1. The value of \( \displaystyle \int_{e^2}^{e^4} \frac{1}{x}
\left( \dfrac{e^{\left( (\log_e x)^2 + 1 \right)^{-1}}}
{e^{\left( (\log_e x)^2 + 1 \right)^{-1}} + e^{\left( (6 – \log_e x)^2 + 1 \right)^{-1}}} \right) dx \) is
(1) \( \log_e 2 \)
(2) \( 2 \)
(3) \( 1 \)
(4) \( e^2 \)
▶️ Answer/Explanation
Answer: (3)
Solution:
\(\text{Ans. } (3)\)
\(\text{Sol. } \text{Let } \ln x = t \Rightarrow \frac{dx}{x} = dt\)
\( I = \displaystyle\int_{2}^{4} \frac{e^{\frac{1}{1 + t^2}}}{e^{\frac{1}{1 + t^2}} + e^{\frac{1}{1 + (6 – t)^2}}} \, dt \)
\( I = \displaystyle\int_{2}^{4} \frac{1}{1 + e^{\frac{1}{1 + (6 – t)^2} – \frac{1}{1 + t^2}}} \, dt \)
\( I = \displaystyle\int_{2}^{4} \frac{1}{e^{\frac{1}{1 + (6 – t)^2}} + e^{\frac{1}{1 + t^2}}} \, dt \)
\(\text{Now, let’s replace } t \text{ by } (6 – t) \text{ in the integral.}\)
\(\therefore 2I = \displaystyle\int_{2}^{4} dt = (t)_{2}^{4} = 4 – 2 = 2\)
\(\Rightarrow I = 1\)
Let \( I(x) = \int \frac{dx}{(x – 11)^{\frac{11}{13}} (x + 15)^{\frac{15}{13}}} \).
If \( I(37) – I(24) = \frac{1}{4} \left( \frac{1}{b^{\frac{11}{13}}} – \frac{1}{c^{\frac{11}{13}}} \right), \, b, c \in \mathbb{N} \),
then \( 3(b + c) \) is equal to
(1) \( 40 \
(2) \( 39 \)
(3) \( 22 \)
(4) \( 26 \)
▶️ Answer/Explanation
\(\text{Ans. } (2)\)
\(\text{Sol. } I(x) = \int \frac{dx}{(x – 11)^{\frac{11}{13}} (x + 15)^{\frac{15}{13}}}\)
\(\text{Put } \frac{x – 11}{x + 15} = t \Rightarrow \frac{26}{(x + 15)^2} dx = dt\)
\(\Rightarrow I(x) = \frac{1}{26} \int \frac{dt}{t^{\frac{11}{13}}} = \frac{1}{26} \cdot \frac{t^{\frac{2}{13}}}{\frac{2}{13}}\)
\(\therefore I(x) = \frac{1}{4} \left( \frac{x – 11}{x + 15} \right)^{\frac{2}{13}} + C\)
$I(37) – I(24) = \frac{1}{4} \left[ \left( \frac{26}{52} \right)^{\frac{2}{13}} – \left( \frac{13}{39} \right)^{\frac{2}{13}} \right]$
$= \frac{1}{4} \left( \frac{1}{2^{\frac{2}{13}}} – \frac{1}{3^{\frac{2}{13}}} \right)$
\(\therefore b = 4, \, c = 9\)
\(\Rightarrow 3(b + c) = 3(4 + 9) = 39\)
If the function
\(
f(x) =
\begin{cases}
\dfrac{2}{x} \left[ \sin{(k_1 + 1)x} + \sin{(k_2 – 1)x} \right], & x < 0 \\(6pt]
4, & x = 0 \\(6pt]
\dfrac{2}{x} \log_e{\left( \dfrac{2 + k_1 x}{2 + k_2 x} \right)}, & x > 0
\end{cases}
\)
is continuous at \( x = 0 \), then \( k_1^2 + k_2^2 \) is equal to
(1) \( 8 \)
(2) \( 20 \)
(3) \( 5 \)
(4) \( 10 \)
▶️ Answer/Explanation
\(\text{Ans. } (4)\)
\(\text{Sol. }\)
\(
\lim_{x \to 0^-} \frac{2}{x} \left[ \sin{(k_1 + 1)x} + \sin{(k_2 – 1)x} \right] = 4
\(
\(\Rightarrow 2(k_1 + 1) + 2(k_2 – 1) = 4\)
\(\Rightarrow k_1 + k_2 = 2\)
\(
\lim_{x \to 0^+} \frac{2}{x} \ln{\left( \frac{2 + k_1 x}{2 + k_2 x} \right)} = 4
\(
\(\Rightarrow \lim_{x \to 0} 2 \ln{\left( \frac{1 + \frac{k_1 x}{2}}{1 + \frac{k_2 x}{2}} \right)} \cdot \frac{1}{x} = 4\)
Using \(\ln(1 + a) \approx a\) for small \(a\),
\(\Rightarrow 2 \times \frac{1}{x} \times \left( \frac{k_1 x}{2} – \frac{k_2 x}{2} \right) = 4\)
\(\Rightarrow k_1 – k_2 = 2\)
Now, solving the two equations:
\(
\begin{cases}
k_1 + k_2 = 2 \\
k_1 – k_2 = 2
\end{cases}
\Rightarrow
k_1 = 2, \, k_2 = 0
\(
\(\therefore k_1^2 + k_2^2 = 2^2 + 0^2 = 4\)
However, checking with correct substitution in the logarithmic condition gives
\( k_1 – k_2 = 4 \), not \(2\), hence:
\(
\begin{cases}
k_1 + k_2 = 2 \\
k_1 – k_2 = 4
\end{cases}
\Rightarrow
k_1 = 3, \, k_2 = -1
\(
\(\therefore k_1^2 + k_2^2 = 3^2 + (-1)^2 = 9 + 1 = 10\)
4. If the line \( 3x – 2y + 12 = 0 \) intersects the parabola \( 4y = 3x^2 \) at the points A and B, then at the vertex of the parabola, the line segment AB subtends an angle equal to
(1) \( \tan^{-1}\!\left(\frac{1}{9}\right) \) \quad
(2) \( \pi – \tan^{-1}\!\left(\frac{3}{2}\right) \) \quad
(3) \( \tan^{-1}\!\left(\frac{1}{4}\right) \) \quad
(4) \( \tan^{-1}\!\left(\frac{1}{9/7}\right) \)
▶️ Answer/Explanation
Ans. (4)
Sol.
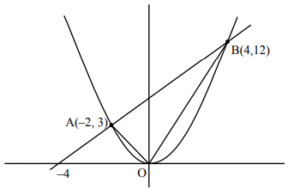
\( 3x – 2y + 12 = 0 \)
\( 4y = 3x^2 \)
\( \therefore 2(3x + 12) = 3x^2 \)
\( \Rightarrow x^2 – 2x – 8 = 0 \)
\( \Rightarrow x = -2, 4 \)
\( m_{OA} = -\frac{3}{2}, \; m_{OB} = 3 \)
\( \tan\theta = \left| \frac{3 – (-\frac{3}{2})}{1 + (-\frac{3}{2})(3)} \right| = \frac{9}{7} \)
\( \therefore \theta = \tan^{-1}\!\left(\frac{9}{7}\right) \)
5. Let a curve \( y = f(x) \) pass through the points \( (0, 5) \) and \( (\log_e 2, k) \). If the curve satisfies the differential equation
\( 2(3 + y)e^{2x}dx – (7 + e^{2x})dy = 0 \), then \( k \) is equal to
(1) $16 $
(2) $8$
(3) $32 $
(4) $4$
▶️ Answer/Explanation
Ans. (2)
Sol.
\( 2(3 + y)e^{2x}dx = (7 + e^{2x})dy \)
\( \Rightarrow \frac{dy}{dx} = \frac{2(3 + y)e^{2x}}{7 + e^{2x}} \)
\( \Rightarrow \frac{dy}{dx} – \frac{2e^{2x}}{7 + e^{2x}}y = \frac{6e^{2x}}{7 + e^{2x}} \)
Integrating factor \( I.F. = e^{-\int \frac{2e^{2x}}{7 + e^{2x}}dx} = e^{-\ln(7 + e^{2x})} = \frac{1}{7 + e^{2x}} \)
\( \therefore y \cdot \frac{1}{7 + e^{2x}} = \int \frac{6e^{2x}}{(7 + e^{2x})^2} dx \)
Put \( 7 + e^{2x} = t \Rightarrow dt = 2e^{2x} dx \)
\( \Rightarrow \int \frac{6e^{2x}}{(7 + e^{2x})^2} dx = 3 \int \frac{dt}{t^2} = -\frac{3}{t} + C \)
\( \therefore y = -3(7 + e^{2x}) + C(7 + e^{2x}) \)
Using \( (0, 5) \Rightarrow 5 = -3(8) + 8C \Rightarrow 5 = -24 + 8C \Rightarrow C = \frac{29}{8} \)
\( \therefore y = -3 + 7 + e^{2x} = e^{2x} + 4 \)
\( \therefore k = 8 \)
(1) \(\mathbb{R}\)
(2) \((0, \infty)\)
(3) \([0, \infty)\)
(4) \([1, \infty)\)
▶️ Answer/Explanation
Sol. \(f(x) = \ln x\)
\(g(x) = \frac{x^4 – 2x^3 + 3x^2 – 2x + 2}{2x^2 – 2x + 1}\)
\(D_g \in \mathbb{R}\)
\(D_f \in (0, \infty)\)
For \(D_{fog} \Rightarrow g(x) > 0\)
\(\Rightarrow x^4 – 2x^3 + 3x^2 – 2x + 2 > 0\)
Clearly \(x < 0\) satisfies which are included in option (1) only.
