IB DP Chemistry Mock Exam HL Paper 1A Set 1 - 2025 Syllabus
IB DP Chemistry Mock Exam HL Paper 1A Set 1
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam HL Paper 1A Set 1. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
The diagram shows how protons, neutrons, and electrons behave when they move through an electric field.
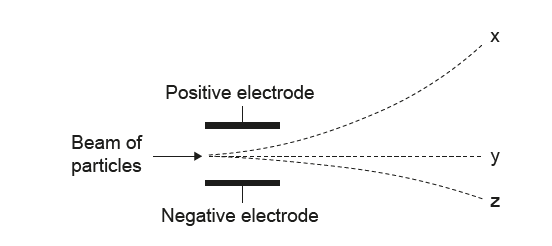

Which option correctly identifies each particle?
| Option | Protons | Electrons | Neutrons |
|---|---|---|---|
| A | y | z | x |
| B | x | y | z |
| C | z | x | y |
| D | x | z | y |
▶️ Answer/Explanation
• The path bending strongly upward (toward the negative electrode) must be protons → z.
• The path bending strongly downward (toward the positive electrode) must be electrons → x.
• The straight path must be neutrons →y
✅ Answer: (C)
• The path bending strongly downward (toward the positive electrode) must be electrons → x.
• The straight path must be neutrons →y
✅ Answer: (C)
Question
Which diagram illustrates the correct ground-state electron configuration of a nitrogen atom?
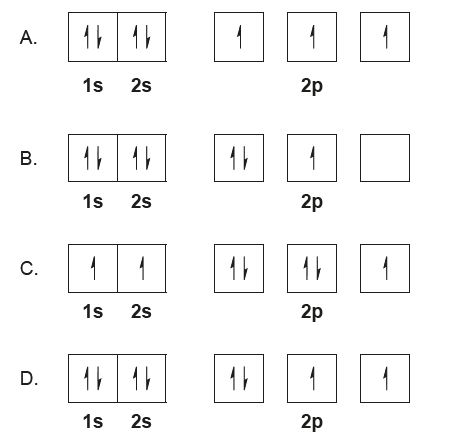
▶️ Answer/Explanation
Detailed solution
Nitrogen (Z=7) electron configuration: 1s² 2s² 2p³
- Hund’s rule: electrons occupy orbitals singly before pairing
- 2p orbitals should have three unpaired electrons
- This matches option (A): 1s: ↑↓, 2s: ↑↓, 2p: ↑, ↑, ↑
✅ Answer: (A)
Question
Which row gives the correct formula and electron configuration of the species present in calcium nitride?
| Formula | Electron configuration of the Ca species | Electron configuration of the N species |
|---|---|---|
| A. Ca2N3 | [Ar] | [Ne] |
| B. Ca2N3 | [Ar] 4s2 | [He] 2s2 2p3 |
| C. Ca2N2 | [Ar] 4s2 | [He] 2s2 2p3 |
| D. Ca2N2 | [Ar] | [Ne] |
▶️ Answer/Explanation
Detailed solution
Calcium nitride:
- Formula: Ca3N2 (Ca2+ and N3−)
- Ca2+: loses 2 electrons → [Ar] configuration (without 4s2)
- N3−: gains 3 electrons → [Ne] configuration (1s2 2s2 2p6)
✅ Answer: (D) – Ca2N2 with [Ar] and [Ne] configurations
Note: While the exact formula should be Ca3N2, among the given options, (D) has the correct electron configurations for the ionic species.
