Which of the following statements is incorrect for antibiotics?
(A) An antibiotic should be effective in low concentrations.
(B) An antibiotic must be a product of metabolism.
(C) An antibiotic should promote the growth or survival of microorganisms
(D) An antibiotic is a synthetic substance produced as a structural analogue of naturally occurring antibiotic.
▶️ Answer/Explanation
Answer: (C) An antibiotic should promote the growth or survival of microorganisms
Explanation:
Antibiotics is an antimicrobial substance, it inhibits the growth of microbes. It should not promote growth or survival of microorganism. Low concentration of antibiotics is enough to increase antimicrobial resistance.
▶️ Answer/Explanation
’25 volume’ hydrogen peroxide means
(A) 1 L marketed solution contains 250g of H₂O₂.
(B) 1 L marketed solution contains 25g of H₂O₂.
(C) 100 mL marketed solution contains 25 g of H₂O₂.
(D) 1 L marketed solution contains 75 g of H₂O₂.
▶️ Answer/Explanation
Answer: (D) 1 L marketed solution contains 75 g of H₂O₂.
Explanation:
Strength in g/L = N × Eq.wt = (V.S/5.6) × Eq.wt = (25/5.6) × 17 = 75.89 ≈ 76 g/L
Match List I with List II
| List-I (Elements) | List-II (Colour imparted to the flame) |
|---|---|
| A. K | I. Brick Red |
| B. Ca | II. Violet |
| C. Sr | III. Apple Green |
| D. Ba | IV. Crimson Red |
Choose the correct answer from the options given below:
(A) A- IV, B-III, C-II, D- I
(B) A-II, B-I, C-III, D-IV
(C) A-II, B-IV, C-I, D-III
(D) A-II, B-I, C-IV, D-III
▶️ Answer/Explanation
Answer: (D) A-II, B-I, C-IV, D-III
Explanation:
K – Violet
Ca – Brick Red
Sr – Crimson Red
Ba – Apple Green
Match List I with List II
| List-I (Cations) | List-II (Group reagents) |
|---|---|
| A. Pb²⁺, Cu²⁺ | I. H₂S gas in presence of dilute HCl |
| B. Al³⁺, Fe³⁺ | II. (NH₄)₂CO₃ in presence of NH₄OH |
| C. Co²⁺, Ni²⁺ | III. NH₄OH in presence of NH₄Cl |
| D. Ba²⁺, Ca²⁺ | IV. H₂S in presence of NH₄OH |
Choose the correct answer from the options given below:
(A) A- III, B-I, C-IV, D- II
(B) A-I, B-III, C-II, D-IV
(C) A-I, B-III, C-IV, D-II
(D) A-IV, B-II, C-III, D-I
▶️ Answer/Explanation
Answer: (C) A-I, B-III, C-IV, D-II
Explanation:
Pb²⁺, Cu²⁺ – H₂S gas in presence of dilute HCl
Al³⁺, Fe³⁺ – NH₄OH in presence of NH₄Cl
Co²⁺, Ni²⁺ – H₂S in presence of NH₄OH
Ba²⁺, Ca²⁺ – (NH₄)₂CO₃ in presence of NH₄OH
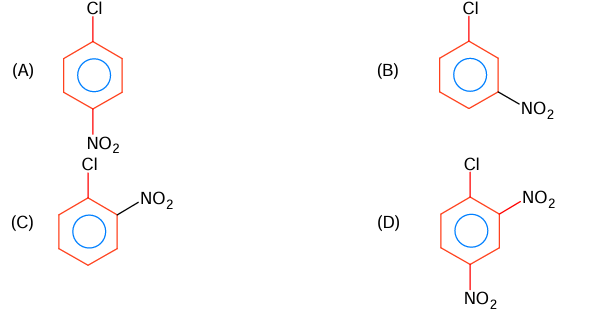
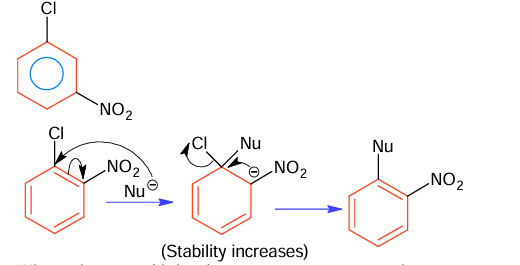
The correct order in aqueous medium of basic strength in case of methyl substituted amines is:
(A) Me₂NH > MeNH₂ > Me₃N > NH₃
(B) Me₂NH > Me₃N > MeNH₂ > NH₃
(C) NH₃ > Me₃N > MeNH₂ > Me₂NH
(D) Me₃N > Me₂NH > MeNH₂> NH₃
▶️ Answer/Explanation
Answer: (A) Me₂NH > MeNH₂ > Me₃N > NH₃
Explanation:
Basic strength order of methyl substituted amines are 2° > 1° > 3° > NH₃
Basic strength in aqueous solutions decided by (i) I-effect (ii) H-bonding (iii) steric factor.