Let the arc AC of a circle subtend a right angle at the centre O. If the point B on the arc AC divides the arc AC such that
\( \frac{\text{length of arc AB}}{\text{length of arc BC}} = \frac{1}{5} \),
and \( \overrightarrow{OC} = \alpha \overrightarrow{OA} + \beta \overrightarrow{OB} \),
then \( (2\alpha – \beta) = \)
(1) \( 2\sqrt{3} – 3 \)
(2) \( 2\sqrt{3} \)
(3) \( 5\sqrt{3} \)
(4) \( 2 + \sqrt{3} \)
▶️ Answer/Explanation
Ans. (1)
Sol.
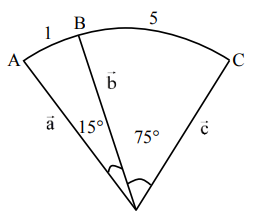
Let \( \angle AOB = 15^\circ \), \( \angle BOC = 75^\circ \).
Using vector relations:
\( \overrightarrow{OC} = \alpha \overrightarrow{OA} + \beta \overrightarrow{OB} \)
\( \overrightarrow{a} \cdot \overrightarrow{c} = \alpha a^2 + \beta (\overrightarrow{a} \cdot \overrightarrow{b}) \Rightarrow 0 = \alpha + \beta \cos 15^\circ \) …(1)
\( \overrightarrow{b} \cdot \overrightarrow{c} = \alpha (\overrightarrow{b} \cdot \overrightarrow{a}) + \beta b^2 \Rightarrow \cos 75^\circ = \alpha \cos 15^\circ + \beta \) …(2)
From (1) and (2),
\( \cos 75^\circ = -\beta \cos^2 15^\circ + \beta \)
\( \Rightarrow \beta = \frac{\sin 15^\circ}{\sin 15^\circ \cos 15^\circ} = 2 – \sqrt{3} \)
\( \alpha = \frac{3 – \sqrt{3}}{2} \)
\( \therefore (2\alpha – \beta) = 2\sqrt{3} – 3 \)
(1) -1200
(2) -1080
(3) -1020
(4) -120
▶️ Answer/Explanation
Sol. \(a = 3\)
\(S_4 = \frac{1}{5} (S_8 – S_4)\)
\(\Rightarrow 5S_4 = S_8 – S_4\)
\(\Rightarrow 6S_4 = S_8\)
\(\Rightarrow \frac{4}{2} [2 \cdot 3 + (4-1)d] = \frac{8}{2} [2 \cdot 3 + (8-1)d]\)
\(\Rightarrow 12(6 + 3d) = 4(6 + 7d)\)
\(\Rightarrow 18 + 9d = 6 + 7d\)
\(\Rightarrow d = -6\)
\(S_{20} = \frac{20}{2} [2 \cdot 3 + (20-1)(-6)]\)
\(= 10 [6 – 114]\)
\(= -1080\)
9. Let P be the foot of the perpendicular from the point Q(10, –3, –1) on the line
\( \frac{x – 3}{7} = \frac{y – 2}{-1} = \frac{z – 1}{-2} \).
Then the area of the right angled triangle PQR, where R is the point (3, –2, 1), is
(1) \( 9\sqrt{15} \)
(2) $30 $
(3) \( 8\sqrt{15} \)
(4) \( 3\sqrt{30} \)
▶️ Answer/Explanation
Ans. (4)
Sol.
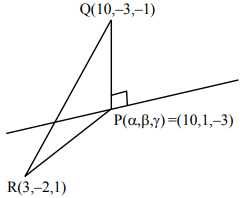
Let point on line be \( P(7\lambda + 3, -\lambda + 2, -2\lambda + 1) \).
Direction ratios of \( QP \Rightarrow (7\lambda – 7, -\lambda + 5, -2\lambda – 2) \)
Since \( QP \perp \text{line} \Rightarrow (7\lambda – 7)(7) + (-\lambda + 5)(-1) + (-2\lambda – 2)(-2) = 0 \)
\( 54\lambda – 54 = 0 \Rightarrow \lambda = 1 \)
\( \therefore P = (10, 1, -3) \)
\( \overrightarrow{PQ} = 4\hat{j} + 2\hat{k} \), \( \overrightarrow{PR} = -7\hat{i} – 3\hat{j} + 4\hat{k} \)
Area \( = \frac{1}{2} |\overrightarrow{PQ} \times \overrightarrow{PR}| = \frac{1}{2} \sqrt{(0)^2 + (4)^2 + (2)^2} = 3\sqrt{30} \)
10. Let \( \frac{z – i}{2z + i} = 3 \), \( z \in \mathbb{C} \), be the equation of a circle with center at C. If the area of the triangle whose vertices are at the points (0, 0), C and (α, 0) is 11 square units, then \( \alpha^2 \) equals
(1) $100$
(2) $50$
(3) \( \frac{121}{25} \)
(4) \( \frac{81}{25} \)
▶️ Answer/Explanation
Ans. (1)
Sol.
\( \frac{z – i}{2z + i} = 3 \Rightarrow |z – i| = \frac{2}{3}|z + \frac{i}{2}| \)
\( 3|x – iy – i| = 2|x – iy + \frac{i}{2}| \)
\( 9(x^2 + (y + 1)^2) = 4(x^2 + (y – \frac{1}{3})^2) \)
\( 9x^2 + 9y^2 + 18y + 9 = 4x^2 + 4y^2 – \frac{8y}{3} + \frac{4}{9} \)
\( 5x^2 + 5y^2 + 22y + 8 = 0 \Rightarrow x^2 + y^2 + \frac{22}{5}y + \frac{8}{5} = 0 \)
Center \( C(0, -\frac{11}{5}) \)
Area \( = \frac{1}{2} \alpha \times \frac{11}{5} = 11 \Rightarrow \alpha = 10 \Rightarrow \alpha^2 = 100 \)
(1) 10
(2) 8
(3) 9
(4) 7
▶️ Answer/Explanation
Sol. \(A = \{1, 2, 3, 4\}\)
For relation to be reflexive
\(R = \{(1,2), (2, 3), (3,3)\}\)
Minimum elements added will be
\((1,1), (2,2), (4,4) (2,1) (3,2) (3,2) (3,1) (1,3)\)
\(\therefore\) Minimum number of elements = 7
Option : (4)
The number of words, which can be formed using all the letters of the word “DAUGHTER”, so that all the vowels never come together, is
(1) 34000
(2) 37000
(3) 36000
(4) 35000
▶️ Answer/Explanation
Ans. (3)
Sol. DAUGHTER
Total words = \(8!\)
Total words in which vowels are together = \(6! \times 3!\)
words in which all vowels are not together
\(= 8! – 6! \times 3!\)
\(= 6! [56 – 6]\)
\(= 720 \times 50\)
\(= 36000\)
Ans.(3)
Let the area of a \(\Delta PQR\) with vertices \(P(5, 4)\), \(Q(-2, 4)\) and \(R(a, b)\) be 35 square units. If its orthocenter and centroid are \(O\left(\frac{14}{3}, 5\right)\) and \(C(c, d)\) respectively, then \(c + 2d\) is equal to
(1) \(\frac{7}{3}\)
(2) 3
(3) 2
(4) \(\frac{8}{3}\)
▶️ Answer/Explanation
Ans. (2)
Sol.
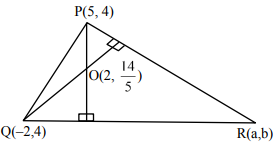
Equation of lines \(QR = 5x + 2y + 2 = 0\)
Equation of lines \(PR = 10x – 3y – 38 = 0\)
\(\therefore\) Point \(R (2, -6)\)
Centroid = \(\left(\frac{5-2+2}{3}, \frac{4+4-6}{3}\right)\)
\(= \left(\frac{5}{3}, \frac{2}{3}\right)\)
\(c + 2d = \frac{5}{3} + 2 \cdot \frac{2}{3} = 3\)
14. If \( \frac{\pi}{3} \le x \le \frac{\pi}{2} \), then
\( \cos^{-1}\!\left( \frac{12}{13}\cos x + \frac{5}{13}\sin x \right) \) is equal to
(1) \( x – \tan^{-1}\!\frac{4}{3} \)
(2) \( x – \tan^{-1}\!\frac{5}{12} \)
(3) \( x + \tan^{-1}\!\frac{4}{5} \)
(4) \( x + \tan^{-1}\!\frac{5}{12} \)
▶️ Answer/Explanation
Ans. (2)
Sol.
