Covalent bonding & structures — part (b)(i)
• 1(g): Covalent bonding & structures (Diamond and Graphite) — parts (b)(i), (b)(ii)
• 1(g): Simple molecular vs. giant covalent structures — part (b)(ii)
▶️ Answer/Explanation
(a)(i)
M1: Four electrons (two shared pairs) between the carbon and each oxygen atom.
M2: Rest of the molecule correct (lone pairs on oxygen atoms correctly shown).
(a)(ii)
M1: Shared pair(s) of electrons.
M2: (The electrons are) attracted to the (two) nuclei.
REJECT: singular “nucleus”. Must be plural for M2.
(b)(i)
M1: Graphite has delocalised electrons.
M2: The delocalised electrons can move or flow (throughout the structure), carrying charge.
IGNORE: sea of electrons, free electrons, references to layers. Any mention of ions scores 0.
(b)(ii)
M1: Diamond has a giant covalent / macromolecular structure.
M2: To melt diamond, strong covalent bonds must be broken.
M3: \( C_{60} \) fullerene has a simple molecular structure.
M4: To melt \( C_{60} \), only weak intermolecular forces (of attraction) are overcome.
M5: More energy is needed to break the strong covalent bonds in diamond than to overcome the weak intermolecular forces in \( C_{60} \).
ACCEPT: Breaking bonds in \( C_{60} \) if intermolecular forces are clearly mentioned.
REJECT: Mention of breaking covalent bonds in \( C_{60} \).
This question is about the reduction of metal oxides.
(a) Solid oxides of copper can be reduced by reacting them with methane gas.
Complete the equation for the reaction between copper(II) oxide and methane.
Include state symbols.
CuO(………..) + ………CH4(………..) → ………Cu(………..) + ………CO2(………..) + ………H2O(……….)
(b) A teacher uses this apparatus to demonstrate the reaction between a different oxide of copper and methane.
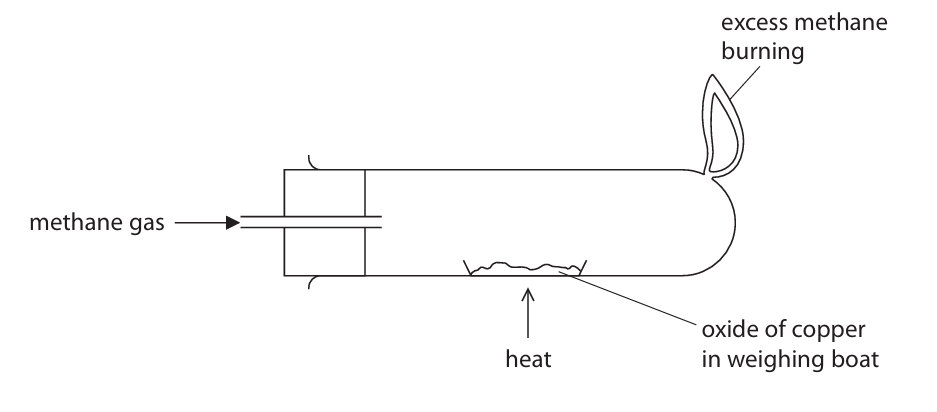
(i) The teacher heats the oxide of copper until the reaction is complete. The table shows the teacher’s results.
| Mass in g | |
|---|---|
| empty weighing boat | 15.05 |
| weighing boat + oxide of copper | 18.63 |
| weighing boat + copper | 18.23 |
Use the teacher’s results to show that the empirical formula of this oxide of copper is Cu2O.
(ii) The teacher wears safety glasses and a lab coat during the demonstration. Give one other safety precaution that she should take.
(c) Iron forms when iron(III) oxide is heated with carbon.
The equation for the reaction is
\[ \text{Fe}_2\text{O}_3 + 3\text{C} \rightarrow 2\text{Fe} + 3\text{CO} \]
(i) State how the equation shows that iron(III) oxide is reduced.
(ii) State why carbon monoxide should not be released into the atmosphere.
(iii) Calculate the maximum mass, in tonnes, of iron that can be produced when 30.0 tonnes of iron(III) oxide are reacted with an excess of carbon.
\[ [1 \, \text{tonne} = 1.0 \times 10^6 \, \text{g}] \]
(iv) A mixture of 25 000 mol of iron(III) oxide and 840 000 g of carbon is heated. Use this equation to show that the iron(III) oxide is in excess.
\[ \text{Fe}_2\text{O}_3 + 3\text{C} \rightarrow 2\text{Fe} + 3\text{CO} \]
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(d): Reactivity series — part (c)(i) (reduction defined as loss of oxygen)
• 2(e): Extraction and uses of metals — parts (a), (b), (c)
• 4(b): Crude oil (combustion products) — part (c)(ii) (toxicity of carbon monoxide)
• Practical Skills: Experimental safety — part (b)(ii)
▶️ Answer/Explanation
(a)
\[ 4\text{CuO(s)} + \text{CH}_4\text{(g)} \rightarrow 4\text{Cu(s)} + \text{CO}_2\text{(g)} + 2\text{H}_2\text{O(l/g)} \]
M1 correct balancing (4,1,4,1,2). M2 correct state symbols.
(b)(i)
Mass of copper oxide = \( 18.63 – 15.05 = 3.58 \text{ g} \)
Mass of copper produced = \( 18.23 – 15.05 = 3.18 \text{ g} \)
Mass of oxygen in oxide = \( 3.58 – 3.18 = 0.40 \text{ g} \)
Moles of copper = \( \frac{3.18}{63.5} = 0.0501 \text{ mol} \)
Moles of oxygen = \( \frac{0.40}{16.0} = 0.0250 \text{ mol} \)
Ratio Cu : O = \( 0.0501 : 0.0250 = 2 : 1 \)
Therefore, empirical formula = \(\text{Cu}_2\text{O}\).
(b)(ii)
Any one from:
• Use a safety screen / wear heat-proof gloves / tie hair back.
• Position the class at a safe distance.
• Conduct the experiment in a fume cupboard.
• Ignite the excess methane immediately to prevent buildup.
(c)(i)
Iron(III) oxide loses oxygen (to form iron).
(c)(ii)
Carbon monoxide is poisonous / toxic. It reduces the blood’s ability to carry oxygen.
(c)(iii)
\( M_r(\text{Fe}_2\text{O}_3) = (2 \times 56) + (3 \times 16) = 160 \)
Mass of \(\text{Fe}_2\text{O}_3\) in grams = \( 30.0 \times 10^6 \text{ g} \)
Moles of \(\text{Fe}_2\text{O}_3\) = \( \frac{30.0 \times 10^6}{160} = 187,500 \text{ mol} \)
From equation: moles of Fe = \( 2 \times \text{moles of Fe}_2\text{O}_3 = 2 \times 187,500 = 375,000 \text{ mol} \)
Mass of Fe = \( 375,000 \times 56 = 21,000,000 \text{ g} = 21.0 \text{ tonnes} \)
Maximum mass of iron = 21.0 tonnes.
(c)(iv)
Moles of carbon = \( \frac{840,000}{12} = 70,000 \text{ mol} \)
From equation, 3 mol C react with 1 mol \(\text{Fe}_2\text{O}_3\).
Therefore, moles of \(\text{Fe}_2\text{O}_3\) needed = \( \frac{70,000}{3} \approx 23,333 \text{ mol} \)
Moles of \(\text{Fe}_2\text{O}_3\) available = 25,000 mol.
Since available (25,000 mol) > needed (23,333 mol), iron(III) oxide is in excess.