Reaction of thionyl chloride with white phosphorus forms a compound [A], which on hydrolysis gives [B], a diabasic acid. [A] and [B] are respectively.
(A) P₄O₆ and H₃PO₃
(B) POCl₃ and H₃PO₄
(C) PCl₃ and H₃PO₃
(D) PCl₅ and H₃PO₄
▶️ Answer/Explanation
Answer: (C) PCl₃ and H₃PO₃
Explanation:
P₄ + SOCl₂ → PCl₃ + SO₂ + S₂Cl₂ (white P)
PCl₃ + H₂O → H₃PO₃ + 3HCl
H₃PO₃ is a dibasic acid.
The radius of the 2nd orbit of Li²⁺ is x. The expected radius of the 3rd orbit of Be³⁺ is
(A) \(\frac{27x}{16}\)
(B) \(\frac{16x}{27}\)
(C) \(\frac{4x}{9}\)
(D) \(\frac{9x}{4}\)
▶️ Answer/Explanation
Answer: (A) \(\frac{27x}{16}\)
Explanation:
For Li²⁺: r₂ = x = (r₁)ₕ × (2²/3) = 4(r₁)ₕ/3 ⇒ (r₁)ₕ = 3x/4
For Be³⁺: r₃ = (r₁)ₕ × (3²/4) = (r₁)ₕ × 9/4 = (3x/4) × (9/4) = 27x/16
Inert gases have positive electron gain enthalpy. Its correct order is
(A) He < Ne < Kr < Xe
(B) He < Xe < Kr < Ne
(C) Xe < Kr < Ne < He
(D) He < Kr < Xe < Ne
▶️ Answer/Explanation
Answer: (B) He < Xe < Kr < Ne
Explanation:
(+ve) electron gain enthalpy of inert gases because octet is complete. When size increase (+ve) electron gain enthalpy decreases.
He (+48 kJ/mole) < Xe (+77 kJ/mole) < Kr (+96 kJ/mole) < Ne (+116 kJ/mole)
He has smallest size therefore highest tendency to accept e⁻.
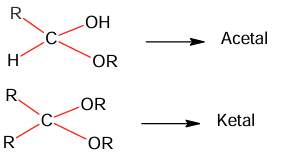
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: Acetal / Ketal is stable in basic medium.
Reason R: The light leaving tendency of alkoxide ion gives the stability to acetal / ketal in basic medium.
In the light of the above statements, choose the correct answer from the options given below:
(A) A is true but R is false
(B) A is false but R is true
(C) Both A and R are true but R is NOT the correct explanation of A
(D) Both A and R are true and R is the correct explanation of A
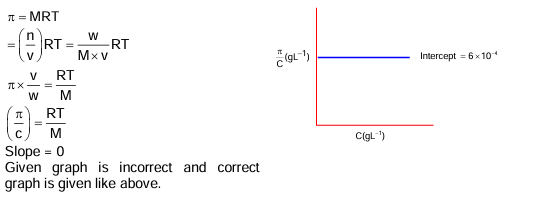
A cubic solid is made up of two elements X and Y. Atoms of X are present on every alternate corner and one at the center of cube. Y is at 1/3 of the total faces. The empirical formula of the compound is.
(A) X₂Y₁.₅
(B) XY₂.₅
(C) X₂.₅Y
(D) X₁.₅Y₂
▶️ Answer/Explanation
Answer: (D) X₁.₅Y₂ or X₃Y₂
Explanation:
Atoms of X = (1/8 × 4) + (1 × 1) = 3/2
Atoms of Y = (1/3 × 6) × 1/2 = 1
Formula = X₃/₂Y₁ = X₃Y₂
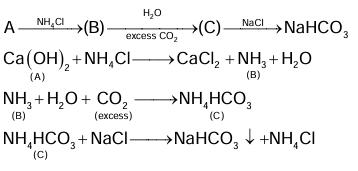
Compound A reacts with NH₄Cl and forms a compound B. Compound B reacts with H₂O and excess of CO₂ to form compound C which on passing through or reaction with saturated NaCl solution forms sodium hydrogen carbonate. Compound A, B and C, are respectively.