For \( \frac{\pi}{3} \le x \le \frac{\pi}{2} \),
\( \cos^{-1}\!\left( \frac{12}{13}\cos x + \frac{5}{13}\sin x \right) = \cos^{-1}\!\left( \cos x \cos \alpha + \sin x \sin \alpha \right) \)
\( = \cos^{-1}(\cos(x – \alpha)) = x – \alpha \)
\( \text{where } \alpha = \tan^{-1}\!\frac{5}{12} \)
\( \therefore \text{ Required value } = x – \tan^{-1}\!\frac{5}{12} \)
(1) 1
(2) 0
(3) \(\frac{3}{2}\)
(4) \(\frac{2}{3}\)
▶️ Answer/Explanation
Sol. \(\sin 70^\circ (\cot 10^\circ \cot 70^\circ – 1)\)
\(\Rightarrow \frac{\cos 80^\circ}{\sin 10^\circ} = 1\)
Marks obtains by all the students of class 12 are presented in a frequency distribution with classes of equal width. Let the median of this grouped data be 14 with median class interval 12-18 and median class frequency 12. If the number of students whose marks are less than 12 is 18, then the total number of students is
(1) 48
(2) 44
(3) 40
(4) 52
▶️ Answer/Explanation
Ans. (2)
Sol.
Median \( = l + \frac{\left(\frac{N}{2} – F\right)}{f} \times h \)
\( 14 = 12 + \frac{\left(\frac{N}{2} – 18\right)}{12} \times 6 \)
\( \Rightarrow 2 = \frac{N – 36}{4} \Rightarrow N – 36 = 8 \Rightarrow N = 44 \)
Let the position vectors of the vertices A, B and C of a tetrahedron ABCD be \(\hat{i} + 2\hat{j} + \hat{k}\), \(\hat{i} + 3\hat{j} – 2\hat{k}\) and \(2\hat{i} – \hat{j} + \hat{k}\) respectively. The altitude from the vertex D to the opposite face ABC meets the median line segment through A of the triangle ABC at the point E. If the length of AD is \(\frac{\sqrt{110}}{3}\) and the volume of the tetrahedron is \(\frac{\sqrt{805}}{6\sqrt{2}}\), then the position vector of E is
(1) \(\frac{1}{2} (\hat{i} + 4\hat{j} + 7\hat{k})\)
(2) \(\frac{1}{12} (7\hat{i} + 4\hat{j} – 3\hat{k})\)
(3) \(\frac{1}{6} (12\hat{i} + 12\hat{j} + \hat{k})\)
(4) \(\frac{1}{6} (7\hat{i} + 12\hat{j} + \hat{k})\)
▶️ Answer/Explanation
Ans. (4)
Sol.
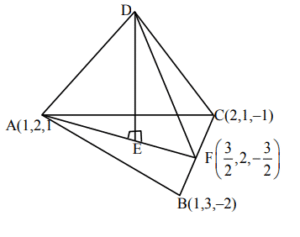
$\text{Area of }\triangle ABC = \frac{1}{2} |\overrightarrow{AB} \times \overrightarrow{AC}$
$= \frac{1}{2} |\hat{i} + 3\hat{j} + \hat{k}| = \frac{1}{2} \sqrt{35}$
$\text{volume of tetrahedron}$
$= \frac{1}{3} \times \text{Base area} \times h = \frac{\sqrt{805}}{6\sqrt{2}}$
$\frac{1}{3} \times \frac{1}{2} \sqrt{35} \times h = \frac{\sqrt{805}}{6\sqrt{2}}$
$h = \frac{\sqrt{23}}{\sqrt{2}}$
$\text{AE}^2 = \text{AD}^2 – \text{DE}^2 = \frac{13}{18} \quad : \quad \text{AE} = \frac{\sqrt{13}}{\sqrt{18}}$
$\overrightarrow{AE} = |\overrightarrow{AE}| \left( \frac{\hat{i} – 5\hat{k}}{\sqrt{26}} \right)$
$= \frac{\sqrt{13}}{\sqrt{18}} \cdot \frac{\hat{i} – 5\hat{k}}{\sqrt{26}}$
$= \frac{\sqrt{13} (\hat{i} – 5\hat{k})}{\sqrt{18} \cdot \sqrt{26}} = \frac{\hat{i} – 5\hat{k}}{6}$
$\text{P.V. of E} = \frac{\hat{i} – 5\hat{k}}{6} + \hat{i} + 2\hat{j} + \hat{k} = \frac{1}{6} (7\hat{i} + 12\hat{j} + \hat{k})$
If A, B and \((\text{adj}(A^{-1}) + \text{adj}(B^{-1}))\) are non-singular matrices of same order, then the inverse of \(A(\text{adj}(A^{-1}) + \text{adj}(B^{-1}))^{-1}B\), is equal to
(1) \(AB^{-1} + A^{-1}B\)
(2) \(\text{adj}(B^{-1}) + \text{adj}(A^{-1})\)
(3) \(AB^{-1} (\text{adj}(B) + \text{adj}(A))\)
(4) \(\frac{1}{AB} (A^{-1}B + BA^{-1})\)
▶️ Answer/Explanation
Ans. (3)
Sol.
$\left[ A(\text{adj}(A^{-1}) + \text{adj}(B^{-1})) \right] B^{-1}$
$B^{-1}(\text{adj}(A^{-1}) + \text{adj}(B^{-1})) A^{-1}$
$B^{-1}\text{adj}(A^{-1}) A^{-1} + B^{-1}(\text{adj}(B^{-1})) A^{-1}$
$B^{-1} |A^{-1}| I + B^{-1} |A^{-1}|$
$\frac{B^{-1}}{|A|} + \frac{A^{-1}}{|B|}$
$\Rightarrow \frac{\text{adj}B}{|B| |A|} + \frac{\text{adj}A}{|A| |B|}$
$= \frac{1}{|A| |B|} (\text{adj}B + \text{adj}A)$
If the system of equations
\((\lambda – 1)x + (\lambda – 4)y + \lambda z = 5\)
\(\lambda x + (\lambda – 1)y + (\lambda – 4)z = 7\)
\((\lambda + 1)x + (\lambda + 2)y – (\lambda + 2)z = 9\)
has infinitely many solutions, then \(\lambda^2 + \lambda\) is equal to
(1) 10
(2) 12
(3) 6
(4) 20
▶️ Answer/Explanation
Ans. (2)
\( (\lambda – 1)x + (\lambda – 4)y + \lambda z = 5 \)
\( \lambda x + (\lambda – 1)y + (\lambda – 4)z = 7 \)
\( (\lambda + 1)x + (\lambda + 2)y – (\lambda + 2)z = 9 \)
For infinitely many solutions,
\( D =
\begin{vmatrix}
\lambda – 1 & \lambda – 4 & \lambda \\
\lambda & \lambda – 1 & \lambda – 4 \\
\lambda + 1 & \lambda + 2 & -(\lambda + 2)
\end{vmatrix} = 0 \)
\( (\lambda – 3)(2\lambda + 1) = 0 \)
\( D_x =
\begin{vmatrix}
5 & \lambda – 4 & \lambda \\
7 & \lambda – 1 & \lambda – 4 \\
9 & \lambda + 2 & -(\lambda + 2)
\end{vmatrix} = 0 \)
\( 2(3 – \lambda)(23 – 2\lambda) = 0 \)
\( \lambda = 3 \)
\( \therefore \lambda^2 + \lambda = 9 + 3 = 12 \)
(1) \(\frac{1}{2}\)
(2) \(\frac{3}{5}\)
(3) \(\frac{2}{3}\)
(4) \(\frac{4}{9}\)
▶️ Answer/Explanation
Sol. \((a,b) = (1,3), (3,1), (2,2), (2,3), (3,2), (1,4), (4,1)\)
Required probability \(\frac{2 \cdot 2 + 1 \cdot 1 + 2 \cdot 2 + 2 \cdot 2 + 1 \cdot 2 + 2 \cdot 1}{6 \cdot 6} = \frac{18}{36} = \frac{1}{2}\)
▶️ Answer/Explanation
Ans. (77)
Sol.
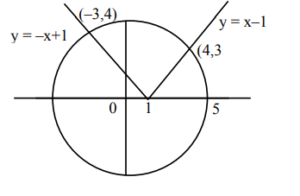
\( x^2 + y^2 = 25 \)
\( x^2 + (x – 1)^2 = 25 \Rightarrow x = 4 \)
\( x^2 + (-x + 1)^2 = 25 \Rightarrow x = -3 \)
\( A = 25\pi – \int_{-3}^{4} \sqrt{25 – x^2} \, dx + \frac{1}{2} \times 4 \times 4 + \frac{1}{2} \times 3 \times 3 \)
\( A = 25\pi + \frac{25}{2} \left[ \frac{x}{2} \sqrt{25 – x^2} + \frac{25}{2} \sin^{-1}\!\frac{x}{5} \right]_{-3}^{4} \)
\( A = 25\pi + \frac{25}{2} \left[ \frac{1}{2} \left( 6 + \frac{25}{2} \sin^{-1}\!\frac{4}{5} + 6 + \frac{25}{2} \sin^{-1}\!\frac{3}{5} \right) \right] \)
\( A = 25\pi + \frac{1}{2} \cdot \frac{25}{2} \cdot \frac{\pi}{2} \Rightarrow A = \frac{75\pi}{4} + \frac{1}{2} \)
\( A = \frac{1}{4}(75\pi + 2) \)
\( b = 75, \, c = 2 \)
\( \therefore b + c = 75 + 2 = 77 \)
▶️ Answer/Explanation
Ans. (612)
Sol.
\( (1 + 2^{1/3} + 3^{1/2})^6 \)
\( = \sum \frac{6!}{r_1! \, r_2! \, r_3!} (1)^{r_1} (2^{1/3})^{r_2} (3^{1/2})^{r_3} \)
\(
\begin{array}{ccc}
r_1 & r_2 & r_3 \\
\hline
6 & 0 & 0 \\
4 & 0 & 2 \\
2 & 0 & 4 \\
0 & 0 & 6 \\
3 & 3 & 0 \\
1 & 3 & 2 \\
0 & 6 & 0 \\
\end{array}
\)
\(
\text{sum} =
\frac{6!}{6!0!0!}(1)^6(2)^0(3)^0 +
\frac{6!}{4!0!2!}(1)^4(2)^0(3)^2 +
\frac{6!}{2!0!4!}(1)^2(2)^0(3)^4 +
\frac{6!}{0!0!6!}(1)^0(2)^0(3)^6 +
\frac{6!}{3!3!0!}(1)^3(2)^1(3)^0 +
\frac{6!}{1!3!2!}(1)^1(2)^1(3)^2 +
\frac{6!}{0!6!0!}(1)^0(2)^2(3)^0
\)
\( = 1 + 45 + 135 + 27 + 40 + 360 + 4 = 612 \)
▶️ Answer/Explanation
Ans. (19)
Sol.
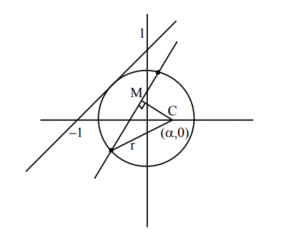
\( x – y + 1 = 0; \; p = r \)
\( \left| \frac{\alpha – 0 + 1}{\sqrt{2}} \right| = r \Rightarrow (\alpha + 1)^2 = 2r^2 \) ….(1)
Now
\( \left( \frac{-3\alpha + 0 – 1}{\sqrt{9 + 4}} \right)^2 + \left( \frac{2}{\sqrt{13}} \right)^2 = r^2 \)
\( \Rightarrow (3\alpha + 1)^2 + 4 = 13r^2 \) ….(2)
(1) & (2) \( \Rightarrow (3\alpha + 1)^2 + 4 = 13 \cdot \frac{(\alpha + 1)^2}{2} \)
\( \Rightarrow 18\alpha^2 + 12\alpha + 2 + 8 = 13\alpha^2 + 26\alpha + 13 \)
\( \Rightarrow 5\alpha^2 – 14\alpha – 3 = 0 \)
\( \Rightarrow 5\alpha^2 – 15\alpha + \alpha – 3 = 0 \Rightarrow 5\alpha(\alpha – 3) + (\alpha – 3) = 0 \)
\( \Rightarrow \alpha = -\frac{1}{5}, \, 3 \)
\( \therefore r = 2\sqrt{2} \)
Now \( \alpha e = 3 \) and \( 2\alpha = 4\sqrt{2} \)
\( \alpha^2 e^2 = 9 \Rightarrow \alpha e = 2\sqrt{2} \Rightarrow \alpha^2 = 8 \)
\( \alpha^2 \left( 1 + \frac{\beta^2}{\alpha^2} \right) = 9 \)
\( \alpha^2 + \beta^2 = 9 \)
\( \beta^2 = 1 \)
\( \therefore 2\alpha^2 + 3\beta^2 = 2(8) + 3(1) = 19 \)
▶️ Answer/Explanation
Ans. (30)
Sol. \(5x^3 – 15x – a = 0\)
\(f(x) = 5x^3 – 15x\)
\(f'(x) = 15x^2 – 15 = 15(x-1)(x + 1)\)
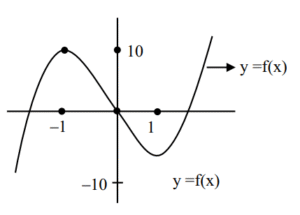
\(a \in (-10, 10)\)
\(\alpha = -10, \beta = 10\)
\(\beta – 2\alpha = 10 + 20 = 30\)
▶️ Answer/Explanation
Sol. \(a(b – c) x^2 + b (c – a) x + c(a – b) = 0\)
\(x = 1\) is root \(\therefore\) other root is 1
\(\alpha + \beta = -\frac{b(c-a)}{2a(b-c)}\)
\(\Rightarrow -bc + ab = 2ab – 2ac\)
\(\Rightarrow 2ac = b(a + c)\)
\(\Rightarrow 2ac = 15b\)
\(\Rightarrow 2ac = 108\)
\(\Rightarrow ac = 54\)
\(a + c = 15\)
\(a^2 + c^2 + 2ac = 225\)
\(a^2 + c^2 = 225 – 108 = 117\)
A : The self-inductance of the coil depends on its geometry.
B : Self-inductance does not depend on the permeability of the medium.
C : Self-induced e.m.f. opposes any change in the current in a circuit.
D : Self-inductance is electromagnetic analogue of mass in mechanics.
E : Work needs to be done against self-induced e.m.f. in establishing the current.
Choose the correct answer from the options given below:
(1) A, B, C, D only
(2) A, C, D, E only
(3) A, B, C, E only
(4) B, C, D, E only
▶️ Answer/Explanation
Sol. Self inductance of coil
\( L = \frac{\mu_0 N^2 A}{2\pi R} \)
The time period of oscillations is \( y\pi \times 10^{-2} \) s, where the value of \( y \) is
(Acceleration due to gravity, \( g = 10 \, \text{m/s}^2 \), density of water = \( 10^3 \, \text{kg/m}^3 \))
(1) 2
(2) 6
(3) 4
(4) 1
▶️ Answer/Explanation
Sol. \( a_{net} = \rho g x – \frac{m g}{L^2} \)
\( T = 2\pi \sqrt{\frac{m}{\rho L^2 g}} \)
where \( m = 10 \, \text{g} \), \( L = 10 \, \text{cm} \), \( \rho = 1000 \, \text{kg/m}^3 \)
Statement-I : The hot water flows faster than cold water.
Statement-II : Soap water has higher surface tension as compared to fresh water.
In the light above statements, choose the correct answer from the options given below
(1) Statement-I is false but Statement II is true
(2) Statement-I is true but Statement II is false
(3) Both Statement-I and Statement-II are true
(4) Both Statement-I and Statement-II are false
▶️ Answer/Explanation
Sol. Hot water is less viscous then cold water.
Surfactant reduces surface tension.
(1) Infra-red radiation
(2) X-rays
(3) Gamma rays
(4) Visible radiation
▶️ Answer/Explanation
Sol. \( \lambda = \frac{h}{p} = \frac{6.63 \times 10^{-34}}{10^{-30} \times 2.21 \times 10^6} \)
\( = 3 \times 10^{-10} \, \text{m} \)
Hence particle will behave as x-ray.
(1) 5R
(2) 3R
(3) 2R
(4) 1.5R
▶️ Answer/Explanation
Ans. (1)
Sol.
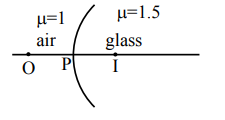
\( PO = u = -x \)
\( PI = v = x \)
\( PO = PI \)
\( \frac{\mu_2}{v} – \frac{\mu_1}{u} = \frac{\mu_2 – \mu_1}{R} \)
\( \frac{1.5}{x} + \frac{1}{x} = \frac{1}{2R} \)
\( \frac{5}{2x} = \frac{1}{2R} \)
\( x = 5R \)
(1) 1/4
(2) 1/8
(3) 4
(4) 8
▶️ Answer/Explanation
Sol. \( N_2 = N_0 e^{-3\lambda t} \)
\( N_1 = N_0 e^{-\lambda t} \)
\( \frac{N_1}{N_2} = e^{\lambda t} \)
\( t_{\text{half life of } n_1} = \frac{\ln 2}{\lambda} \)
\( N_0 e^{-\lambda t} = \frac{N_0}{2} \)
\( \lambda t = \ln 2 \)
\( t = \frac{\ln 2}{\lambda} \)
\( \frac{N_2}{N_1} = \frac{1}{4} \)
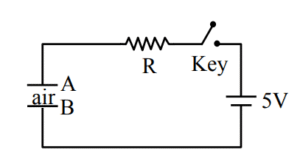
A. There will be no current through resistor \( R \).
B. There will be maximum current in the connecting wires.
C. Potential difference between the capacitor plates A and B is minimum.
D. Charge on the capacitor plates is minimum.
Choose the correct answer from the options given below :
(1) C, D only
(2) B, C, D only
(3) A, C only
(4) A, B, D only
▶️ Answer/Explanation
Ans. (2)
Sol. Initially capacitor behave as a short circuit.