(A) Ca(OH)₂, NH₃, NH₄HCO₃
(B) CaCl₂, NH₃, NH₄HCO₃
(C) Ca(OH)₂, NH₄⁺, (NH₄)₂CO₃
(D) CaCl₂, NH₄⁺, (NH₄)₂CO₃
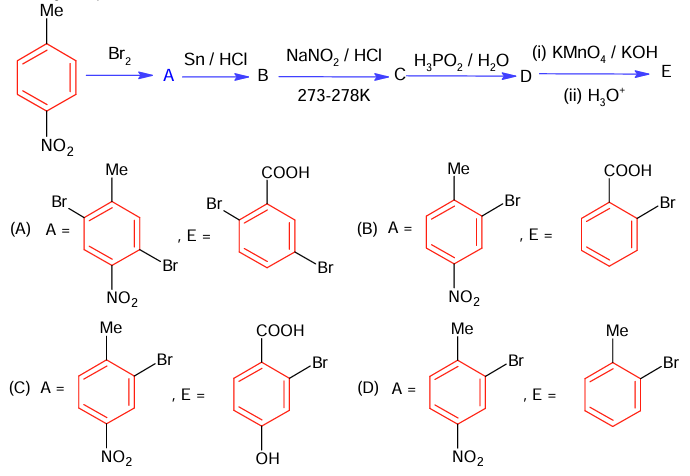
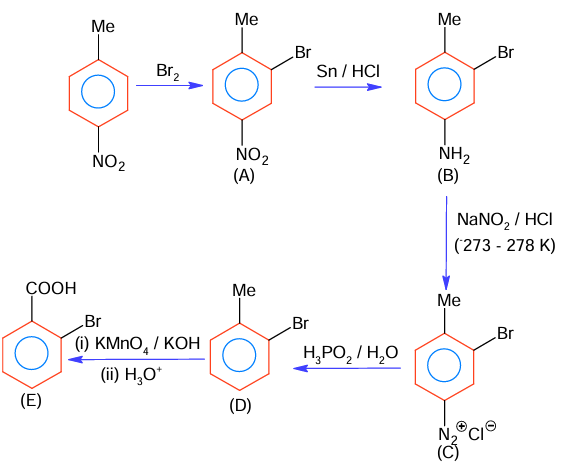
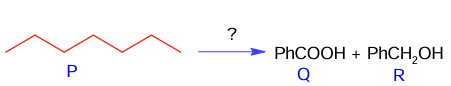
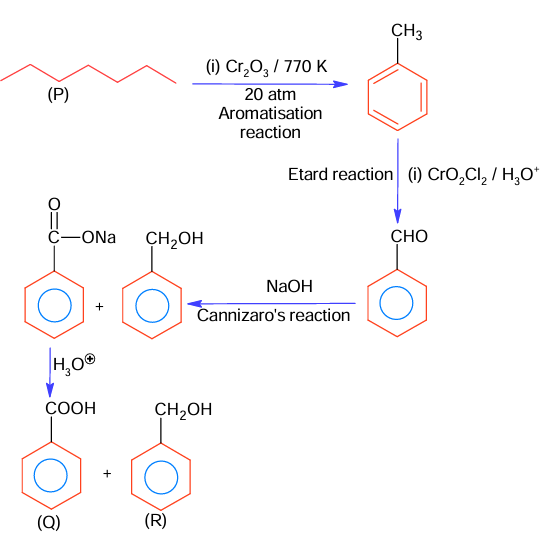
The correct sequence of reagent for the preparation of Q and R is
(A) (i) KMnO₄, OH⁻ ; (ii) Mo₂O₃, Δ ; (iii) NaOH ; (iv) H₃O⁺
(B) (i) CrO₂Cl₂, H₃O⁺, (ii) Cr₂O₃, 770K , 20 atm (iii) NaOH ; (iv) H₃O⁺
(C) (i) Cr₂O₃, 770K , 20 atm; (ii) CrO₂Cl₂, H₃O⁺, (iii) NaOH ; (iv) H₃O⁺
(D) (i) Mo₂O₃, Δ ; (ii) CrO₂Cl₂, H₃O⁺, (iii) NaOH ; (iv) H₃O⁺
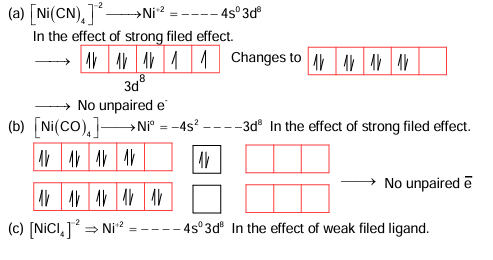
The number of paramagnetic species from the following is ______.