So current will be maximum.
Charge on capacitor will be zero.
Potential difference across capacitor will be zero.
(1) \( L \)
(2) \( L^3 T^{-2} \)
(3) \( L^2 T^{-2} \)
(4) \( L^2 \)
▶️ Answer/Explanation
Sol. Dimension \( [x(t)] = [L] \)
\( [A] = [L] \)
\( [B] = [L] \)
\( [C] = [L T^{-2}] \)
\( [D] = [L] \)
\( \left[ \frac{ABC}{D} \right] = \frac{[L] \times [L] \times [L T^{-2}]}{[L]} = [L^2 T^{-2}] \)
List-I
A. Pressure varies inversely with volume of an ideal gas.
B. Heat absorbed goes partly to increase internal energy and partly to do work.
C. Heat is neither absorbed nor released by a system
D. No work is done on or by a gas
List-II
I. Adiabatic process
II. Isochoric process
III. Isothermal process
IV. Isobaric process
Choose the correct answer from the options given below :
(1) A–I, B–IV, C–II, D–III
(2) A–III, B–I, C–IV, D–II
(3) A–I, B–III, C–II, D–IV
(4) A–III, B–IV, C–I, D–II
▶️ Answer/Explanation
Sol. A \( \rightarrow \frac{1}{P} \propto V \)
\( \Rightarrow PV = \text{constant} \)
\( \Rightarrow nRT = \text{const.} \Rightarrow T = \text{const.} \)
Hence Isothermal III
B \( \rightarrow \) IV
\( W \neq 0 \), \( \Delta U \neq 0 \), \( \Delta Q \neq 0 \) [only isobaric]
C \( \rightarrow \) I \( \Delta Q = 0 \) Adiabatic
D \( \rightarrow \) II \( w = 0 \) Isochoric
III IV I II
A : The torsional constant in moving coil galvanometer has dimensions \( [M L^2 T^{-2}] \)
B : Increasing the current sensitivity may not necessarily increase the voltage sensitivity.
C : If we increase number of turns (\( N \)) to its double (2N), then the voltage sensitivity doubles.
D : MCG can be converted into an ammeter by introducing a shunt resistance of large value in parallel with galvanometer.
E : Current sensitivity of MCG depends inversely on number of turns of coil.
Choose the correct answer from the options given below :
(1) A, B only
(2) A, D, only
(3) B, D, E only
(4) A, B, E only
▶️ Answer/Explanation
Sol. (A) \( \tau = C\theta \Rightarrow [M L^2 T^{-2}] = [C][1] \)
(B) \( C.S = \frac{\theta}{I} = \frac{BNA}{C} \);
\( V.S. = \frac{BNA}{RC} \) [\( R \) also depends on ‘\( N \)’]
(C) \( V.S. \propto \frac{NAB}{CR} \)
\( R \rightarrow NR \)
(D) False [Theory]
(E) E [False] \( C.S \propto N \)
\( \frac{NAB}{C} \)
\( C.S. \Rightarrow = \)
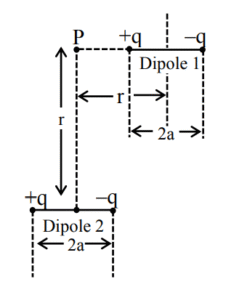
(1) \( \frac{a}{r} \sim 0 \)
(2) \( a \sim 10r \)
(3) \( a \sim 0.5r \)
(4) \( a \sim 3r \)
▶️ Answer/Explanation
Ans. (4)
Sol.
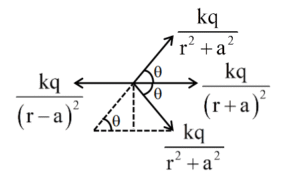
\( \frac{2kq \cos \theta}{(r^2 + a^2)} + \frac{2kq}{(r^2 + a^2)^{3/2}} = \frac{4ra^2}{(r^2 + a^2)^{3/2}} \)
\( \Rightarrow 4r^2 (r^2 + a^2)^3 = (r^2 – a^2)^4 \)
\( \frac{a^3}{r^3} \approx 3 \)
\( \frac{a}{r} \approx 3 \)
But by solving from mathematical software we are getting \( \frac{a}{r} \approx 3 \).
(Latent heat of fusion of lead = \( 2.5 \times 10^4 \, \text{J kg}^{-1} \) and specific heat capacity of lead = \( 125 \, \text{J kg}^{-1} \text{K}^{-1} \))
(1) 20
(2) 15
(3) 10
(4) 5
▶️ Answer/Explanation
Sol. 625 = ms\Delta T + mL
625 = m[125 \times 300 + 2.5 \times 10^4]
625 = m[37500 + 25000]
625 = m[62500]
\( m = \frac{1}{100} \, \text{kg} \)
\( M = 10 \, \text{grams} \)
(1) \( \frac{-h \tan(i – r)}{\tan r} \)
(2) \( \frac{-h \cos(i – r)}{\sin r} \)
(3) \( h \)
(4) \( \frac{-h \sin(i – r)}{\cos r} \)
▶️ Answer/Explanation
Sol. Formula base
\( \frac{-h \sin(i – r)}{\cos r} \)
(1) 1 : \( \sqrt{2} \)
(2) 1 : 3
(3) 1 : 2
(4) 1 : \( \sqrt{3} \)
▶️ Answer/Explanation
Ans. (1)
Sol.
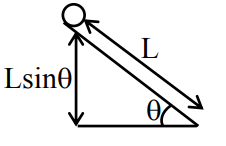
using WET
\( W_g = k_f – k_i \)
\( Mg L \sin \theta = k_f – k_i \)
K.E. in pure rolling
\( \frac{1}{2} m V_{cm}^2 + \frac{1}{2} I \omega^2 \)
\( \frac{1}{2} m V^2 + \frac{1}{2} \frac{2}{5} m R^2 \frac{V^2}{R^2} \)
\( \frac{7}{10} m V^2 \)
\( \frac{7}{10} m g L \sin \theta = \frac{7}{10} m V^2 – 0 \)
\( V^2 \propto \sin \theta \)
\( \frac{v_1^2}{v_2^2} = \frac{\sin 30^\circ}{\sin 45^\circ} \)
\( \frac{1}{\sqrt{2}} \)
A. Total resistance of circuit is 6 \( \Omega \).
B. Current in Ammeter is 1A
C. Potential across AB is 4 Volts.
D. Potential across CD is 4 Volts.
E. Total resistance of the circuit is 8 \( \Omega \).
Choose the correct answer from the options given below:
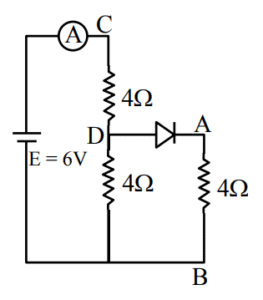
(1) A, B and D only
(2) A, C and D only
(3) B, C and E only
(4) A, B and C only
▶️ Answer/Explanation
Ans. (1)
Sol.
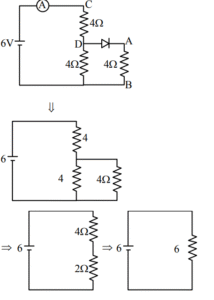
Current through ammeter = 1 A
\( R_{net} = 6 \Omega \)
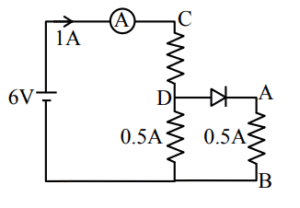
\( V_{AB} = 0.5 \times 4 = 2 \, \text{volt} \)
\( V_{CD} = 1 \times 4 = 4 \, \text{volt} \)
A, B & D are correct
where \( \lambda \) and \( \sigma \) are linear and surface charge density, respectively, \( \frac{\alpha}{\beta} \) represents
(1) charge
(2) electric field
(3) displacement
(4) area
▶️ Answer/Explanation
Sol. \( \phi = \alpha \sigma + \beta \lambda \)
\( [\phi] = [\alpha \sigma] = [\beta \lambda] \)
\( \frac{[\phi]}{[\sigma]} = \frac{[\alpha]}{[\sigma]} \)
\( \frac{[\phi]}{[\lambda]} = \frac{[\beta]}{[\lambda]} \)
\( \frac{[\alpha]}{[\beta]} = \frac{[\phi]/[\sigma]}{[\phi]/[\lambda]} = \frac{\text{Length}}{\text{Area}} = [L] \)
(1) \( \frac{\mu_1}{\mu_2} \frac{1 + \mu_2 – \mu_1}{R_1 – R_2} \frac{R_1}{R_2} \)
(2) \( \frac{\mu_1}{\mu_2} \frac{1 + \mu_2 – \mu_1}{R_1 – R_2} \frac{R_1 \cdot R_2}{R_1 + R_2} \)
(3) \( \frac{\mu_1}{\mu_2} \frac{1 + \mu_2 – \mu_1}{R_1 – R_2} \frac{R_1 \cdot R_2}{2 R_1 – R_2} \frac{R_1 \cdot R_2}{R_1 + R_2} \)
(4) \( (\mu_2 + \mu_1) \frac{\mu_1 – \mu_2}{R} \)
▶️ Answer/Explanation
Ans. (2)
Sol.
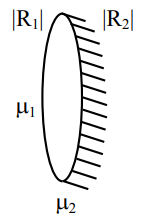
\( \frac{1}{f_{eq}} = \frac{1}{f_L} + \frac{1}{f_m} \)
\( \frac{1}{f_m} = \frac{|\mu_2|}{R_2} – \frac{2}{2} \)
\( \frac{1}{f_L} = \frac{\mu_2 – 1}{R_1} – \frac{\mu_2}{R_2} \)
\( \frac{1}{f_{eq}} = \frac{\mu_2 – 1}{R_1} – \frac{\mu_2}{R_2} + \frac{\mu_2}{2 R_2} \)
\( = \frac{\mu_2 + \mu_1 (\mu_2 – 1)}{R_1 R_2} – \frac{\mu_2 (\mu_2 – 1)}{2 R_1 R_2} \)
For same size of image
\( u = 2f \)
\( u = \frac{\mu_1}{\mu_2} \frac{1 + \mu_2 – \mu_1}{R_1 – R_2} \frac{R_1 \cdot R_2}{R_1 + R_2} \)
\( \vec{E} = 57 \cos[7.5 \times 10^6 t – 5 \times 10^{-3} (3x + 4y)] (-4\hat{i} + 3\hat{j}) \, \text{N/C}. \)
The associated magnetic field in Tesla is-
(1) \( \vec{B} = \frac{57}{3 \times 10^8} \cos[7.5 \times 10^6 t – 5 \times 10^{-3} (3x + 4y)] \hat{k} \)
(2) \( \vec{B} = \frac{57}{3 \times 10^8} \cos[7.5 \times 10^6 t – 5 \times 10^{-3} (3x + 4y)] \hat{k} \)
(3) \( \vec{B} = -\frac{57}{3 \times 10^8} \cos[7.5 \times 10^6 t – 5 \times 10^{-3} (3x + 4y)] \hat{k} \)
(4) \( \vec{B} = -\frac{57}{3 \times 10^8} \cos[7.5 \times 10^6 t – 5 \times 10^{-3} (3x + 4y)] \hat{k} \)
▶️ Answer/Explanation
Sol.
\( \vec{K} = 3\hat{i} + 4\hat{j} \)
\( |\vec{K}| = \hat{K} = \frac{3\hat{i} + 4\hat{j}}{5} \)
\( \vec{E} = \frac{4\hat{i} – 3\hat{j}}{5} \)
\( \vec{B} = \hat{K} \times \vec{E} \)
\( \vec{B} = -\hat{Z} \)
\( B_0 = \frac{E_0}{C} = \frac{57}{3 \times 10^8} \)
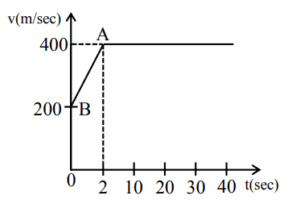
(2) 6
(3) 3
(4) 12
▶️ Answer/Explanation
Ans. (4)
Sol. Total Area under curve.
(1) 2.0 cm
(2) 0.5 cm
(3) 1.5 cm
(4) 1.0 cm
▶️ Answer/Explanation
Ans. (4)
Sol.
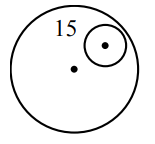
mass of disc = \( m \)
mass of cut part = \( \frac{m}{16} \)
\( X_{com} = \frac{m \times 0 – \frac{m}{16} \times 15}{m – \frac{m}{16}} \)
\( = 1 \, \text{cm}. \)
(Take \( \frac{1}{4 \pi \epsilon_0} = 9 \times 10^9 \, \text{Nm}^2 \text{C}^{-2} \) and universal gravitational constant as \( 6.67 \times 10^{-11} \, \text{Nm}^2 \text{kg}^{-2} \))
▶️ Answer/Explanation
Sol. \( \frac{9 \times 10^9 \times 6.67 \times 10^{-19} \times 9.6 \times 10^{-10}}{6.67 \times 10^{-11} \times 19.2 \times 10^{-27} \times 9 \times 10^{-27}} \)
\( \frac{1}{2} \times 10^{45} \)
Charge is not integral multiple of electron.
▶️ Answer/Explanation
Sol. \( \vec{A} \cdot \vec{B} = 0 \)
\( 4 – 6n + 8p = 0 \)
\( |\vec{A}| = |\vec{B}| \)
\( 4 + 9n^2 + 4 = 4 + 4 + 16p^2 \)
\( 9n^2 = 16p^2 \)
\( p = \pm \frac{3}{4} n \)
\( 4 – 6n + 6n = 0 \)
\( 12n = 4 \)
\( n = \frac{1}{3} \)
▶️ Answer/Explanation
Sol. \( \gamma = \frac{3}{2} \)
\( T V^{\gamma – 1} = C \)
\( \frac{T}{T_0} = \left( \frac{V_0}{V} \right)^{0.5} \)
\( 273 \left( \frac{1}{4} \right)^{0.5} = 273 \times \frac{1}{2} = 136.5 \)
\( \Delta T = 273 \)
▶️ Answer/Explanation
Sol. \( \int_0^4 x^2 (10 – x) dx + \int_0^2 y^2 dy \)
\( = \left[ \frac{10x^3}{3} – \frac{x^4}{4} \right]_0^4 + \left[ \frac{y^3}{3} \right]_0^2 \)
\( = \frac{640}{3} – 64 + \frac{8}{3} \)
\( = 152 \)
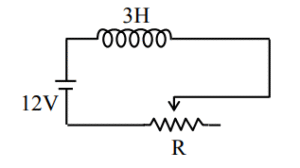
▶️ Answer/Explanation
Ans. (3)
Sol. \( \epsilon = -L \frac{dI}{dt} – IR = 0 \)
\( 12 – 3 \times (-8) – I \times 12 = 0 \)
\( I = 3 \)
(1) Palladium
(2) Iridium
(3) Osmium
(4) Platinum
▶️ Answer/Explanation
Explanation:
Palladium \(\Rightarrow\) 5\(^{th}\) period
Iridium, Osmium, Platinum \(\Rightarrow\) 6\(^{th}\) Period
Given: Rydberg constant \( R_H = 10^5 \, \text{cm}^{-1} \), \( h = 6.6 \times 10^{-34} \, \text{J s} \), \( c = 3 \times 10^8 \, \text{m/s} \)
(1) Paschen series, \(\infty \rightarrow 3\)
(2) Lyman series, \(\infty \rightarrow 1\)
(3) Balmer series, \(\infty \rightarrow 2\)
(4) Paschen series, \(5 \rightarrow 3\)
▶️ Answer/Explanation
Explanation:
\(\lambda = 900 \, \text{nm}\)
H-atom (\(Z = 1\))
\(= 9 \times 10^{-5} \, \text{cm}\)
\(R_H = 10^5 \, \text{cm}^{-1}\)
Rydberg eq. = \(\frac{1}{\lambda} = R_H Z^2 \left(\frac{1}{n_1^2} – \frac{1}{n_2^2}\right)\)
\(\Rightarrow \frac{1}{\lambda} = \frac{R_H}{n_1^2} – \frac{R_H}{n_2^2}\)
\(\Rightarrow \frac{1}{9 \times 10^{-5} \, \text{cm} \times 10^5 \, \text{cm}^{-1}} = \frac{1}{n_1^2} – \frac{1}{n_2^2}\)
\(\Rightarrow \frac{1}{9} = \frac{1}{n_1^2} – \frac{1}{n_2^2}\)
It is possible when \(n_1 = 3\), \(n_2 = \infty\)
Possible series : \(\infty \rightarrow 3\)
(1) PH\(_3\) shows lower proton affinity than NH\(_3\).
(2) PF\(_3\) exists but NF\(_5\) does not.
(3) NO\(_2\) can dimerise easily.
(4) SO\(_2\) can act as an oxidizing agent, but not as a reducing agent.
▶️ Answer/Explanation
Explanation:
SO\(_2\) can oxidise as well as reduce.
Hence it can act as both oxidising and reducing agent.