\[ \left[ Ni(CN)_4 \right]^{2-}, \left[ Ni(CO)_4 \right], \left[ NiCl_4 \right]^{2-} \]
\[ \left[ Fe(CN)_6 \right]^{4-}, \left[ Cu(NH_3)_4 \right]^{2+} \]
\[ \left[ Fe(CN)_6 \right]^{3-} \text{ and } \left[ Fe(H_2O)_6 \right]^{2+} \]
Consider the cell
\[ Pt(s) | H_2(g)(1atm)| H_{(aq)}^+, [H^+] = 1) || Fe_{(aq)}^{3+}, Fe_{(aq)}^{2+} | Pt(s) \]
Given \( E^0 \) \[ Fe^{3+/Fe^{2+}} = 0.771 \, V \text{ and } E^0 = 0V, T = 298K \]
If the potential of the cell is 0.712 V, the ratio of concentration of \( Fe^{2+} \) to \( Fe^{3+} \) is ______ (Nearest integer)
▶️ Answer/Explanation
\[ E = E^0 – \frac{0.059}{n} \log \frac{\left[ Fe^{2+} \right]}{\left[ Fe^{3+} \right]} \]
\[ 0.712 = 0.771 – 0.059 \log \frac{\left[ Fe^{2+} \right]}{\left[ Fe^{3+} \right]} \]
\[ 0.059 \log \frac{\left[ Fe^{2+} \right]}{\left[ Fe^{3+} \right]} = 0.059 \]
\[ \log \frac{\left[ Fe^{2+} \right]}{\left[ Fe^{3+} \right]} = 1 \]
\[ \frac{\left[ Fe^{2+} \right]}{\left[ Fe^{3+} \right]} = 10 \]
A litre of buffer solution contains 0.1 mole of each of NH\(_3\) and NH\(_4\)Cl. On the addition of 0.02 mole of HCl by dissolving gaseous HCl, the pH of the solution is found to be ______×10\(^{-3}\)(Nearest integer)
Given: pKb(NH\(_3\)) = 4.745, log2 = 0.301, log3 = 0.477, T = 298 K
An athlete is given 100g of glucose (C\(_6\)H\(_{12}\)O\(_6\)) for energy. This is equivalent to 1800 kJ of energy. The 50% of this energy gained is utilized by the athlete for sports activities at the event. In order to avoid storage of energy, the weight of extra water be would need to perspire is ______ g (Nearest integer)
Given: The enthalpy of evaporation of water is 45 kJ mol\(^{-1}\), Molar mass of C, H & O are 12, 1 and 16 g mol\(^{-1}\).
For the first order reaction A→B, the half life is 30 min. The time taken for 75% completion of the reaction is ______ min. (Nearest integer)
Given: log2 = 0.3010, log3 = 0.4771, log5 = 0.6989
▶️ Answer/Explanation
\[t1/2=30 min
k = \frac{0.693}{t_{1/2}} = \frac{0.693}{30} \text{ min}^{-1} \]
For 75% completion, \[ t = \frac{2.303}{k} \log \frac{100}{25} = \frac{2.303 \times 30}{0.693} \log 4 \]
\[ = \frac{69.09}{0.693} \times 0.602 = 99.7 \times 0.602 = 60 \text{ min} \]
The density of a monobasic strong acid (Molar mass 24.2 g/mol) is 1.21 kg/L. The volume of its solution required for the complete neutralization of 25mL of 0.24 NaOH is ______ ×10\(^{-2}\)mL (Nearest integer)
▶️ Answer/Explanation
d = 1.21kg/L = 1.21g/mℓ
Molar mass = 24.2 g / mole
Millimole of acid = millimole of base
= 25 × 0.24 = 6
Mass of acid = \(\frac{mm}{1000}\) × molar mass
= \(\frac{6 × 24.2}{1000}\) g
∴ Volume = \(\frac{mass}{density}\)
= \(\frac{6 × 24.2}{1.21} × 10^{-3}\) = \(\frac{145.2}{1.21} × 10^{-3}\)
= \(120 × 10^{-3}\) mℓ
= \(12 × 10^{-2}\) mℓ
In sulphur estimation, 0.471 g of an organic compound gave 1.4439 g of barium sulphate. The percentage of sulphur in the compound is ______ (Nearest integer)
Given: Atomic mass Ba: 137u, S: 32u, O:16u
▶️ Answer/Explanation
= \(\frac{32}{233} × \frac{1.4439}{0.471} × 100\)
= \(\frac{32}{233} × \frac{144.39}{0.471}\)
= \(\frac{4620.48}{109.743} = 42.10\)
≈ 42
How many of the following metal ions have similar value of spin magnetic moment in gaseous state? ______.
Given: Atomic number : V:23; Cr:24; Fe: 26; Ni:28
V\(^{3+}\) , Cr\(^{3+}\) ,Fe\(^{2+}\) , Ni\(^{3+}\)