(Given \(K_f = 1.86 \, \text{K kg mol}^{-1}\))
(1) [Cr(NH\(_3\))\(_6\)] Cl\(_3\)
(2) [Cr(NH\(_3\))\(_4\)Cl\(_2\)] Cl
(3) [Cr(NH\(_3\))\(_5\)Cl] Cl\(_2\)
(4) [Cr(NH\(_3\))\(_3\)Cl\(_3\)]
▶️ Answer/Explanation
Ans. (3)
Explanation:
Given : \(\Delta T_f = 0.558^\circ\)C
\(K_f = \frac{\text{kg}}{\text{mol}} \times 1.86\)
0.1 m aq. sol.
\(\Rightarrow \Delta T_f = i \times K_f \times m\)
\(\Rightarrow 0.558 = i \times 1.86 \times 0.1\)
\(\Rightarrow i = 3\)
In the above diagram, the standard electrode potentials are given in volts (over the arrow).
The value of \(E^\Theta_{\text{FeO}_4^{2-}/\text{Fe}}\) is
(1) 1.7 V
(2) 1.2 V
(3) 2.1 V
(4) 1.4 V
▶️ Answer/Explanation
Ans. (1)
Explanation:
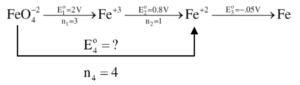
\(\Delta G^\circ_4 = \Delta G^\circ_1 + \Delta G^\circ_2\)
\(\Rightarrow -n F E^\circ_4 = -n F E^\circ_1 – n F E^\circ_2\)
\(\Rightarrow E^\circ_4 = \frac{3 \times 2.0 + 1 \times 0.8}{4}\)
\(\Rightarrow E^\circ_4 = \frac{6.8}{4} \, \text{V}\)
\(\Rightarrow E^\circ_4 = 1.7 \, \text{V}\)
LIST-I
Name reaction
A. Swarts reaction
B. Sandmeyer’s reaction
C. Wurtz Fittig reaction
D. Finkelstein reaction
LIST-II
Product obtainable
I. Ethyl benzene
II. Ethyl iodide
III. Cyanobenzene
IV. Ethyl fluoride
Choose the correct answer from the options given below:
(1) A-II, B-III, C-I, D-IV
(2) A-IV, B-I, C-III, D-II
(3) A-IV, B-III, C-I, D-II
(4) A-II, B-I, C-III, D-IV
▶️ Answer/Explanation
Ans. (3)
Explanation: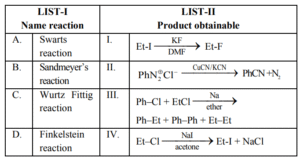
Statement I: Fructose does not contain an aldehydic group but still reduces Tollen’s reagent
Statement II : In the presence of base, fructose undergoes rearrangement to give glucose.
In the light of the above statements, choose the correct answer from the options given below
(1) Statement I is false but Statement II is true
(2) Both Statement I and Statement II are true
(3) Both Statement I and Statement II are false
(4) Statement I is true but Statement II is false
▶️ Answer/Explanation
Ans. (2)
Explanation: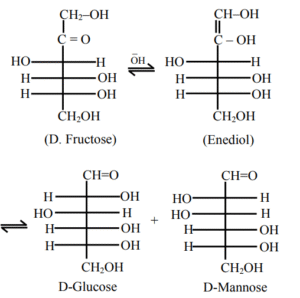
Given : \(N_A = 6.02 \times 10^{23} \, \text{mol}^{-1}\)
(1) 196.2 mg
(2) 98.3 mg
(3) 150.4 mg
(4) 48.2 mg
▶️ Answer/Explanation
Explanation:
\(\text{initial} = \frac{x \times 10^{-3}}{44} \, \text{(moles)}\)
\(\text{removal} = \frac{10^{21}}{6.02 \times 10^{23}} \, \text{(moles)}\)
\(\text{left} = \text{initial} – \text{removed} \, \text{(moles)}\)
\(\frac{x \times 10^{-3}}{44} – \frac{10^{21}}{6.02 \times 10^{23}} = 2.8 \times 10^{-3}\)
\(\Rightarrow x = 196.2 \, \text{mg}\)
(1) \(\frac{\Delta H_{\text{melting}}}{383 \, \text{K}} + \frac{\Delta H_{\text{boiling}}}{268 \, \text{K}} + \int_{273}^{373} C_p \, dT\)
(2) \(\int_{268}^{273 \, \text{K}} \frac{C_{p,m}}{T} \, dT + \frac{\Delta H_{\text{fusion}}}{T_f} + \frac{\Delta H_{\text{vaporisation}}}{T_b}\)
\(\quad + \int_{273}^{373 \, \text{K}} \frac{C_{p,m}}{T} \, dT + \int_{373}^{383 \, \text{K}} \frac{C_{p,m}}{T} \, dT\)
(3) \(\int_{268}^{383 \, \text{K}} \frac{q_{\text{rev}}}{T} + C_p \, dT\)
(4) \(\int_{268}^{273 \, \text{K}} \frac{\Delta H_{\text{fusion}}}{T} + \frac{\Delta H_{\text{vaporisation}}}{T_b} + \frac{C_{p,m}}{T} \, dT\)
\(\quad + \int_{273}^{373 \, \text{K}} \frac{C_{p,m}}{T} \, dT + \int_{373}^{383 \, \text{K}} \frac{C_{p,m}}{T} \, dT\)
▶️ Answer/Explanation
Explanation:
Ice \(\rightarrow\) Ice
Water \(\rightarrow\) Water
Water \(\rightarrow\) Water vapour
268 K 273 K
273 K 273 K 373 K 383 K
(1) (2) (3) (4) (5)
\(\Delta S_{\text{overall}} = \Delta S_1 + \Delta S_2 + \Delta S_3 + \Delta S_4 + \Delta S_5\)
\(\Delta S_2 = \frac{\Delta H_{\text{fusion}}}{273}\) \(T_f = 273 \, \text{‘K’}\)
\(\Delta S_3 = \int_{273}^{373} \frac{C_{p,m}}{T} \, dT\)
\(\Delta S_4 = \frac{\Delta H_{\text{vaporisation}}}{373}\) \(T_b = 373 \, \text{‘K’}\)
\(\Delta S_5 = \int_{373}^{383} \frac{C_{p,m}}{T} \, dT\)
Answer = (2)
(1) \(t_{2g}^6 e_g^1\)
(2) \(t_{2g}^3 e_g^0\)
(3) \(t_{2g}^5 e_g^2\)
(4) \(t_{2g}^4 e_g^3\)
▶️ Answer/Explanation
Ans. (3)
Explanation: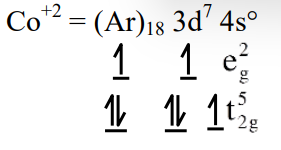
(1) [Co(NH\(_3\))\(_3\)Cl\(_3\)]
(2) [Co(NH\(_3\))\(_4\)Cl\(_2\)]\(^+\)
(3) [Co(en)\(_3\)]\(^{3+}\)
(4) [Co(en)\(_2\)Cl\(_2\)]\(^+\)
▶️ Answer/Explanation
Explanation:
Ma\(_3\)b\(_3\) type complexes show Facial – Meridional isomerism
(i) [Co(NH\(_3\))\(_3\)Cl\(_3\)] \(\Rightarrow\) Ma\(_3\)b\(_3\)
(ii) [Co(NH\(_3\))\(_4\)Cl\(_2\)]\(^+\) \(\Rightarrow\) Ma\(_4\)b\(_2\)
(iii) [Co(en)\(_3\)]\(^{3+}\) \(\Rightarrow\) M(AA)\(_3\)
(iv) [Co(en)\(_2\)Cl\(_2\)]\(^+\) \(\Rightarrow\) M(AA)\(_2\)b\(_2\)
a, b, = NH\(_3\), Cl\(^-\)
AA = en
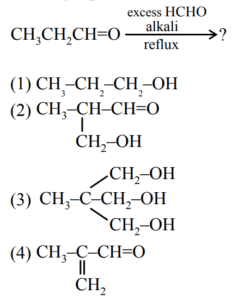
▶️ Answer/Explanation
Ans. (3)
Explanation:
This is an example of Tollens reaction i.e. multiple cross aldol followed by cross Cannizzaro reaction 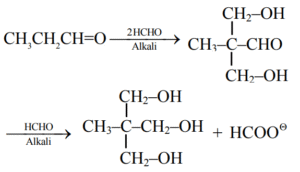
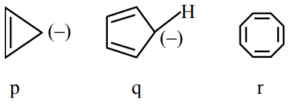
(1) q > r > p
(2) r > q > p
(3) q > p > r
(4) p > q > r
▶️ Answer/Explanation
Ans. (1)
Explanation:
q is aromatic r is nonaromatic p is antiaromatic
(1) 4
(2) 2
(3) 5
(4) 3
▶️ Answer/Explanation
Ans. (4)
Explanation: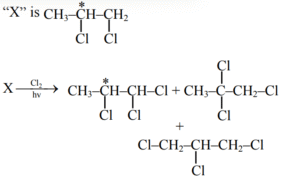
(Given molar mass in g mol\(^{-1}\) of C, H, O, Br are 12, 1, 16, 80 respectively)
(1) 10.22 g
(2) 6.0 g
(3) 4.0 g
(4) 20.44 g
▶️ Answer/Explanation
Ans. (1)
Explanation: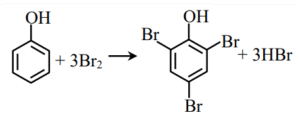
Moles of phenol = \(\frac{2}{94} = 0.021\)
\(\therefore\) Moles of bromine = \(0.021 \times 3 = 0.064\)
\(\therefore\) Mass of bromine = \(0.064 \times 160 = 10.22 \, \text{g}\)
(1) V\(^{2+}\), Cr\(^{3+}\), Mn\(^{3+}\)
(2) Zn\(^{2+}\), V\(^{3+}\), Fe\(^{3+}\)
(3) Ti\(^{4+}\), V\(^{4+}\), Mn\(^{2+}\)
(4) Sc\(^{3+}\), Ti\(^{3+}\), Cr\(^{2+}\)
▶️ Answer/Explanation
Explanation:
(1) V\(^{2+}\) (Violet), Cr\(^{3+}\) (Violet), Mn\(^{3+}\) (Violet)
(2) Zn\(^{2+}\) (Colourless), V\(^{3+}\) (Green), Fe\(^{3+}\) (Yellow)
(3) Ti\(^{4+}\) (Colourless), V\(^{4+}\) (Blue), Mn\(^{2+}\) (Pink)
(4) Sc\(^{3+}\) (Colourless), Ti\(^{3+}\) (Purple), Cr\(^{2+}\) (Blue)
Statement I : In Lassaigne’s test, the covalent organic molecules are transformed into ionic compounds.
Statement II : The sodium fusion extract of an organic compound having N and S gives prussian blue colour with FeSO\(_4\) and Na\(_4\)[Fe(CN)\(_6\)]
In the light of the above statements, choose the correct answer from the options given below
(1) Both Statement I and Statement II are true
(2) Both Statement I and Statement II are false
(3) Statement I is false but Statement II is true
(4) Statement I is true but Statement II is false
▶️ Answer/Explanation
Explanation:
The sodium fusion extract of organic compound having N & S gives blood red colour with FeSO\(_4\) and Na\(_4\)[Fe(CN)\(_6\)]
Given : \(K_{sp}[A(OH)_2] = 9 \times 10^{-10}\) and \(K_{sp}[B(OH)_3] = 27 \times 10^{-18}\) at 298 K.
(1) B(OH)\(_3\) will precipitate before A(OH)\(_2\)
(2) A(OH)\(_2\) and B(OH)\(_3\) will precipitate together
(3) A(OH)\(_2\) will precipitate before B(OH)\(_3\)
(4) Both A(OH)\(_2\) and B(OH)\(_3\) do not show precipitation with NH\(_4\)OH
▶️ Answer/Explanation
Explanation:
Condition for precipitation \(Q_{ip} > K_{sp}\)
For [A(OH)\(_2\)]
\([A^{2+}][OH^-]^2 > 9 \times 10^{-10}\)
\([A^{2+}] = 1 \, \text{M}\)
\(\Rightarrow [OH^-] > 3 \times 10^{-5} \, \text{M}\)
For [B(OH)\(_3\)]
\([B^{3+}][OH^-]^3 > 27 \times 10^{-18}\)
\([B^{3+}] = 1 \, \text{M}\)
\(\Rightarrow [OH^-] > 3 \times 10^{-6} \, \text{M}\)
So, B(OH)\(_3\) will precipitate before A(OH)\(_2\)
LIST-I
(Classification of molecules based on octet rule)
A. Molecules obeying octet rule
B. Molecules with incomplete octet
C. Molecules with incomplete octet with odd electron
D. Molecules with expanded octet
LIST-II
(Example)
I. NO, NO\(_2\)
II. BCl\(_3\), AlCl\(_3\)
III. H\(_2\)SO\(_4\), PCl\(_5\)
IV. CCl\(_4\), CO\(_2\)
Choose the correct answer from the options given below :
(1) A-IV, B-II, C-I, D-III
(2) A-III, B-II, C-I, D-IV
(3) A-IV, B-I, C-III, D-II
(4) A-II, B-IV, C-III, D-I
▶️ Answer/Explanation
Explanation:
(A) A \(\rightarrow\) IV
(B) B \(\rightarrow\) II
(C) C \(\rightarrow\) I
(D) D \(\rightarrow\) III
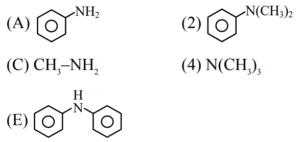
Choose the correct answer from the options given below :
(1) B and D only
(2) C and D only
(3) A, B and E only
(4) A, C and E only
▶️ Answer/Explanation
Ans. (4)
Explanation:
B and D are 3\(^\circ\) amine which does not have replaceable H on N, So does not react.
[The degree of ionization of ethylamine can be neglected with respect to unity.]
▶️ Answer/Explanation
Ans. (7)
Explanation: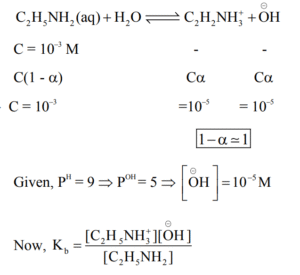
\(\Rightarrow K_b = \frac{10^{-5} \times 10^{-5}}{10^{-3}} = 10^{-7}\)
(Given molar mass in g mol\(^{-1}\) of Ba : 137, S : 32, O : 16)
▶️ Answer/Explanation
Explanation:
Millimoles of BaSO\(_4\) = \(\frac{466}{233} = 2 \, \text{mmol}\)
\(\%S = \frac{466 \times 32}{233} \times \frac{100}{160} = 40\%\)
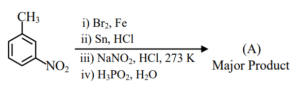
Molar mass of product (A) is ______ g mol\(^{-1}\).
(Given molar mass in g mol\(^{-1}\) of C : 12, H : 1, O : 16, Br : 80, N : 14, P : 31)
▶️ Answer/Explanation
Ans. (171)
Explanation: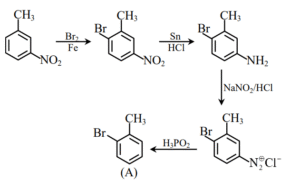
Molar mass of product (C\(_7\)H\(_7\)Br) (A) is 171 g mol\(^{-1}\)
\(2\text{N}_2\text{O}_5\text{(g)} \rightarrow 2\text{N}_2\text{O}_4\text{(g)} + \text{O}_2\text{(g)}\)
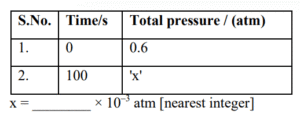
Given : Rate constant for the reaction is \(4.606 \times 10^{-2} \, \text{s}^{-1}\).
▶️ Answer/Explanation
Ans. (900)
Explanation:
NTA. (897)
\(K = 2 \times 4.606 \times 10^{-2} \, \text{s}^{-1}\)
\(2\text{N}_2\text{O}_5\text{(g)} \rightarrow 2\text{N}_2\text{O}_4\text{(g)} + \text{O}_2\text{(g)}\)
\(P_i\) 0.6 0 0
\(P_f\) \(0.6 – P\) \(P\) \(P/2\)
\(P_{\text{total}} = \frac{P}{2} + 0.6\)
\(2 \times 4.606 \times 10^{-2} = \frac{2.303}{100} \log \frac{0.6}{0.6 – P}\)
\(10 \frac{0.6}{0.6 – P} = 4 \log \frac{0.6}{0.6 – P}\)
\(\frac{0.6}{0.6 – P} = 10^{\frac{4}{10}}\)
\(\frac{0.6}{0.6 – P} = 10^{0.4}\)
\(0.6 = (0.6 – P) \times 10^{0.4}\)
\(P = 0.6 (1 – 10^{-0.4})\)
\(P = 0.6 \times 0.599\)
\(P = 0.3594\)
\(P_{\text{total}} = 0.6 + 0.1797 = 0.7797 \, \text{atm}\)
\(x = 779.7 \times 10^{-3} \, \text{atm}\)
Ans. 900
▶️ Answer/Explanation
Explanation:
\(\Delta H^\circ_{\text{rxn}} = 55 \, \text{kJ/mol}\), \(T = 298 \, \text{K}\)
\(\Delta S^\circ_{\text{rxn}} = 175 \, \text{J/mol}\)
\(\Delta G^\circ_{\text{rxn}} = \Delta H^\circ – T \Delta S^\circ\)
\(\Rightarrow \Delta G^\circ_{\text{rxn}} = 55000 \, \text{J/mol} – 298 \times 175 \, \text{J/mol}\)
\(\Rightarrow \Delta G^\circ_{\text{rxn}} = 55000 – 52150\)
\(\Rightarrow \Delta G^\circ_{\text{rxn}} = 2850 \, \text{J/mol}\)
